Abstract
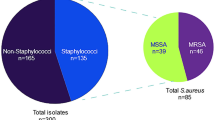
Staphylococcus aureus is among the microorganisms more frequently associated with subclinical bovine mastitis. S. aureus may produce several virulence factors. This study aimed at determining the frequency of virulence factors such as enterotoxins, toxic shock syndrome toxin 1, and ica adhesion genes. In addition, we assessed antimicrobial drug resistance in S. aureus isolated from clinical and subclinical cases of mastitis. A total of 88 cows with clinical or subclinical mastitis were sampled, resulting in 38 S. aureus isolates, from which 25 (65.78%) carried toxin genes, including seb, sec, sed, tst, and icaD adhesion gene. These S. aureus isolates belong to 21 ribotypes and three S. aureus strains belonged to the same ribotype producing ica adhesion gene. Approximately 90% of S. aureus strains obtained in our study demonstrated multiple resistance to different antimicrobial agents. The most efficacious antimicrobial agents against the isolates were gentamicin, amoxicillin, and norfloxacin. Gentamicin was the most efficacious agent inhibiting 78.95% of the S. aureus isolates. The least efficacious were penicillin, streptomycin, and ampicillin. Our results can help in understanding the relationship between virulence factors and subclinical mastitis caused by S. aureus. Further research about diversity of S. aureus isolates and genes responsible for the pathogenicity of subclinical mastitis is essential.


Similar content being viewed by others
References
Aarestrup, F.M., Wegener, H.C., Jensen, N.E., Jonsson, O., Myllys, V., Thorberg, B.M., Waage, S., and Rosdahl, V.T., 1997. A study of phage- and ribotype patterns of Staphylococcus aureus isolated from bovine mastitis in the Nordic countries, Acta Veterinaria Scandinavica, 38, 243–252
Akineden, O., Annemuller, C., Hassan, A.A., Lammler, C., Wolter, W., and Zschock, M., 2001. Toxin genes and other characteristics of Staphylococcus aureus isolates from milk of cows with mastitis, Clinical and Diagnostic Laboratory Immunology, 8, 959–964
Babra, C., Tiwari, J.G., Pier, G., Thein, T.H., Sunagar, R., Sundareshan, S., Isloor, S., Hegde, N.R., de Wet, S., Deighton, M., Gibson, J., Costantino, P., Wetherall, J., and Mukkur, T., 2013. The persistence of biofilm-associated antibiotic resistance of Staphylococcus aureus isolated from clinical bovine mastitis cases in Australia, Folia Microbiologica, 58, 469–474
Barkema, H.W., Schukken, Y.H., and Zadoks, R.N., 2006. Invited review: the role of cow, pathogen, and treatment regimen in the therapeutic success of bovine Staphylococcus aureus mastitis, Journal of Dairy Science, 89, 1877–1895
Bauer, A.W., Kirby, W.M., Sherris, J.C., and Turck, M., 1966. Antibiotic susceptibility testing by a standardized single disk method, American Journal of Clinical Pathology, 45, 493–496
Becker, K., and Eiff, C.v., 2011. Staphylococcus, Micrococcus, and other catalase-positive cocci. In: J. Versalovic et al. (eds), Manual for clinical microbiology, 2011, (American Society for Microbiology, Washington, DC, USA), 308–330
Becker, K., Roth, R., and Peters, G., 1998. Rapid and specific detection of toxigenic Staphylococcus aureus: use of two multiplex PCR enzyme immunoassays for amplification and hybridization of staphylococcal enterotoxin genes, exfoliative toxin genes, and toxic shock syndrome toxin 1 gene, Journal of Clinical Microbiology, 36, 2548–2553
Bouchet, V., Huot, H., and Goldstein, R., 2008. Molecular genetic basis of ribotyping, Clinical Microbiology Reviews, 21, 262–273
CLSI, Clinical and Laboratory Standards Institute. 2003. Padronização dos testes de sensibilidade a antimicrobianos por disco-difusão. Norma aprovada - Oitava edição. Clinical and Laboratory Standards Institute. 2003,
Cramton, S.E., Gerke, C., Schnell, N.F., Nichols, W.W., and Götz, F., 1999. The intercellular adhesion (ica) locus is present in Staphylococcus aureus and is required for biofilm formation, Infection and Immunity, 67, 5427–5433
Cuny, C., Claus, H., and Witte, W., 1996. Discrimination of S. aureus strains by PCR for r-RNA gene spacer size polymorphism and comparison to SmaI macrorestriction patterns, Zentralblatt für Bakteriologie, 283, 466–476
De Vliegher, S., Fox, L.K., Piepers, S., McDougall, S., and Barkema, H.W., 2012. Invited review: mastitis in dairy heifers: nature of the disease, potential impact, prevention, and control, Journal of Dairy Science, 95, 1025–1040
