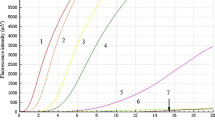
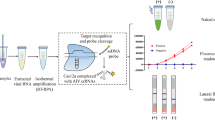
Abstract
Avian influenza virus (AIV) infection can lead to severe economic losses in the poultry industry and causes a serious risk for humans. A rapid and simple test for suspected viral infection cases is crucial. In this study, a reverse transcription recombinase-aided amplification assay (RT-RAA) for the rapid detection of all AIV subtypes was developed. The reaction temperature of the assays is at 39 °C and the detection process can be completed in less than 20 min. The specificity results of the assay showed that this method had no cross-reaction with other main respiratory viruses that affect birds, including Newcastle disease virus (NDV) and infectious bronchitis virus (IBV). The analytical sensitivity at a 95% confidence interval was 102 RNA copies per reaction. In comparison with a published assay for reverse transcription quantitative real-time polymerase chain reaction (RT-qPCR), the κ value of the RT-RAA assay in 384 avian clinical samples was 0.942 (p < 0.001). The sensitivity and specificity of the RT-RAA assay for avian clinical sample detection was determined as 97.59% (95% CI 93.55–99.23%) and 96.79% (95% CI 93.22–98.59%), respectively. The RT-RAA assay for AIV in this study provided an effective and practicable tool for AIV molecular detection.


Similar content being viewed by others
References
Alonso WJ, Yu C, Viboud C, Richard SA, Schuck-Paim C, Simonsen L, Mello WA, Miller MA (2015) A global map of hemispheric influenza vaccine recommendations based on local patterns of viral circulation. Sci Rep 5:17214. https://doi.org/10.1038/srep17214
Chen Y, Chen YF (2014) Global distribution patterns of highly pathogenic H5N1 avian influenza: environmental vs. socioeconomic factors. Comptes Rendus Biologies 337:459–465. https://doi.org/10.1016/j.crvi.2014.06.001
Fouchier R, Munster V, Wallensten A, Bestebroer TM, Herfst S, Smith D, Rimmelzw Aa NGF, Olsen B, Osterhaus AD (2005) Characterization of a novel influenza A virus hemagglutinin subtype (H16) obtained from black-headed gulls. J Virol 79:2814. https://doi.org/10.1128/JVI.79.5.2814-2822.2005
Guo Y, Li J, Cheng X (1999) Discovery of men infected by avian influenza A (H9N2) virus. Zhonghua Shi Yan He Lin Chuang Bing Du Xue Za Zhi 13:105–108
Tong S, Li Y, Rivailler P, Conrardy C, Castillo DA, Chen LM, Recuenco S, Ellison JA, Davis CT, York IA, Turmelle AS, Moran D, Rogers S, Shi M, Tao Y, Weil MR, Tang K, Rowe LA, Sammons S, Xu X, Frace M, Lindblade KA, Cox NJ, Anderson LJ, Rupprecht CE, Donis RO (2012) A distinct lineage of influenza A virus from bats. Proc Natl Acad Sci USA 109:4269–4274. https://doi.org/10.1073/pnas.1116200109
Tong S, Zhu X, Li Y, Shi M, Zhang J, Bourgeois M, Yang H, Chen X, Recuenco S, Gomez J, Chen LM, Johnson A, Tao Y, Dreyfus C, Yu W, McBride R, Carney PJ, Gilbert AT, Chang J, Guo Z, Davis CT, Paulson JC, Stevens J, Rupprecht CE, Holmes EC, Wilson IA, Donis RO (2013) New world bats harbor diverse influenza A viruses. PLoS Pathog 9:e1003657. https://doi.org/10.1371/journal.ppat.1003657
Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y (1992) Evolution and ecology of influenza A viruses. Microbiol Rev 56:152–179. https://doi.org/10.1128/mr.56.1.152-179.1992
