Abstract
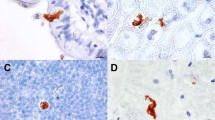
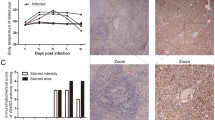
Rabbit Haemorrhagic Disease (RHD) is a lethal infection caused by calicivirus that kills 90% of the infected adult rabbits within 3 days. The calicivirus replicates in the liver and causes a fulminant hepatitis. Most studies on the pathology of RHD have been focused on the fulminant liver disease. This may not be the only mechanism in the pathogenesis of RHD: calicivirus infection may also induce leukopenia in the infected adult rabbits. We show now by flow cytometry analysis that the calicivirus induces an early decrease in B and T cells, in both spleen and liver. The depletion of B and T cells was associated with apoptosis labelled by annexin V. These changes occurred in rabbits before they showed enzymatic evidence of liver damage and persisted after liver transaminase values were very high. We conclude that depletion of lymphocytes caused by the calicivirus infection precedes or attends liver damage. The relative contribution of this lymphocyte depletion for the pathogenesis of the fatal calicivirus infection of rabbits remains to be investigated.





Similar content being viewed by others
References
Baize S, Leroy EM, Georges AJ, Georges-Courbot MC, Capron M, Bedjabaga I, Lansoud-Soukate J, Mavoungou E (2002) Inflammatory responses in Ebola virus-infected patients. Clin Exp Immunol 128: 163–168.
Bertagnoli S, Gelfi J, Petit F, Vautherott JF, Rasschaert D, Laurentt S, Le Gallf G, Boilletot E, Chantal J, Boucraut-Baralon C (1996) Protection of rabbits against rabbit viral haemorrhagic disease with a vaccinia-RHDV recombinant virus. Vaccine 14 (6): 506–510.
Bradfute SB, Braun DR, Shamblin JD, Geisbert JB, Paragas J, Garrison A, Hensley LE, Geisbert TW (2007) Lymphocyte Death in a Mouse Model of Ebola Virus Infection. J Infect Dis 196 (S2): 296-304.
Cooke BD (2002) Rabbit haemorrhagic disease: field epidemiology and the management of wild rabbit populations. Rev sci tech Off int Epiz 2: 347–358.
Ferreira PG, Costa-e-Silva A, Monteiro E, Oliveira MJR, Águas AP (2004) Transient decrease in blood heterophils and sustained liver damage caused by calicivirus infection of young rabbits that are naturally resistant to rabbit haemorrhagic disease. Res Vet Sci 76: 83–94.
Ferreira PG, Costa-e-Silva A, Oliveira MJR, Monteiro E, Águas AP (2006a) Severe leukopenia and liver biochemistry changes in adult rabbits after calicivirus infection. Res Vet Sci 80: 218–225.
Ferreira PG, Costa-e-Silva A, Oliveira MJR, Monteiro E, Águas AP (2006b) Liver enzymes and ultrastructure in rabbit haemorrhagic disease (RHD). Vet Res Comm 30: 393–401.
Ferreira PG, Dinís M, Costa-e-Silva A, Oliveira MJR, Águas AP (2008) Adult rabbits acquire resistance to lethal calicivirus infection by adoptive transfer of sera from infected young rabbits. Vet Immuno Imunopathol 121: 364–69.
Kotani M, Yamamura Y, Tamatani T, Kitamura F, Miyasaka M (1993) Generation and characterisation of monoclonal antibodies against rabbit CD4, CD5 and CD11a cell surface antigens. J Immunol Methods 157: 241–252.
Laurent S, Vautherot JF, Madelaine MF, Gall G, Rasschaert D (1994) Recombinant Rabbit Hemorrhagic Disease Virus Capsid Protein Expressed in Baculovirus Self-Assembles into Virus like Particles and Induces Protection. J Virol 68 (10): 6794–6798.
Leroy EM, Baize S, Volchkov VE, Fisher-Hoch SP, Georges-Courbot MC, Lansoud-Soukate J, Capro M, Debré P, McCormick JB, Georges AJ (2000) Human asymptomatic Ebola infection and strong inflammatory response. The Lancet 355: 2210–2215.
Mahanty S, Bray M (2004) Pathogenesis of filoviral haemorrhagic fevers. The Lancet, Infect Dis 4: 487–498.
Marcato PS, Benazzi C, Vecchi G, Galleotti M, Della Salda L, Sarli G, Lucidi P (1991) Clinical and pathological features of viral haemorrhagic disease of rabbits and the European brown hare syndrome. Rev Sci Tech Off Int Epiz 10: 371–392.
Mitro S, Krauss H (1993) Rabbit hemorrhagic disease: a review with special reference to its epizootiology. Eur J Epid 9: 70–78.
Niedzwiedzka-Rystwej P, Pawlikowska M, Hukowska-Szematowicz B, et al. (2009) Immunological and genetic studies of RHD (rabbit haemorrhagic disease) virus strains. Central Eur J Immuno 34 (2):61-67.
Ohlinger VF, Haas B, Meyers G, Weiland F, Thiel H-J (1990) Identification and characterization of the virus causing rabbit hemorrhagic disease. J Virol 64: 3331–3336.
Parra F, Prieto M (1990) Purification and characterization of a calicivirus as the causative agent of a lethal hemorrhagic disease in rabbits. J Virol 64: 4013–4015.
Ramiro-Ibáñez F, Martín-Alonso JM, Palencia PG, Parra F, Alonso C (1999) Macrophage tropism of rabbit hemorrhagic disease virus is associated with vascular pathology. Virus Res 60: 21–28.
Shien JH, Shieh HK, Lee LH (2000) Experimental infections of rabbits with rabbit haemorrhagic disease virus monitored by polymerase chain reaction. Res Vet Sci 68: 255–259.
Tokarz-Deptula B, Deptula W (2004) T and B lymphocytes and their subpopulations in peripheral blood in rabbits experimentally infected with Fr-2 strain of viral haemorrhagic disease (VHD) virus. Bull Vet.Inst Pulawy 48 (4): 367–370.
Tokarz-Deptula B, Deptula T (2009) Selected immunological factors in rabbits infected with four strains (Fr-1, Fr-2, SGM, Mal) of the RHD virus (rabbit haemorrhagic disease). Central Eur J Immuno 34 (4): 222-226.
Acknowledgements
We are grateful to Professor Francisco Parra (University of Oviedo) for the offer of the initial sample of calicivirus suspension that was used to infect the rabbits. We thank Dr. José Carlos Oliveira (Santo António Hospital), Mrs. Maria Júlia Reis (Santo António Hospital) and Mrs. Manuela Silva for excellent technical assistance. This work was funded by grants from FCT, Portugal.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marques, R.M., Costa-e-Silva, A., Águas, A.P. et al. Early acute depletion of lymphocytes in calicivirus-infected adult rabbits. Vet Res Commun 34, 659–668 (2010). https://doi.org/10.1007/s11259-010-9437-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-010-9437-7