Abstract
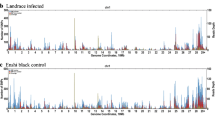
The host genetic makeup plays a significant role in causing the within-breed variation among individuals after vaccination. The present study was undertaken to elucidate the genetic basis of differential immune response between high and low responder Landlly (Landrace X Ghurrah) piglets vis-à-vis CSF vaccination. For the purpose, E2 antibody response against CSF vaccination was estimated in sampled animals on the day of vaccination and 21-day post-vaccination as a measure of humoral immune response. Double-digestion restriction associated DNA (ddRAD) sequencing was undertaken on 96 randomly chosen Landlly piglets using Illumina HiSeq platform. SNP markers were called using standard methodology. Genome-wide association study (GWAS) was undertaken in PLINK program to identify the informative SNP markers significantly associated with differential immune response. The results revealed significant SNPs associated with E2 antibody response against CSF vaccination. The genome-wide informative SNPs for the humoral immune response against CSF vaccination were located on SSC10, SSC17, SSC9, SSC2, SSC3 and SSC6. The overlapping and flanking genes (500Kb upstream and downstream) of significant SNPs were CYB5R1, PCMTD2, WT1, IL9R, CD101, TMEM64, TLR6, PIGG, ADIPOR1, PRSS37, EIF3M, and DNAJC24. Functional enrichment and annotation analysis were undertaken for these genes in order to gain maximum insights into the association of these genes with immune system functionality in pigs. The genetic makeup was associated with differential immune response against CSF vaccination in Landlly piglets while the identified informative SNPs may be used as suitable markers for determining variation in host immune response against CSF vaccination in pigs.





Similar content being viewed by others
Data Availability
The data in the current study are available from the corresponding author upon reasonable request.
References
Al Adhami H, Bardet AF, Dumas M, Cleroux E, Guibert S, Fauque P, Acloque H, Weber M (2022) A comparative methylome analysis reveals the conservation and divergence of DNA methylation patterns and functions in vertebrates. BMC Biol 20(1):1–8. https://doi.org/10.1186/s12915-022-01270-x
Ardouin T, Briquet-Duhazé S, Annoot E (2017) Le champ de la formation et de la professionnalisation des adultes: Attentes sociales, pratiques, lexique et postures identitaires. E-book 302p. http://digital.casalini.it/9782140043802
Baeza MC, Corva PM, Soria LA, Rincon G, Medrano JF, Pavan E, Villarreal EL, Schor A, Melucci L, Mezzadra C, Miquel MC (2011) Genetic markers of body composition and carcass quality in grazing Brangus steers. Genet Mol Res 10(4):3146–3156. https://doi.org/10.4238/2011.December.19.3
Bai X, Yang T, Putz AM, Wang Z, Li C, Fortin F, Harding JCS, Dyck MK, Dekkers JCM, Field CJ, Plastow GS, Vandenbroek D, DeVries B, Dion N, Blanchette S, Rathje T, Duggan M, Kemp R, Charagu P, Mathur P (2021) Investigating the genetic architecture of Disease resilience in pigs by genome-wide association studies of complete blood count traits collected from a natural Disease challenge model. BMC Genomics 22(1):1–5. https://doi.org/10.1186/s12864-021-07835-4
Beiting DP, Hidano S, Baggs JE, Geskes JM, Fang Q, Wherry EJ, Hunter CA, Roos DS, Cherry S (2015) The orphan nuclear receptor TLX is an enhancer of STAT1-mediated transcription and immunity to Toxoplasma Gondii. PLoS Biol 13(7):e1002200. https://doi.org/10.1371/journal.pbio.1002200
Bharati J, Mohan NH, Kumar S, Gogoi J, Kumar S, Jose B, Punetha M, Borah S, Kumar A, Sarkar M (2021) Transcriptome profiling of different developmental stages of corpus luteum during the estrous cycle in pigs. Genomics 113(1):366–379. https://doi.org/10.1016/j.ygeno.2020.12.008
