Abstract
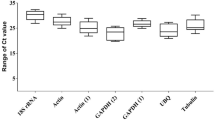
Valsa mali var. mali (Vmm), is the predominant species of apple valsa canker in China. Modern analysis of genes involved in virulence or pathogenicity usually implicate gene expression analysis most often performed using real-time quantitative polymerase chain reaction (RT-qPCR). However, for relative gene expression analysis pertinent reference genes have to be validated before using them as internal reference. This has not been reported for Vmm, so far. Therefore, eight commonly used housekeeping genes (ACT, CYP, EF1-α, G6PDH, GAPDH, L13, TUB, and UBQ) were cloned and evaluated for their expression stability by geNorm and NormFinder. Overall, all of the candidate reference genes were found to be suitable for gene expression analysis. After analysis of 10 samples from different strains and abiotic stress treatments, G6PDH appeared to be the most suitable reference gene, whereas GAPDH was the least suitable. Moreover, taking G6PDH combined with L13 or CYP as reference genes, improved the reliability of RT-qPCR significantly. The influence of the reference system on expression data was demonstrated by analyzing Vmmpg-1 encoding an endo-polygalacturonase gene. Pectinases are considered key pathogenicity factors for this fungus. In order to better understand the role of pectinases in pathogenicity of Vmm, RT-qPCR was used for expression analysis. Our results may provide a guideline for future studies on gene expression of V. mali var. mali by using RT-qPCR.





Similar content being viewed by others
References
Abe K, Kotoda N, Kato H, Soejima J (2007) Resistance sources to valsa canker (Valsa ceratosperma) in a germplasm collection of diverse Malus species. Plant Breeding 126(4):449–453
Altschul Gish SF, Miller W, Myers W, Lipman EW, David J (1990) Basic local alignment search tool. J Mol Biol 215(3):403–410
Andersen CL, Jensen JL, Ørntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64(15):5245
Bohle K, Jungebloud A, Göcke Y, Dalpiaz A, Cordes C, Horn H, Hempel DC (2007) Selection of reference genes for normalisation of specific gene quantification data of Aspergillus niger. J Biotechnol 132(4):353–358
Bustin S (2002) Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J Mol Endocrinol 29(1):23–39
Bustin S, Dorudi S (1998) Molecular assessment of tumour stage and disease recurrence using PCR-based assays. Mol Med Today 4(9):389–396
Expósito-Rodríguez M, Borges A, Borges-Pérez A, Pérez J (2008) Selection of internal control genes for quantitative real-time RT-PCR studies during tomato development process. BMC Plant Biol 8(1):131
Feng H, Huang X, Zhang Q, Wei G, Wang X, Kang Z (2012) Selection of suitable inner reference genes for relative quantification expression of microRNA in wheat. Plant Physiol Biochem 51:116–122
Frohman MA, Dush MK, Martin GR (1988) Rapid production of full-length cDNAs from rare transcripts: amplification using a single gene-specific oligonucleotide primer. Proc Natl Acad Sci USA 85(23):8998–9002
Gachon C, Mingam A, Charrier B (2004) Real-time PCR: what relevance to plant studies? J Exp Bot 55(402):1445–1454
Glare E, Divjak M, Bailey M, Walters E (2002) β-Actin and GAPDH housekeeping gene expression in asthmatic airways is variable and not suitable for normalising mRNA levels. Thorax 57(9):765–770
Hoondal G, Tiwari R, Tewari R, Dahiya N, Beg Q (2002) Microbial alkaline pectinases and their industrial applications: a review. Appl Microbiol Biotechnol 59(4):409–418
Huggett J, Dheda K, Bustin S, Zumla A (2005) Real-time RT-PCR normalisation; strategies and considerations. Genes Immun 6(4):279–284
Ke X, Huang L, Han Q, Gao X, Kang Z (2013) Histological and cytological investigations of the infection and colonization of apple bark by Valsa mali var. mali. Australas Plant Pathol 42(1):85–93
Lee JM, Roche JR, Donaghy DJ, Thrush A, Sathish P (2010) Validation of reference genes for quantitative RT-PCR studies of gene expression in perennial ryegrass (Lolium perenne L.). BMC Mol Biol 11(1):8
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408
Mariño-Ramírez L, Demidenko NV, Logacheva MD, Penin AA (2011) Selection and validation of reference genes for quantitative real-time PCR in buckwheat (Fagopyrum esculentum) based on transcriptome sequence data. PLoS ONE 6(5):e19434
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497
Nailis H, Coenye T, Van Nieuwerburgh F, Deforce D, Nelis HJ (2006) Development and evaluation of different normalization strategies for gene expression studies in Candida albicans biofilms by real-time PCR. BMC Mol Biol 7(1):25
Paolacci AR, Tanzarella OA, Porceddu E, Ciaffi M (2009) Identification and validation of reference genes for quantitative RT-PCR normalization in wheat. BMC Mol Biol 10(1):11
Ruan W, Lai M (2007) Actin, a reliable marker of internal control? Clin Chim Acta 385(1–2):1–5
Sakuma T (1990) Valsa canker. In: Jones AL, Aldwinckle HS (eds) Compendium of apple and pear disease. American Phytopathological Society Press, St Paul, MN, USA, pp 39–40
Steiger MG, Mach RL, Mach-Aigner AR (2010) An accurate normalization strategy for RT-qPCR in Hypocrea jecorina (Trichoderma reesei). J Biotechnol 145(1):30–37
Svircev A, Biggs A, Miles N (1991) Isolation and partial purification of phytotoxins from liquid cultures of Leucostoma cincta and Leucostoma persoonii. Can J Bot 69(9):1998–2003
Tamura O, Saito I (1982) Histopathological changes of apple bark infected by Valsa ceratosperma (Tode ex Fr.) maire during dormant and growing periods. Ann Phytopath Soc Japan 48(4):490–498
Teste M-A, Duquenne M, François JM, Parrou J-L (2009) Validation of reference genes for quantitative expression analysis by real-time RT-PCR in Saccharomyces cerevisiae. BMC Mol Biol 10(1):99
Thellin O, Zorzi W, Lakaye B, De Borman B, Coumans B, Hennen G, Grisar T, Igout A, Heinen E (1999) Housekeeping genes as internal standards: use and limits. J Biotechnol 75(2):291–295
Tong Z, Gao Z, Wang F, Zhou J, Zhang Z (2009) Selection of reliable reference genes for gene expression studies in peach using real-time PCR. BMC Mol Biol 10(1):71
Traquair J (1987) Oxalic acid and calcium oxalate produced by Leucostoma cincta and L. persoonii in culture and in peach bark tissues. Can J Bot 65(9):1952–1956
Udvardi MK, Czechowski T, Scheible WR (2008) Eleven golden rules of quantitative RT-PCR. Plant Cell 20(7):1736–1737
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3(7):research0034
Wang X, Wei J, Huang L, Kang Z (2011) Re-evaluation of pathogens causing valsa canker on apple in China. Mycologia 103(2):317–324
Acknowledgments
This study was financially supported by the National Natural Science Foundation of China (NO.31171796 and NO.31101476), the Program for Agriculture (nyhyzx201203034-03) and the 111 Project (B07049). Special thanks are due to Dr. H. Buchenauer and Dr. Xiaoyu Qiang for correcting the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhiyuan Yin and Xiwang Ke contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yin, Z., Ke, X., Huang, D. et al. Validation of reference genes for gene expression analysis in Valsa mali var. mali using real-time quantitative PCR. World J Microbiol Biotechnol 29, 1563–1571 (2013). https://doi.org/10.1007/s11274-013-1320-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1320-6