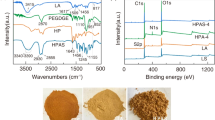
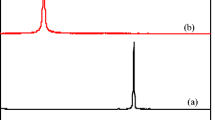
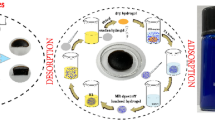
Abstract
Macroscopic utilization of nanomaterial provides a new idea for the research and development of novel adsorbent, which can enhance efficiency in the adsorption and elution process. In this paper, nano-polypyrrole (PPy) was dispersed into two inexpensive and renewable biomass materials, gelatin (Gel) and chitosan (CS), to fabricate a novel photo/electric-sensitive hydrogel, Gel/CS/PPy. The micro-network of Gel/CS/PPy shows a high adsorption rate of 94.2% for acid fuchsine (AF). Furthermore, with the addition of polypyrrole, Gel/CS/PPy has the characteristic of photo/electric response, which can improve the elution efficiency of AF from the adsorbent. The results showed that the elution efficiency could be increased by 4 times with photo-assistance, and about 2 times with electro-assistance. Predictably, using the methods described in this article, high-quality adsorbents can be designed for more organic pollutants.

Graphical abstract









Similar content being viewed by others
References
Barakat, M. A., & Sahiner, N. (2008). Cationic hydrogels for toxic arsenate removal from aqueous environment. Journal of Environmental Management, 88, 955–961. https://doi.org/10.1016/j.jenvman.2007.05.003.
Gornall, J. L., & Terentjev, E. M. (2008). Helix–coil transition of gelatin: helical morphology and stability. Soft Matter, 4, 544–549. https://doi.org/10.1039/B713075A.
Khan, M., & Lo, I. M. (2017). Removal of ionizable aromatic pollutants from contaminated water using nano γ-Fe2O3 based magnetic cationic hydrogel: sorptive performance, magnetic separation and reusability. Journal of Hazardous Materials, 322, 195–204. https://doi.org/10.1016/j.jhazmat.2016.01.051.
Khan, M., & Lo, I. M. (2016). A holistic review of hydrogel applications in the adsorptive removal of aqueous pollutants: recent progress, challenges, and perspectives. Water Research, 106, 259–271. https://doi.org/10.1016/j.watres.2016.10.008.
Lewis, R. W., Bertsch, P. M., & McNear, D. H. (2019). Nanotoxicity of engineered nanomaterials (ENMs) to environmentally relevant beneficial soil bacteria–a critical review. Nanotoxicology, 13, 1–37. https://doi.org/10.1080/17435390.2018.1530391.
Li, S., Xu, J., Yao, G., & Liu, H. (2019a). Self-adhesive, self-healable, and triple-responsive hydrogel doped with polydopamine as an adsorbent toward methylene blue. Industrial & Engineering Chemistry Research, 58, 17075–17087. https://doi.org/10.1021/acs.iecr.9b03359.
Li, C., Yan, Y., Zhang, Q., Zhang, Z., Huang, L., Zhang, J., Xiong, Y., & Tan, S. (2019b). Adsorption of Cd2+ and Ni2+ from aqueous single-metal solutions on graphene oxide-chitosan-poly (vinyl alcohol) hydrogels. Langmuir, 35, 4481–4490. https://doi.org/10.1021/acs.langmuir.8b04189.
Li, Q., Wu, J., Tang, Z., Xiao, Y., Huang, M., & Lin, J. (2010). Application of poly (acrylic acid-g-gelatin)/polypyrrole gel electrolyte in flexible quasi-solid-state dye-sensitized solar cell. Electrochimica Acta, 55, 2777–2781. https://doi.org/10.1016/j.electacta.2009.12.072.
Ozay, O., Ekici, S., Baran, Y., Aktas, N., & Sahiner, N. (2009). Removal of toxic metal ions with magnetic hydrogels. Water Research, 43, 4403–4411. https://doi.org/10.1016/j.watres.2009.06.058.
Pakdel, P. M., & Peighambardoust, S. J. (2018). A review on acrylic based hydrogels and their applications in wastewater treatment. Journal of Environmental Management, 217, 123–143. https://doi.org/10.1016/j.jenvman.2018.03.076.