Bharati J, De K, Paul S, Kumar S, Yadav AK, Doley J, Mohan NH, Das BC (2022) Mobilizing pig resources for capacity development and livelihood security, in agriculture, livestock production and aquaculture. Springer, pp 219–242. https://doi.org/10.1007/978-3-030-93262-6_12
Blome S, Staubach C, Henke J, Carlson J, Beer M (2017) Classical swine Fever—an updated review. Viruses 9(4):86. https://doi.org/10.3390/v9040086
Cabrera-Ortega AA, Feinberg D, Liang Y, Rossa C, Graves DT (2017) The role of forkhead box 1 (FOXO1) in the immune system: dendritic cells, T cells, B cells, and hematopoietic stem cells. Crit Rev Immunol 37(1):1–13. https://doi.org/10.1615/CritRevImmunol.2017019636
Caubit X, Gubellini P, Andrieux J, Roubertoux PL, Metwaly M, Jacq B, Fatmi A, Had-Aissouni L, Kwan KY, Salin P, Carlier M, Liedén A, Rudd E, Shinawi M, Vincent-Delorme C, Cuisset JM, Lemaitre MP, Abderrehamane F, Duban B, Lemaitre JF, Woolf AS, Bockenhauer D, Severac D, Dubois E, Zhu Y, Sestan N, Garratt AN, Goff K-L, Fasano L (2016) L TSHZ3 deletion causes an autism syndrome and defects in cortical projection neurons. Nat Genet 48. https://doi.org/10.1038/ng.3681
Chokas AL, Bickford JS, Barilovits SJ, Rogers RJ, Qiu X, Newsom KJ, Beachy DE, Nick HS (2014) A TEAD1/p65 complex regulates the eutherian-conserved MnSOD intronic enhancer, eRNA transcription and the innate immune response. Biochim Biophys Acta - Gene Regul Mech 1839. https://doi.org/10.1016/j.bbagrm.2014.06.012
Coronado L, Perera CL, Rios L, Frías MT, Pérez LJ (2021) A critical review about different vaccines against classical swine Fever virus and their repercussions in endemic regions. Vaccines. https://doi.org/10.3390/vaccines9020154.
Dewulf J, Laevens H, Koenen F, Mintiens K, De Kruif A (2001) An experimental Infection with classical swine Fever virus in pregnant sows: transmission of the virus, course of the Disease, antibody response, and effect on gestation. J Vet Med Ser B 48(8):583–591. https://doi.org/10.1046/j.1439-0450.2001.00467.x
Dong B, Liang J, Li D, Song W, Song J, Zhu M, Zhao S, Ma Y, Yang T (2021) Identification of a prognostic signature Associated with the Homeobox Gene family for Bladder Cancer. Front Mol Biosci 8. https://doi.org/10.3389/fmolb.2021.688298
Edwards S, Fukusho A, Lefèvre PC, Lipowski A, Pejsak Z, Roehe P, Westergaard J (2000) Classical swine Fever: the global situation. Vet Microbiol 73(2–3):103–119. https://doi.org/10.1016/S0378-1135(00)00138-3
Fritzemeier J, Teuffert J, Greiser-Wilke I, Staubach C, Schlüter H, Moennig V (2000) Epidemiology of classical swine Fever in Germany in the 1990s. Vet Microbiol 77(1–2):29–41. https://doi.org/10.1016/S0378-1135(00)00254-6
Garrison E, Marth G (2012) Haplotype-based variant detection from short-read sequencing. arXiv preprint. https://doi.org/10.48550/arXiv.1207.3907.
Gogarten SM, Bhangale T, Conomos MP, Laurie CA, McHugh CP, Painter I, Zheng X, Crosslin DR, Levine D, Lumley T, Nelson SC, Rice K, Shen J, Swarnkar R, Weir BS, Laurie CC (2012) GWASTools: an R/Bioconductor package for quality control and analysis of genome-wide association studies. Bioinformatics 28(24):3329–3331. https://doi.org/10.1093/bioinformatics/bts610
Gong YF, Lu X, Wang ZP, Hu F, Luo YR, Cai SQ, Qi CM, Li S, Niu XY, Qiu XT, Zeng J, Zhang Q (2010) Detection of quantitative trait loci affecting hematological traits in swine via genome scanning. BMC Genet 11. https://doi.org/10.1186/1471-2156-11-56
Gopi B, Singh RV, Kumar S, Kumar S, Chauhan A, Kumar A, Singh SV (2020) Single-nucleotide polymorphisms in CLEC7A, CD209, and TLR4 gene and their association with susceptibility to paratuberculosis in Indian cattle. J Genet 99(1):14. https://doi.org/10.1007/s12041-019-1172-4
Haller O, Staeheli P, Kochs G (2007) Interferon-induced Mx proteins in antiviral host defense. Biochimie. https://doi.org/10.1016/j.biochi.2007.04.015.
