Abstract
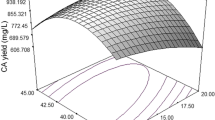
Total of 171 alkaliphilic actinomycetes were evaluated for extracellular RNase production and Streptomyces sp. M49-1 was selected for further experiments. Fermentation optimization for RNase production was implemented in two steps using response surface methodology with central composite design. In the first step, the effect of independent fermentation variables including temperature, initial pH and process time were investigated. After identification of carbon and nitrogen sources affecting the production by one variable at a time method, concentrations of glucose and yeast extract and also inoculum size were chosen for the second central composite design. A maximum RNase activity was obtained under optimal conditions of 4.14 % glucose concentration, 4.63 % yeast extract concentration, 6.7 × 106 spores as inoculum size for 50 ml medium, 42.9 °C, 91.2 h process time and medium initial pH 9.0. Optimum activity of the enzyme is achieved at pH 11 and temperature 60 °C. The enzyme is highly stable at pH range 9.0–12.0 and at 90 °C after 2 h. Statistical optimization experiments provide 2.25 fold increases in the activity of alkalotolerant and thermostable RNase and shortened the fermentation time compared to that of unoptimized condition. The members of Streptomyces can be promising qualified RNase producer for pharmaceutical industries.


Similar content being viewed by others
References
Arnold U (2008) Aspects of the cytotoxic action of ribonucleases. Curr Pharm Biotechnol 9:161–168
Chacko R, Deshpande M, Shankar V (1996) Extracellular ribonuclease production by Rhizopus stolonifer: influence of metal ions. Curr Microbiol 32:246–251
Cheng S-C, Ogrydziak DM (1986) Extracellular RNase produced by Yarrowia lipolytica. J Bacteriol 168:581–589
Elibol M (2004) Optimization of medium composition for actinorhodin production by Streptomyces coelicolor A3(2) with response surface methodology. Process Biochem 39:1057–1062
Fang EF, Ng TB (2011) Ribonucleases of different origins with a wide spectrum of medicinal applications. Biochim Biophys Acta 1815:65–74
Gomes E, De Silva R, Serzedello A (1998) Ribonuclease production by Aspergillus species. Rev Microbiol 29:187–192
Gundampati RK, Debnath M (2009) Extracellular ribonuclease from Aspergillus niger: process optimization for production. Int J Eng Tech (IJET) 1:317–320
Gundampati RK, Sharma A, Kumari M, Debnath M (2011) Extracellular poly (A) specific ribonuclease from Aspergillus niger ATCC 26550: purification, biochemical, and spectroscopic studies. Process Biochem 46:135–141
Haaland PD (1989) Statistical problem solving. In: Haaland PD (ed) Experimental design in biotechnology. Marcel Dekker Incorporation, New York, pp 1–18
Hebert EJ, Grimsley GR, Hartley RW, Horn G, Schell D, Garcia S, Both V, Sevcik J, Pace CN (1997) Purification of ribonucleases Sa, Sa2, and Sa3 after expression in Escherichia coli. Protein Expr Purif 11:162–168
Hlinkova V, Urbanikova L, Krajcikova D, Sevcik J (2001) Purification, crystallization and preliminary x-ray analysis of two crystal forms of ribonuclease Sa3. Acta Cryst D57:737–739
Hole RC, Singhal RS, Melo JS, D’Souza SF (2004) A rapid plate screening technique for extracellular ribonuclease producing strains. BARC Newsletter 249:91–97
Ilinskaya ON, Dreyer F, Mitkevich VA, Shaw KL, Pace CN, Makarov AA (2002) Changing the net charge from negative to positive makes ribonuclease Sa cytotoxic. Protein Sci 11:2522–2525
Joshi AP, Deshmukh SS (2011) Streptomyces nucleases. Crit Rev Microbiol 37:227–236
Kutzner HJ (1981) The family Streptomycetaceae. In: Starr MP, Stolp H, Trüper HG, Balons A, Schlegel HG (eds) The Prokaryotes—A handbook 1 on habitats, isolation and identification of bacteria. Springer, Berlin, pp 2028–2090
Pace CN, Hebert EJ, Shaw KL, Schell D, Botg V, Krajcikova D, Sevcik J, Wilson KS, Dauter Z, Hartley RW, Grimsley GR (1998) Conformational stability and thermodynamics of folding of ribonucleases Sa, Sa2 and Sa3. J Mol Biol 279:271–286
Patil NS, Deshmukh SS, Shankar V (2005) Extracellular nuclease from a thermophile, Streptomyces thermonitrificans: production, purification and partial characterization of—double strand preferential—deoxyribonuclease activity. Process Biochem 40:1271–1278
Reddy LG, Shankar V (1993) Immobilized nucleases. Crit Rev Biotechnol 13:255–273
Sevcik J, Urbanikova L, Leland PA, Raines RT (2002) X-ray structure of two crystalline forms of a Streptomycete ribonuclease with cytotoxic activity. J Biol Chem 277:47325–47330
Shaw KL, Grimsley GR, Yakovlev GI, Makarov AA, Pace CN (2001) The effect of net charge on the solubility, activity, and stability of ribonuclease Sa. Protein Sci 10:1206–1215
Trefethen JM, Pace CN, Scholtz JM, Brem DN (2005) Charge–charge interactions in the denatured state influence the folding kinetics of ribonuclease Sa. Protein Sci 14:1934–1938
Uchida T, Egami F (1967) Ribonuclease T1 from taka-diastase. Methods Enzymol 12(Part A):229–239
Voss C, Lindau D, Flaschel E (2006) Production of recombinant RNase Ba and its application in downstream processing of plasmid DNA for pharmaceutical use. Biotechnol Prog 22:737–744
Xiong Y-H, Liu J-Z, Song H-Y, Ji L-N (2004) Enhanced production of extracellular ribonuclease from Aspergillus niger by optimization of culture conditions using response surface methodology. Biochem Eng J 21:27–32
Yang H–H, Thayer DW, Yang SF (1979) Reduction of endogenous nucleic acid in a single-cell protein. Appl Environ Microbiol 38:143–147
Zhong L, Srirangan K, Scharer J, Moo-Young M, Fenner D, Crossley L, Honeyman CH, Suen S, Chou CP (2011) Developing an RNase –free bioprocess to produce pharmaceutical-grade plasmid DNA using selective precipitation and membrane chromatography. Sep Purif Technol 83:121–129
Zhou W-W, Niu T-G (2009) Purification and some properties of an extracellular ribonuclease with antiviral activity against tobacco mosaic virus from Bacillus cereus. Biotechnol Lett 31:101–105
Zhou W-W, He Y-L, Niu T-G, Zhong J-J (2009) Optimization of fermentation conditions for production of anti-TMV extracellular ribonuclease by Bacillus cereus using response surface methodology. Bioprocess Biosyst Eng 33:657–663
Acknowledgments
This research was supported by The Research Fund of Ege University with 09.MUH.078 project number. The authors would like to thank Professor Murat Elibol (Department of Bioengineering, Faculty of Engineering, Ege University, Izmir, Turkey) for his valuable comments and suggestions during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Demir, T., Gübe, Ö., Yücel, M. et al. Increased alkalotolerant and thermostable ribonuclease (RNase) production from alkaliphilic Streptomyces sp. M49-1 by optimizing the growth conditions using response surface methodology. World J Microbiol Biotechnol 29, 1625–1633 (2013). https://doi.org/10.1007/s11274-013-1325-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1325-1