Abstract
Objective
The LC–MS/MS-based non-targeted metabolomics method was used to differentially screen serum and urine metabolites of acute kidney injury (AKI) patients and healthy people, to explore potential biomarkers of AKI and analyze related pathways, and explain the potential mechanism and biological significance of AKI.
Methods
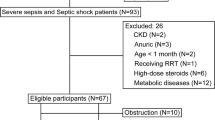
The serum and urine samples from 30 AKI patients and 20 healthy people were selected to conduct a non-targeted metabolomics study by ultra-high-performance liquid chromatography-tandem quadrupole time-of-flight mass spectrometry (UPLC-Q/TOF–MS). The differential metabolites between the two groups were searched by the human metabolome (HMDB) database (https://hmdb.ca/) and the related pathways of these potential biomarkers were identified by searching the Kyoto encyclopedia of genes and genomes (KEGG) database (https://www.kegg.jp/). The total metabolic pathways were analyzed by the MS Peaks to Pathways module of MetaboAnalyst (https://www.metaboanalyst.ca/).
Results
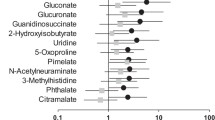
Multivariate data analysis found that serum and urine metabolism in AKI patients was significantly different from healthy people. We found three metabolites in urine (2-S-glutathionyl glutathione acetate, 5-l-Glutamyl-taurine, and l-Phosphoarginine) contributing to the separation of AKI patients from healthy people, and major metabolic pathways associated with these potential biomarkers including cytochrome P450 metabolism, arginine, and proline metabolism.
Conclusion
2-S-glutathionyl glutathione acetate, 5-l-Glutamyl-taurine, and l-Phosphoarginine were associated with AKI patients, which could be selected as potential biomarkers to predicate AKI disease.




Similar content being viewed by others
References
Gonsalez SR, Cortes AL, Silva RCD, Lowe J, Prieto MC, Silva Lara LD (2019) Acute kidney injury overview: from basic findings to new prevention and therapy strategies. Pharmacol Ther 200:1–12. https://doi.org/10.1016/j.pharmthera.2019.04.001
Lewington AJ, Cerda J, Mehta RL (2013) Raising awareness of acute kidney injury: a global perspective of a silent killer. Kidney Int 84(3):457–467. https://doi.org/10.1038/ki.2013.153
Rangaswamy D, Sud K (2018) Acute kidney injury and disease: long-term consequences and management. Nephrology (Carlton) 23(11):969–980. https://doi.org/10.1111/nep.13408
Zuk A, Bonventre JV (2016) Acute kidney injury. Annu Rev Med 67:293–307. https://doi.org/10.1146/annurev-med-050214-013407
Khwaja A (2012) KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 120(4):c179-184. https://doi.org/10.1159/000339789
Teo SH, Endre ZH (2017) Biomarkers in acute kidney injury (AKI). Best Pract Res Clin Anaesthesiol 31(3):331–344. https://doi.org/10.1016/j.bpa.2017.10.003
Beker BM, Corleto MG, Fieiras C, Musso CG (2018) Novel acute kidney injury biomarkers: their characteristics, utility and concerns. Int Urol Nephrol 50(4):705–713. https://doi.org/10.1007/s11255-017-1781-x
Johnson CH, Ivanisevic J, Siuzdak G (2016) Metabolomics: beyond biomarkers and towards mechanisms. Nat Rev Mol Cell Biol 17(7):451–459. https://doi.org/10.1038/nrm.2016.25
Pezzatti J, Boccard J, Codesido S, Gagnebin Y, Joshi A, Picard D, Gonzalez-Ruiz V, Rudaz S (2020) Implementation of liquid chromatography-high resolution mass spectrometry methods for untargeted metabolomic analyses of biological samples: a tutorial. Anal Chim Acta 1105:28–44. https://doi.org/10.1016/j.aca.2019.12.062
Alge JL, Arthur JM (2015) Biomarkers of AKI: a review of mechanistic relevance and potential therapeutic implications. Clin J Am Soc Nephrol 10(1):147–155. https://doi.org/10.2215/CJN.12191213
Tajima S, Yamamoto N, Masuda S (2019) Clinical prospects of biomarkers for the early detection and/or prediction of organ injury associated with pharmacotherapy. Biochem Pharmacol 170:113664. https://doi.org/10.1016/j.bcp.2019.113664
Dudzik D, Barbas-Bernardos C, Garcia A, Barbas C (2018) Quality assurance procedures for mass spectrometry untargeted metabolomics. A review. J Pharm Biomed Anal 147:149–173. https://doi.org/10.1016/j.jpba.2017.07.044