Hickmann FMW, Braccini Neto J, Kramer LM, Huang Y, Gray KA, Dekkers JCM, Sanglard LP, Serão NVL (2021) Host Genetics of Response to Porcine Reproductive and respiratory syndrome in sows: antibody response as an Indicator Trait for Improved Reproductive performance. Front Genet 12. https://doi.org/10.3389/fgene.2021.707873
Holmquist E, Okroj M, Nodin B, Jirström K, Blom AM (2013) Sushi domain-containing protein 4 (SUSD4) inhibits complement by disrupting the formation of the classical C3 convertase. FASEB J 27. https://doi.org/10.1096/fj.12-222042
Hulst MM, Westra DF, Wensvoort G, Moormann RJ (1993) Glycoprotein E1 of hog Cholera virus expressed in insect cells protects swine from hog Cholera. J Virol 67. https://doi.org/10.1128/jvi.67.9.5435-5442.1993
Isken O, Postel A, Bruhn B, Lattwein E, Becher P, Tautz N (2019) CRISPR/Cas9-Mediated knockout of DNAJC14 verifies this chaperone as a pivotal host factor for RNA replication of Pestiviruses. J Virol 93. https://doi.org/10.1128/jvi.01714-18
Karthikeyan A, Kumar A, Chaudhary R, Wara AB, Singh A, Sahoo NR, Baqir M, Mishra BP (2020) Genome-wide association study of birth weight and pre-weaning body weight of crossbred pigs. Indian J Anim Sci 90(2):195–200
Kroczek RA, Henn V (2012) The role of XCR1 and its ligand XCL1 in antigen cross-presentation by murine and human dendritic cells. Front Immunol 3:14. https://doi.org/10.3389/fimmu.2012.00014
Kumar S, Kumar S, Singh RV, Chauhan A, Kumar A, Sulabh S, Bharati J, Singh SV (2019) Genetic association of polymorphisms in bovine TLR2 and TLR4 genes with Mycobacterium avium subspecies paratuberculosis Infection in Indian cattle population. Vet Res Commun. https://doi.org/10.1007/s11259-019-09750-2
Kumar S, Kumar A, Bharati J, Kumari S, Banik S, Das PJ, Panigrahi M, Bhushan B (2022) Network analysis of key genes regulating humoral immune response against live attenuated C-strain CSF virus immunization in Landlly pigs. Pharma Innov J 11:3744–3750
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25(14):1754–1760
Li H, Rostami A (2010) IL-9: basic biology, signaling pathways in CD4 + T cells and implications for autoimmunity. J Neuroimmune Pharmacol 5(2):198–209. https://doi.org/10.1007/s11481-009-9186-y
Li Y, Jin Q, Ding P, Zhou W, Chai Y, Li X, Wang Y, Zhang G (2020) Gold nanoparticles enhance immune responses in mice against recombinant classical swine Fever virus E2 protein. Biotechnol Lett 42. https://doi.org/10.1007/s10529-020-02853-w
Li F, Li B, Niu X, Chen W, Li Y, Wu K, Li X, Ding H, Zhao M, Chen J, Yi L (2022) The development of classical swine Fever marker vaccines in recent years. Vaccines 10(4):603. https://doi.org/10.3390/vaccines10040603
Liu X, Wang X, Wang Q, Luo M, Guo H, Gong W, Tu C, Sun J (2018) The eukaryotic translation initiation factor 3 subunit E binds to classical swine Fever virus NS5A and facilitates viral replication. Virology 515. https://doi.org/10.1016/j.virol.2017.11.019
Magnusson U, Bossé J, Mallard BA, Rosendal S, Wilkie BN (1997) Antibody response to Actinobacillus pleuropneumoniae antigens after vaccination of pigs bred for high and low immune response. Vaccine 15. https://doi.org/10.1016/S0264-410X(96)00294-0
Malik YS, Bhat S, Vinodh Kumar OR, Yadav AK, Sircar S, Ansari MI, Sarma DK, Rajkhowa TK, Ghosh S, Dhama K (2020) Classical swine Fever virus biology, clinicopathology, diagnosis, vaccines and a meta-analysis of prevalence: a review. Pathogens 9(6):500. https://doi.org/10.3390/pathogens9060500
Mehrotra A, Bhushan B, Kumar A, Panigrahi M, Karthikeyan A, Singh A, Tiwari AK, Pausch H, Dutt T, Mishra BP (2021) A 1.6 mb region on SSC2 is associated with antibody response to classical swine Fever vaccination in a mixed pig population. Anim Biotechnol. https://doi.org/10.1080/10495398.2021.1873145
Mi H, Muruganujan A, Huang X, Ebert D, Mills C, Guo X, Thomas PD (2019) Protocol update for large-scale genome and gene function analysis with the PANTHER classification system (v.14.0). Nat Protoc 14. https://doi.org/10.1038/s41596-019-0128-8.