Qi, X., Liu, R., Chen, M., Li, Z., Qin, T., Qian, Y., Zhao, S., Liu, M., & Shen, J. (2019). Removal of copper ions from water using polysaccharide-constructed hydrogels. Carbohydrate Polymers, 209, 101–110. https://doi.org/10.1016/j.carbpol.2019.01.015.
Satapathy, M., Nyambat, B., Chiang, C. W., Chen, C. H., Wong, P. C., Ho, P. H., Jheng, P. R., Burnouf, T., Tseng, C. L., & Chuang, E. Y. (2018). A gelatin hydrogel-containing nano-organic PEI–Ppy with a photothermal responsive effect for tissue engineering applications. Molecules, 23, 1256. https://doi.org/10.3390/molecules23061256.
Soleimani, K., Tehrani, A. D., & Adeli, M. (2018). Bioconjugated graphene oxide hydrogel as an effective adsorbent for cationic dyes removal. Ecotoxicology and Environmental Safety, 147, 34–42. https://doi.org/10.1016/j.ecoenv.2017.08.021.
Tang, Q., Sun, X., Li, Q., Lin, J., & Wu, J. (2009). Preparation and electrical conductivity of SiO 2/polypyrrole nanocomposite. Journal of Materials Science, 44, 849–854. https://doi.org/10.1007/s10853-008-3137-5.
Wang, X. C., Hao, X. L., He, B. Y., & Qiang, T. T. (2013). Study on preparation and adsorption performance of gelatin microspheres for Cr (VI). In Advanced Materials Research (Vol. 641, pp. 1005–1009). https://doi.org/10.4028/www.scientific.net/AMR.641-642.1005.
Xing, J., Yang, B., Shen, Y., Wang, Z., Wang, F., Shi, X., & Zhang, Z. (2019b). Selective removal of acid fuchsin from aqueous solutions by rapid adsorption onto polypyrrole crosslinked cellulose/gelatin hydrogels. Journal of Dispersion Science and Technology, 40, 1–9. https://doi.org/10.1080/01932691.2018.1518147.
Xing, J., Shen, Y., Yang, B., Feng, D., Wang, W., & Bai, B. (2018a). A green method based on electro-assisted and photo-assisted regeneration for removal of chromium (VI) from aqueous solution. Water Science and Technology, 2017, 896–902. https://doi.org/10.2166/wst.2018.260.
Xing, J., Shen, Y., Dang, W., Yang, B., Fu, W., Wang, W., & Bai, B. (2019a). Fabrication of a photoelectric-sensitive imprinting polymer by PPy-cross-linked Gel/CS complex and its comprehensive treatment of Cr (VI). Polymer Bulletin, 1–14. https://doi.org/10.1007/s00289-019-02780-5.
Xing, J., Zhu, C., Chowdhury, I., Tian, Y., Du, D., & Lin, Y. (2018b). Electrically switched ion exchange based on polypyrrole and carbon nanotube nanocomposite for the removal of chromium (VI) from aqueous solution. Industrial & Engineering Chemistry Research, 57, 768–774. https://doi.org/10.1021/acs.iecr.7b03520.
Yadav, S., Mehrotra, G. K., Bhartiya, P., Singh, A., & Dutta, P. K. (2020). Preparation, physicochemical and biological evaluation of quercetin based chitosan-gelatin film for food packaging. Carbohydrate Polymers, 227, 115348. https://doi.org/10.1016/j.carbpol.2019.115348.
Yu, Z., Zhang, X., & Huang, Y. (2013). Magnetic chitosan–iron (III) hydrogel as a fast and reusable adsorbent for chromium (VI) removal. Industrial & Engineering Chemistry Research, 52, 11956–11966. https://doi.org/10.1021/ie400781n.
Funding
This work was supported by Fundamental Research Funds for the Central Universities of China (NO. 310829161015), Natural Science Basic Research Program of Shaanxi (NO. 2020JM-226), Yulin Municipal Science and Technology Bureau Science Foundation, and the National Training Projects of the University Students’ Innovation and Entrepreneurship Program (NO. S201910710240).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xing, J., Yang, B., Dang, W. et al. Preparation of Photo/Electro-Sensitive Hydrogel and Its Adsorption/Desorption Behavior to Acid Fuchsine. Water Air Soil Pollut 231, 231 (2020). https://doi.org/10.1007/s11270-020-04582-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04582-2