Chambers MC, Maclean B, Burke R, Amodei D, Ruderman DL, Neumann S, Gatto L, Fischer B, Pratt B, Egertson J, Hoff K, Kessner D, Tasman N, Shulman N, Frewen B, Baker TA, Brusniak MY, Paulse C, Creasy D, Flashner L, Kani K, Moulding C, Seymour SL, Nuwaysir LM, Lefebvre B, Kuhlmann F, Roark J, Rainer P, Detlev S, Hemenway T, Huhmer A, Langridge J, Connolly B, Chadick T, Holly K, Eckels J, Deutsch EW, Moritz RL, Katz JE, Agus DB, MacCoss M, Tabb DL, Mallick P (2012) A cross-platform toolkit for mass spectrometry and proteomics. Nat Biotechnol 30(10):918–920. https://doi.org/10.1038/nbt.2377
Chai YC, Ashraf SS, Rokutan K, Johnston RB Jr, Thomas JA (1994) S-thiolation of individual human neutrophil proteins including actin by stimulation of the respiratory burst: evidence against a role for glutathione disulfide. Arch Biochem Biophys 310(1):273–281. https://doi.org/10.1006/abbi.1994.1167
Nagarkoti S, Dubey M, Awasthi D, Kumar V, Chandra T, Kumar S, Dikshit M (2018) S-Glutathionylation of p47phox sustains superoxide generation in activated neutrophils. Biochim Biophys Acta Mol Cell Res 1865(2):444–454. https://doi.org/10.1016/j.bbamcr.2017.11.014
Wan X, Li X, Wang Q, Zheng B, Zhou C, Kang X, Hu D, Bao H, Peng A (2019) Metabolitic profiling of amino acids in paraquat-induced acute kidney injury. Clin Exp Nephrol 23(4):474–483. https://doi.org/10.1007/s10157-019-01702-z
Moonen L, D’Haese PC, Vervaet BA (2018) Epithelial cell cycle behaviour in the injured kidney. Int J Mol Sci. https://doi.org/10.3390/ijms19072038
Zhang A, Zhou X, Zhao H, Zou S, Ma CW, Liu Q, Sun H, Liu L, Wang X (2017) Metabolomics and proteomics technologies to explore the herbal preparation affecting metabolic disorders using high resolution mass spectrometry. Mol Biosyst 13(2):320–329. https://doi.org/10.1039/c6mb00677a
Xu L, Wang Y, Ma Z, Tang X, Gao Y (2020) Urine metabolomics study on potential hepatoxic biomarkers identification in rats induced by aurantio-obtusin. Front Pharmacol 11:1237. https://doi.org/10.3389/fphar.2020.01237
Zeng W, Huang KE, Luo Y, Li DX, Chen W, Yu XQ, Ke XH (2020) Nontargeted urine metabolomics analysis of the protective and therapeutic effects of Citri Reticulatae Chachiensis Pericarpium on high-fat feed-induced hyperlipidemia in rats. Biomed Chromatogr 34(4):e4795. https://doi.org/10.1002/bmc.4795
Popolo A, Adesso S, Pinto A, Autore G, Marzocco S (2014) L-Arginine and its metabolites in kidney and cardiovascular disease. Amino Acids 46(10):2271–2286. https://doi.org/10.1007/s00726-014-1825-9
George JA, Gounden V (2019) Novel glomerular filtration markers. Adv Clin Chem 88:91–119. https://doi.org/10.1016/bs.acc.2018.10.005
Oliva-Damaso E, Oliva-Damaso N, Rodriguez-Esparragon F, Payan J, Baamonde-Laborda E, Gonzalez-Cabrera F, Santana-Estupinan R, Rodriguez-Perez JC (2019) Asymmetric (ADMA) and Symmetric (SDMA) dimethylarginines in chronic kidney disease: a clinical approach. Int J Mol Sci. https://doi.org/10.3390/ijms20153668
Funding
This study was supported by the National Key Laboratory of Tea Tree Biology and Resource Utilization of Anhui Agricultural University in 2015 Open Fund (SKLTOF20150106).
Author information
Authors and Affiliations
Contributions
Study concept and design: CC, PZ, WC, GB. Data extraction and analysis: PZ, CC, WC, GB. Manuscript drafting: CC, PZ, YF. Manuscript revision: WC, GB, YF. Manuscript review and editing: CC, PZ, WC, GB.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Medical Research Ethics Committee of the first affiliated hospital of the University of Science and Technology of China (Anhui Provincial Hospital). The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, C., Zhang, P., Bao, G. et al. Discovery of potential biomarkers in acute kidney injury by ultra-high-performance liquid chromatography-tandem quadrupole time-of-flight mass spectrometry (UPLC-Q/TOF–MS). Int Urol Nephrol 53, 2635–2643 (2021). https://doi.org/10.1007/s11255-021-02829-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-021-02829-3