Michael H, Li Y, Wang Y, McCusker CT (2021) Trained immunity induced by in vivo peptide-based STAT6 inhibition prevents ragweed allergy in mice. Allergy Asthma Clin Immunol 17. https://doi.org/10.1186/s13223-021-00542-5
Mitchell HD, Eisfeld AJ, Sims AC, McDermott JE, Matzke MM, Webb-Robertson BJM, Tilton SC, Tchitchek N, Josset L, Li C, Ellis AL, Chang JH, Heegel RA, Luna ML, Schepmoes AA, Shukla AK, Metz TO, Neumann G, Benecke AG, Smith RD, Baric RS, Kawaoka Y, Katze MG, Waters KM (2013) A Network Integration Approach to predict conserved regulators related to pathogenicity of Influenza and SARS-CoV respiratory viruses. PLoS ONE 8. https://doi.org/10.1371/journal.pone.0069374
Mkize N, Maiwashe A, Dzama K, Dube B, Mapholi N (2021) Suitability of GWAS as a Tool to Discover SNPs Associated with Tick Resistance in cattle: a review. Pathogens 10(12):1604. https://doi.org/10.3390/pathogens10121604
Moennig V, Floegel-Niesmann G, Greiser-Wilke I (2003) Clinical signs and epidemiology of classical swine Fever: a review of new knowledge. Vet J 165(1):11–20. https://doi.org/10.1016/S1090-0233(02)00112-0
Nicita F, Sabatini L, Alesi V, Lucignani G, Sallicandro E, Sferra A, Bertini E, Zanni G, Palumbo G (2022) Neurological and neuroimaging features of CYB5R3-Related recessive Hereditary Methemoglobinemia type II. Brain Sci 12. https://doi.org/10.3390/brainsci12020182
Okamura T, Onodera W, Tayama T, Kadowaki H, Kojima-Shibata C, Suzuki E, Uemoto Y, Mikawa S, Hayashi T, Awata T, Fujishima-Kanaya N, Mikawa A, Uenishi H, Suzuki K (2012) A genome-wide scan for quantitative trait loci affecting Respiratory Disease and immune capacity in Landrace pigs. Anim Genet 43. https://doi.org/10.1111/j.1365-2052.2012.02359.x
Pathak SK, Kumar A, Bhuwana G, Sah V, Upmanyu V, Tiwari AK, Sahoo AP, Sahoo AR, Wani SA, Panigrahi M, Sahoo NR, Kumar R (2017a) RNA seq analysis for transcriptome profiling in response to classical swine Fever vaccination in indigenous and crossbred pigs. Funct Integer Genomics 17:607–620
Pathak SK, Sah V, Sailo L, Chaudhary R, Singh A, Kumar R, Kumar A (2017b) Expression profiling of immune genes in classical swine Fever vaccinated indigenous and crossbred piglets. Indian J Anim Sci 87(10):1184–1189
Petersen A, Alvarez C, DeClaire S, Tintle NL (2013) Assessing methods for assigning SNPs to genes in gene-based tests of Association using common variants. PLoS ONE 8(5):e62161. https://doi.org/10.1371/journal.pone.0062161
Pockley AG (2003) Heat shock proteins as regulators of the immune response. Lancet 362(9382):0–476. https://doi.org/10.1016/S0140-6736(03)14075-5
Puritz JB, Hollenbeck CM, Gold JR (2014) dDocent: a RADseq, variant-calling pipeline designed for population of non-model organisms. PeerJ 2:e431. https://doi.org/10.7717/peerj.431
Rajkhowa S, Banik S, Mohan NH, Barman K, Das PJ, Kumar S, Kumar S (2018) Annual Report of AICRP on Pig 2018
Rasmussen AL, Okumura A, Ferris MT, Green R, Feldmann F, Kelly SM, Scott DP, Safronetz D, Haddock E, LaCasse R, Thomas MJ, Sova P, Carter VS, Weiss JM, Miller DR, Shaw GD, Korth MJ, Heise MT, Baric RS, De Villena FPM, Feldmann H, Katze MG (2014) Host genetic diversity enables Ebola hemorrhagic Fever pathogenesis and resistance. Science 346(6212):987–991. https://doi.org/10.1126/science.1259595