Wu H, Peng X, Peng X, Cheng L, Lu X, Jin C, Xie T, Yao H, Wu N (2015) Genetic and molecular characterization of H9N2 and H5 avian influenza viruses from live poultry markets in Zhejiang Province, eastern China. Sci Rep 5:17508. https://doi.org/10.1038/srep17508
Gao R, Cao B, Hu Y, Feng Z, Wang D, Hu W, Chen J, Jie Z, Qiu H, Xu K, Xu X, Lu H, Zhu W, Gao Z, Xiang N, Shen Y, He Z, Gu Y, Zhang Z, Yang Y, Zhao X, Zhou L, Li X, Zou S, Zhang Y, Li X, Yang L, Guo J, Dong J, Li Q, Dong L, Zhu Y, Bai T, Wang S, Hao P, Yang W, Zhang Y, Han J, Yu H, Li D, Gao GF, Wu G, Wang Y, Yuan Z, Shu Y (2013) Human infection with a novel avian-origin influenza A (H7N9) virus. N Engl J Med 368:1888–1897. https://doi.org/10.1056/NEJMoa1304459
Novel Swine-Origin Influenza AVIT, Dawood FS, Jain S, Finelli L, Shaw MW, Lindstrom S, Garten RJ, Gubareva LV, Xu X, Bridges CB, Uyeki TM (2009) Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 360:2605–2615. https://doi.org/10.1056/NEJMoa0903810
Uyeki TM, Cox NJ (2013) Global concerns regarding novel influenza A (H7N9) virus infections. N Engl J Med 368:1862. https://doi.org/10.1056/NEJMp1304661
Li YT, Linster M, Mendenhall IH, Su YCF, Smith GJD (2019) Avian influenza viruses in humans: lessons from past outbreaks. Br Med Bull 132:81–95. https://doi.org/10.1093/bmb/ldz036
Chan PK (2002) Outbreak of avian influenza A(H5N1) virus infection in Hong Kong in 1997. Clin Infect Dis 34(Suppl 2):S58-64. https://doi.org/10.1086/338820
Chen Y, Liang W, Yang S, Wu N, Gao H, Sheng J, Yao H, Wo J, Fang Q, Cui D, Li Y, Yao X, Zhang Y, Wu H, Zheng S, Diao H, Xia S, Zhang Y, Chan KH, Tsoi HW, Teng JL, Song W, Wang P, Lau SY, Zheng M, Chan JF, To KK, Chen H, Li L, Yuen KY (2013) Human infections with the emerging avian influenza A H7N9 virus from wet market poultry: clinical analysis and characterisation of viral genome. Lancet 381:1916–1925. https://doi.org/10.1016/S0140-6736(13)60903-4
Chen H, Yuan H, Gao R, Zhang J, Wang D, Xiong Y, Fan G, Yang F, Li X, Zhou J, Zou S, Yang L, Chen T, Dong L, Bo H, Zhao X, Zhang Y, Lan Y, Bai T, Dong J, Li Q, Wang S, Zhang Y, Li H, Gong T, Shi Y, Ni X, Li J, Zhou J, Fan J, Wu J, Zhou X, Hu M, Wan J, Yang W, Li D, Wu G, Feng Z, Gao GF, Wang Y, Jin Q, Liu M, Shu Y (2014) Clinical and epidemiological characteristics of a fatal case of avian influenza A H10N8 virus infection: a descriptive study. Lancet 383:714–721. https://doi.org/10.1016/S0140-6736(14)60111-2
Koopmans M, Wilbrink B, Conyn M, Natrop G, van der Nat H, Vennema H, Meijer A, van Steenbergen J, Fouchier R, Osterhaus A, Bosman A (2004) Transmission of H7N7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet 363:587–593. https://doi.org/10.1016/S0140-6736(04)15589-X
Kudo K, Binh NG, Manabe T, Co DX, Tuan ND, Izumi S, Takasaki J, Minh DH, Thuy PT, Van VT, Hanh TT, Chau NQ (2012) Clinical preparedness for severe pneumonia with highly pathogenic avian influenza A (H5N1): experiences with cases in Vietnam. Respir Investig 50:140–150. https://doi.org/10.1016/j.resinv.2012.08.005
Ostrowsky B, Huang A, Terry W, Anton D, Brunagel B, Traynor L, Abid S, Johnson G, Kacica M, Katz J, Edwards L, Lindstrom S, Klimov A, Uyeki TM (2012) Low pathogenic avian influenza A (H7N2) virus infection in immunocompromised adult, New York, USA, 2003. Emerg Infect Dis 18:1128–1131. https://doi.org/10.3201/eid1807.111913
Widdowson MA, Bresee JS, Jernigan DB (2017) The global threat of animal influenza viruses of zoonotic concern: then and now. J Infect Dis 216:S493–S498. https://doi.org/10.1093/infdis/jix331
