Abstract
Purpose
Most nosocomial urinary tract infections are related to urinary catheters and their biofilm. For the research in this province, animal models are needed to explain the host–pathogen interactions and have an advantage in controlling external interference. This study investigated the validity of the denaturing gradient gel electrophoresis (DGGE) technique for identification of bacteria in a rat model of urinary catheter biofilm.
Methods
After cutting with aseptic technique, the 24-gauge polyurethane IV catheter was placed in the female rat bladder through the urethra. The catheters were kept in the bladder for 2, 4, or 6 weeks for each group. The genomic DNA was isolated from harvested biofilm of the extracted catheter, and DGGE was performed. The band patterns of DGGE results were analysed, and the sequences were compared using the BLAST from the NCBI.
Results
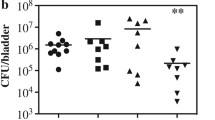
The results show that Pseudomonas aerusinosa, Escherichia coli, Enterococcus spp., and Corynebacterium sp. were the dominant bacterial species, regardless of the indwelling periods, and other species of bacteria, including Burkholderia and Achromobacter, were identified. The changes in bacterial distribution for the different indwelling periods were non-specific.
Conclusions
This study using rat model of urinary catheter suggests that DGGE is a useful method in the analysis of the bacterial community in biofilms. Molecular techniques, including DGGE, are valuable to identify fastidious bacteria in the urinary catheter biofilm. This study may be used as fundamental data for studies involving human materials hereafter.




Similar content being viewed by others
References
Rebmann T, Greene LR (2010) Preventing catheter-associated urinary tract infections: an executive summary of the Association for Professionals in Infection Control and Epidemiology, Inc. Elimination guide. Am J Infect Control 38:644–646
Trautner BW, Hull RA, Darouiche RO (2005) Prevention of catheter-associated urinary tract infection. Curr Opin Infect Dis 18:37–41
Morris NS, Stickler DJ, McLean RJ (1999) The development of bacterial biofilms on indwelling urethral catheters. World J Urol 17:345–350
Trevors JT (2011) Viable but non-culturable (VBNC) bacteria: gene expression in planktonic and biofilm cells. J Microbiol Methods 86:266–273
Ley RE, Turnbaugh PJ, Klein S, Gordon JI (2006) Microbial ecology: human gut microbes associated with obesity. Nature 444:1022–1023
Paster BJ, Boches SK, Galvin JL, Ericson RE, Lau CN, Levanos VA et al (2001) Bacterial diversity in human subgingival plaque. J Bacteriol 183:3770–3783
McCartney AL (2002) Application of molecular biological methods for studying probiotics and the gut flora. Br J Nutr 88(Suppl 1):S29–S37
Johnson DE, Lockatell CV, Hall-Craggs M, Warren JW (1991) Mouse models of short- and long-term foreign body in the urinary bladder: analogies to the bladder segment of urinary catheters. Lab Anim Sci 41:451–455
Krane LS, Gorbachinsky I, Sirintrapun J, Yoo JJ, Atala A, Hodges SJ (2011) Halofuginone-coated urethral catheters prevent periurethral spongiofibrosis in a rat model of urethral injury. J Endourol 25:107–112
Ishii K, Fukui M, Takii S (2000) Microbial succession during a composting process as evaluated by denaturing gradient gel electrophoresis analysis. J Appl Microbiol 89:768–777
Amann RI, Ludwig W, Schleifer KH (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59:143–169
Edwards C (2000) Problems posed by natural environments for monitoring microorganisms. Mol Biotechnol 15:211–223
Rovery C, Greub G, Lepidi H, Casalta JP, Habib G, Collart F et al (2005) PCR detection of bacteria on cardiac valves of patients with treated bacterial endocarditis. J Clin Microbiol 43:163–167
Cherkaoui A, Emonet S, Ceroni D, Candolfi B, Hibbs J, Francois P et al (2009) Development and validation of a modified broad-range 16S rDNA PCR for diagnostic purposes in clinical microbiology. J Microbiol Methods 79:227–231
Kassem II, Esseili MA, Sigler V (2011) Detection and differentiation of staphylococcal contamination of clinical surfaces using denaturing gradient gel electrophoresis. J Hosp Infect 78:187–193
Farnleitner AH, Zibuschka F, Burtscher MM, Lindner G, Reischer G, Mach RL (2004) Eubacterial 16S-rDNA amplicon profiling: a rapid technique for comparison and differentiation of heterotrophic plate count communities from drinking water. Int J Food Microbiol 92:333–345
Linder LE, Curelaru I, Gustavsson B, Hansson HA, Stenqvist O, Wojciechowski J (1984) Material thrombogenicity in central venous catheterization: a comparison between soft, antebrachial catheters of silicone elastomer and polyurethane. JPEN J Parenter Enter Nutr 8:399–406
Sherertz RJ, Carruth WA, Marosok RD, Espeland MA, Johnson RA, Solomon DD (1995) Contribution of vascular catheter material to the pathogenesis of infection: the enhanced risk of silicone in vivo. J Biomed Mater Res 29:635–645
Marosok R, Washburn R, Indorf A, Solomon D, Sherertz R (1996) Contribution of vascular catheter material to the pathogenesis of infection: depletion of complement by silicone elastomer in vitro. J Biomed Mater Res 30:245–250
Martinez–Martinez L, Pascual A, Perea EJ (1990) Effect of three plastic catheters on survival and growth of Pseudomonas aeruginosa. J Hosp Infect 16:311–318
Doyle B, Mawji Z, Horgan M, Stillman P, Rinehart A, Bailey J et al (2001) Decreasing nosocomial urinary tract infection in a large academic community hospital. Lippincotts Case Manag 6:127–136
Puri J, Mishra B, Mal A, Murthy NS, Thakur A, Dogra V et al (2002) Catheter associated urinary tract infections in neurology and neurosurgical units. J Infect 44:171–175
Bouza E, San Juan R, Munoz P, Voss A, Kluytmans J (2001) A European perspective on nosocomial urinary tract infections II. Report on incidence, clinical characteristics and outcome (ESGNI-004 study). European Study Group on Nosocomial Infection. Clin Microbiol Infect 7:532–542
Wade WG (2011) Has the use of molecular methods for the characterization of the human oral microbiome changed our understanding of the role of bacteria in the pathogenesis of periodontal disease? J Clin Periodontol 38(Suppl 11):7–16
Cairns S, Thomas JG, Hooper SJ, Wise MP, Frost PJ, Wilson MJ et al (2011) Molecular analysis of microbial communities in endotracheal tube biofilms. PLoS ONE 6:e14759
Riggio MP, Dempsey KE, Lennon A, Allan D, Ramage G, Bagg J (2010) Molecular detection of transcriptionally active bacteria from failed prosthetic hip joints removed during revision arthroplasty. Eur J Clin Microbiol Infect Dis 29:823–834
Acknowledgments
The authors wish to acknowledge the financial support of Yuhan Corp. and the St. Vincent’s Hospital Research Institute of Medical Science. This study was made possible through the contributions of the research coordinator, Seung June Noh.
Conflict of interest
The authors have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choe, HS., Kim, HJ., Lee, SJ. et al. Evaluation of the bacterial distribution within the biofilm by denaturing gradient gel electrophoresis in the rat model of urinary catheters. Int Urol Nephrol 45, 743–748 (2013). https://doi.org/10.1007/s11255-013-0430-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-013-0430-2