Reiner G, Melchinger E, Kramarova M, Pfaff E, Büttner M, Saalmüller A, Geldermann H (2002) Detection of quantitative trait loci for resistance/susceptibility to pseudorabies virus in swine. J Gen Virol 83. https://doi.org/10.1099/0022-1317-83-1-167
Reiner G, Kliemt D, Willems H, Berge T, Fischer R, Köhler F, Hepp S, Hertrampf B, Daugschies A, Geldermann H, Mackenstedt U, Zahner H (2007) Mapping of quantitative trait loci affecting resistance/susceptibility to Sarcocystis miescheriana in swine. Genomics 89. https://doi.org/10.1016/j.ygeno.2007.01.011
Reiner G, Fischer R, Hepp S, Berge T, Köhler F, Willems H (2008) Quantitative trait loci for white blood cell numbers in swine. Anim Genet 39. https://doi.org/10.1111/j.1365-2052.2008.01700.x
Richert-Spuhler LE, Mar CM, Shinde P, Wu F, Hong T, Greene E, Hou S, Thomas K, Gottardo R, Mugo N, de Bruyn G, Celum C, Baeten JM, Lingappa JR, Lund JM, Wald A, Campbell MS, Corey L, Coombs RW, Hughes JP, Magaret A, McElrath MJ, Morrow R, Mullins JI, Coetzee D, Fife K, Were E, Essex M, Makhema J, Katabira E, Ronald A, Bukusi E, Cohen C, Kapiga S, Manongi R, Farquhar C, John-Stewart G, Kiarie J, Delany-Moretlwe S, Rees H, Gray G, McIntyre J, Mugo NR, Donnell D, Frenkel L, Hendrix CW, Tumwesigye E, Ndase P, Bukusi Eliabeth Wangisi J, Campbell J, Tappero J (2021) CD101 genetic variants modify regulatory and conventional T cell phenotypes and functions. Cell Rep Med 2. https://doi.org/10.1016/j.xcrm.2021.100322
Rossi S, Staubach C, Blome S, Guberti V, Thulke HH, Vos A, Koenen F, Le Potier MF (2015) Controlling of CSFV in European wild boar using oral vaccination: a review front Microbiol. https://doi.org/10.3389/fmicb.2015.01141
Rothschild MF, Hu ZL, Jiang Z (2007) Advances in QTL mapping in pigs. Int J Biol Sci 3(3):192–197. https://doi.org/10.7150/ijbs.3.192
Rümenapf T, Stark R, Meyers G, Thiel HJ (1991) Structural proteins of hog Cholera virus expressed by Vaccinia virus: further characterization and induction of protective immunity. J Virol 65. https://doi.org/10.1128/jvi.65.2.589-597.1991
Sah V, Kumar Amit, Dhar P, Upmanyu V, Tiwari AK, Wani SA, Sahu AR, Kumar Ajay, Badasara SK, Pandey A, Saxena S, Rai A, Mishra BP, Singh RK, Gandham RK (2020) Signature of genome-wide gene expression in classical swine Fever virus-infected macrophages and PBMCs of indigenous vis-a-vis crossbred pigs. Gene 731. https://doi.org/10.1016/j.gene.2020.144356
Sailo L, Kumar A, Sah V, Chaudhary R, Upmanyu V, Tiwari AK, Kumar Ajay, Pandey A, Saxena S, Singh A, Wani SA, Gandham RK, Rai A, Mishra BP, Singh RK (2019) Genome-wide integrated analysis of miRNA and mRNA expression profiles to identify differentially expressed miR-22-5p and miR-27b-5p in response to classical swine fever vaccine virus. Funct Integr Genomics 19, 901–918 (2019). https://doi.org/10.1007/s10142-019-00689-w