Nachbagauer R, Krammer F (2017) Universal influenza virus vaccines and therapeutic antibodies. Clin Microbiol Infect 23:222–228. https://doi.org/10.1016/j.cmi.2017.02.009
Zheng M, Luo J, Chen Z (2014) Development of universal influenza vaccines based on influenza virus M and NP genes. Infection 42:251–262. https://doi.org/10.1007/s15010-013-0546-4
Jiang WM, Liu S, Chen J, Hou GY, Li JP, Cao YF, Zhuang QY, Li Y, Huang BX, Chen JM (2010) Molecular epidemiological surveys of H5 subtype highly pathogenic avian influenza viruses in poultry in China during 2007–2009. J Gen Virol 91:2491–2496. https://doi.org/10.1099/vir.0.023168-0
Starick E, Werner O (2003) Detection of H7 avian influenza virus directly from poultry specimens. Avian Dis 47:1187–1189. https://doi.org/10.1637/0005-2086-47.s3.1187
Spackman E (2014) Avian influenza virus detection and quantitation by real-time RT-PCR. Methods Mol Biol 1161:105–118. https://doi.org/10.1007/978-1-4939-0758-8_10
Xie Z, Pang YS, Liu J, Deng X, Tang X, Sun J, Khan MI (2006) A multiplex RT-PCR for detection of type A influenza virus and differentiation of avian H5, H7, and H9 hemagglutinin subtypes. Mol Cell Probes 20:245–249. https://doi.org/10.1016/j.mcp.2006.01.003
Lutz S, Weber P, Focke M, Faltin B, Hoffmann J, Muller C, Mark D, Roth G, Munday P, Armes N, Piepenburg O, Zengerle R, von Stetten F (2010) Microfluidic lab-on-a-foil for nucleic acid analysis based on isothermal recombinase polymerase amplification (RPA). Lab Chip 10:887–893. https://doi.org/10.1039/b921140c
Shen XX, Qiu FZ, Shen LP, Yan TF, Zhao MC, Qi JJ, Chen C, Zhao L, Wang L, Feng ZS (2019) A rapid and sensitive recombinase aided amplification assay to detect hepatitis B virus without DNA extraction. BMC Infect Dis 19:1–5. https://doi.org/10.1186/s12879-019-3814-9
Rychlik W (2007) OLIGO 7 primer analysis software. Methods Mol Biol 402:35–60. https://doi.org/10.1007/978-1-59745-528-2_2
Das A, Spackman E, Senne D, Pedersen J, Suarez DL (2007) Development of an internal positive control for rapid diagnosis of avian influenza virus infections by real-time reverse transcription. J Clin Microbiol 44:3065–3073. https://doi.org/10.1128/JCM.00639-06
Organization WH (2017) WHO information for the molecular detection of influenza viruses
Funding
This work was supported by the National Key Research and Development Program of China [2017YFC120050].
Author information
Authors and Affiliations
Contributions
KW, SW, QZ, NJ and FZ: contributed to experiment setting, data analysis, writing of the original draft; SW, QZ, KW, NJ, FZ, YL and XY: contributed to design of the experiments and supervision and writing; SW, QZ, NJ, FZ, YL, XY, JL, GH, LY, QC and RZ: contributed to data collection and analysis; QZ, SW, KW, NJ and FZ: contributed to experiment support and writing; SW, QZ, KW, FS and ZP: contributed to provided comments on the data and manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Edited by Juergen Richt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, S., Zhuang, Q., Jiang, N. et al. Reverse transcription recombinase-aided amplification assay for avian influenza virus. Virus Genes 59, 410–416 (2023). https://doi.org/10.1007/s11262-023-01979-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-023-01979-z