Sambrook J, Russel D (2001) Molecular cloning- A laboratory manual, 3rd edition ed Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Sanglard LP, Fernando RL, Gray KA, Linhares DCL, Dekkers JCM, Niederwerder MC, Serão NVL (2020) Genetic Analysis of Antibody Response to Porcine Reproductive and Respiratory Syndrome Vaccination as an Indicator Trait for Reproductive performance in commercial sows. Front Genet 11 https://doi.org/10.3389/fgene.2020.01011.
Scanlan CL, Putz AM, Gray KA, Serão NVL (2019) Genetic analysis of reproductive performance in sows during porcine reproductive and respiratory syndrome (PRRS) and porcine epidemic diarrhea (PED) outbreaks. J Anim Sci Biotechnol 10. https://doi.org/10.1186/s40104-019-0330-0
Scholz H, Kirschner KM (2005) A role for the Wilms’ Tumor protein WT1 in organ development. Physiology 20(1):54–59. https://doi.org/10.1152/physiol.00048.2004
Serão NVL, Matika O, Kemp RA, Harding JCS, Bishop SC, Plastow GS, Dekkers JCM (2014) Genetic analysis of reproductive traits and antibody response in a PRRS outbreak herd. J Anim Sci. https://doi.org/10.2527/jas.2014-7821
Singh A, Kumar A, Sahoo NR, Upmanyu V, Kumar B, Bhushan B, Sharma D (2016) Ssociation of humoral response to classical swine Fever vaccination with single nucleotide polymorphisms of swine leukocyte antigens. J Appl Anim Res 44. https://doi.org/10.1080/09712119.2015.1013965
Singh VK, Rajak KK, Kumar A, Yadav SK (2018) Classical swine Fever in India: current status and future perspective. Trop Anim Health Prod 50:1181–1189. https://doi.org/10.1007/s11250-018-1608-5.
Sørensen KK, Gregersen VR, Christensen OF, Velander IH, Bendixen C (2011) Genomic regions associated with ventro-cranial chronic pleuritis in pigs. J Anim Breed Genet 128. https://doi.org/10.1111/j.1439-0388.2010.00914.x
Suradhat S, Intrakamhaeng M, Damrongwatanapokin S (2001) The correlation of virus-specific interferon-gamma production and protection against classical swine Fever virus Infection. Vet Immunol Immunopathol 83. https://doi.org/10.1016/S0165-2427(01)00389-0
Tan C, Wu Z, Ren J, Huang Z, Liu D, He X, Prakapenka D, Zhang R, Li N, Da Y, Hu X (2017) Genome-wide association study and accuracy of genomic prediction for teat number in Duroc pigs using genotyping-by-sequencing. Genet Sel Evol 49. https://doi.org/10.1186/s12711-017-0311-8
Thissen D, Steinberg L, Kuang D (2002) Quick and easy implementation of the Benjamini-Hochberg procedure for controlling the false positive rate in multiple comparisons. J Educational Behav Stat 27(1):77–83
Uddin MJ, Cinar MU, Große-Brinkhaus C, Tesfaye D, Tholen E, Juengst H, Looft C, Wimmers K, Phatsara C, Schellander K (2011) Mapping quantitative trait loci for innate immune response in the pig. Int J Immunogenet 38. https://doi.org/10.1111/j.1744-313X.2010.00985.x
Uemoto Y, Ichinoseki K, Matsumoto T, Oka N, Takamori H, Kadowaki H, Kojima-Shibata C, Suzuki E, Okamura T, Aso H, Kitazawa H, Satoh M, Uenishi H, Suzuki K (2021) Genome-wide association studies for production, Respiratory Disease, and immune-related traits in Landrace pigs. Sci Rep 11. https://doi.org/10.1038/s41598-021-95339-2
Van Oirschot JT (2003) Vaccinology of classical swine fever: From lab to field, in Vet Microbiol. https://doi.org/10.1016/j.vetmic.2003.09.008
Vohra V, Chhotaray S, Gowane G, Alex R, Mukherjee A, Verma A, Deb SM (2021) Genome-Wide Association Studies in Indian Buffalo revealed genomic regions for Lactation and Fertility. Front Genet 12. https://doi.org/10.3389/fgene.2021.696109
Wan J, Wu Y, Ji X, Huang L, Cai W, Su Z, Wang S, Xu H (2020) IL-9 and IL-9-producing cells in Tumor immunity. Cell Commun Signal 18(1):50. https://doi.org/10.1186/s12964-020-00538-5
Wara AB, Kumar A, Singh A, Karthikeyan AK, Dutt T, Mishra BP (2019) Genome-wide association study of test day’s and 305 days milk yield in crossbred cattle. Indian J Anim Sci 89:861–865
Wimmers K, Murani E, Schellander K, Ponsuksili S (2009) QTL for traits related to humoral immune response estimated from data of a porcine F2 resource population. Int J Immunogenet 36. https://doi.org/10.1111/j.1744-313X.2009.00838.x
Woischke C, Blaj C, Schmidt EM, Lamprecht S, Engel J, Hermeking H, Kirchner T, Horst D (2016) CYB5R1 links epithelial-mesenchymal transition and poor prognosis in Colorectal cancer. Oncotarget 7. https://doi.org/10.18632/oncotarget.8912
Wu T, Ren MX, Chen GP, Jin ZM, Wang G (2016) Rrp15 affects the cell cycle, proliferation, and apoptosis in NIH3T3 cells. FEBS Open Bio 6. https://doi.org/10.1002/2211-5463.12128
Zhang G, Nebert DW (2017) Personalized medicine: genetic risk prediction of drug response. Pharmacol Ther S 0163–7258(17):30050. https://doi.org/10.1016/j.pharmthera.2017.02.036
Zimin AV, Delcher AL, Florea L, Kelley DR, Schatz MC, Puiu D, Hanrahan F, Pertea G, Van Tassell CP, Sonstegard TS, Marçais G, Roberts M, Subramanian P, Yorke JA, Salzberg SL (2009) A whole-genome assembly of the domestic cow, Bos taurus. Genome Biol 10. https://doi.org/10.1186/gb-2009-10-4-r42.
Funding
This work was supported by the Indian Council of Agricultural Research, Centre for Agricultural Bioinformatics project CABin/100644/16103/801/10133.
Author information
Authors and Affiliations
Contributions
Conceptualization: Amit Kumar, Bharat Bhushan and Satish Kumar; Methodology: Satish Kumar, Rajni Chaudhary, Jaya Bharati; Formal analysis and investigation: Satish Kumar, Jaya Bharati, A. Karthikeyan; Writing - original draft preparation: Satish Kumar, Jaya Bharati; Writing - review and editing: Manjit Panigrahi, Kaisa Kaiho, Soni Kumari; Funding acquisition: Amit Kumar; Resources: Amit Kumar, Santanu Banik, G. K. Gaur, Triveni Dutt; Supervision: Amit Kumar, Bharat Bhushan.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All the experimental procedures were performed in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experimentation in Animals (CPCSEA), Ministry of Environment, Forest and Climate Change, Government of India. The methods and protocol had prior approval of Institutional Animal Ethics Committee of ICAR-IVRI with approval no. No. F.I-53/2012- 13/JD(R).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, S., Bhushan, B., Kumar, A. et al. Elucidation of novel SNPs affecting immune response to classical swine fever vaccination in pigs using immunogenomics approach. Vet Res Commun 48, 941–953 (2024). https://doi.org/10.1007/s11259-023-10262-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10262-3