El-Ghodban, A., Ghenghesh, K.S., Marialigeti, K., Esahli, H., and Tawil, A., 2006. PCR detection of toxic shock syndrome toxin of Staphylococcus aureus from Tripoli, Libya, Journal of Medical Microbiology, 55, 179–182
Farahmand-Azar, S., Ahmadi, M., Saei, H.D., and Anassori, E., 2013. Identification of toxic shock syndrome toxin-1 (TSST-1) gene in Staphylococcus aureus isolated from bovine mastitis milk, Archives of Razi Institute, 68, 17–22
Garcia, L.S., 2010. Biochemical tests for the identification of aerobic bacteria. Clinical Microbiology Procedures Handbook, 2010, (ASM Press, Washington, DC, USA), 503–642
Holmberg, O., 1973. Staphylococcus epidermidis isolated from bovine milk. Biochemical properties, phage sensitivity and pathogenicity for the udder, Acta Veterinaria Scandinavica. Supplementum, 1–144
Hunter, P.R., and Gaston, M.A., 1988. Numerical index of the discriminatory ability of typing systems: an application of Simpson’s index of diversity, Journal of Clinical Microbiology, 26, 2465–2466
Iyer, A.P., Albaik, M., and Baghallab, I., 2014. Mastitis in camels in African and Middle East countries, Journal of Bacteriology and Parasitology, 5, 1–11
Johnson, S., Kruger, D., and Labischinski, H., 1995. FemA of Staphylococcus aureus: isolation and immunodetection, FEMS Microbiology Letters, 132, 221–228
Kalorey, D.R., Shanmugam, Y., Kurkure, N.V., Chousalkar, K.K., and Barbuddhe, S.B., 2007. PCR-based detection of genes encoding virulence determinants in Staphylococcus aureus from bovine subclinical mastitis cases, Journal of Veterinary Science, 8, 151–154
Kirkan, S., Göksoy, E.Ö., and Kaya, O., 2005. Identification and antimicrobial susceptibility of Staphylococcus aureus and coagulase negative staphylococci from bovine mastitis in the Aydin Region of Turkey, Turkish Journal of Veterinary and Animal, 29, 791–796
Kolbert, C.P., and Persing, D.H., 1999. Ribosomal DNA sequencing as a tool for identification of bacterial pathogens, Current Opinion in Microbiology, 2, 299–305
Kuroishi, T., Komine, K., Kai, K., Itagaki, M., Kobayashi, J., Ohta, M., Kamata, S., and Kumagai, K., 2003. Concentrations and specific antibodies to staphylococcal enterotoxin-C and toxic shock syndrome toxin-1 in bovine mammary gland secretions, and inflammatory response to the intramammary inoculation of these toxins, The Journal of Veterinary Medical Science, 65, 899–906
Lancette, G.A., and Tatini, S.R., 2001. Staphylococcus aureus. In: F.P. Downes and K. Ito (eds), Compendium of Methods for the Microbiological Examination of Foods, 2001, (American Public Health Association, Washington, DC), 387–403
Mehrotra, M., Wang, G., and Johnson, W.M., 2000. Multiplex PCR for detection of genes for Staphylococcus aureus enterotoxins, exfoliative toxins, toxic shock syndrome toxin 1, and methicillin resistance, Journal of Clinical Microbiology, 38, 1032–1035
Melchior, M.B., Fink-Gremmels, J., and Gaastra, W., 2007. Extended antimicrobial susceptibility assay for Staphylococcus aureus isolates from bovine mastitis growing in biofilms, Veterinary Microbiology, 125, 141–149
Namvar, A.E., Asghari, B., Ezzatifar, F., Azizi, G., and Lari, A.R., 2013. Detection of the intercellular adhesion gene cluster (ica) in clinical Staphylococcus aureus isolates, GMS Hygiene and Infection Control, 8, Doc03
Omoe, K., Hu, D.L., Takahashi-Omoe, H., Nakane, A., and Shinagawa, K., 2003. Identification and characterization of a new staphylococcal enterotoxin-related putative toxin encoded by two kinds of plasmids, Infection and Immunity, 71, 6088–6094
Omoe, K., Imanishi, K., Hu, D.L., Kato, H., Fugane, Y., Abe, Y., Hamaoka, S., Watanabe, Y., Nakane, A., Uchiyama, T., and Shinagawa, K., 2005. Characterization of novel staphylococcal enterotoxin-like toxin type P, Infection and Immunity, 73, 5540–5546
Persson, Y., Nyman, A.K., and Gronlund-Andersson, U., 2011. Etiology and antimicrobial susceptibility of udder pathogens from cases of subclinical mastitis in dairy cows in Sweden, Acta Veterinaria Scandinavica, 53, 1–8
Pyorala, S., and Taponen, S., 2009. Coagulase-negative staphylococci-emerging mastitis pathogens, Veterinary Microbiology, 134, 3–8
Raza, A., Muhammad, G., Sharif, S., and Atta, A., 2013. Biofilm producing Staphylococcus aureus and bovine mastitis: a review, Molecular Microbiology Research, 3, 1–8
Riyaz-Ul-Hassan, S., Verma, V., and Qazi, G.N., 2008. Evaluation of three different molecular markers for the detection of Staphylococcus aureus by polymerase chain reaction, Food Microbiology, 25, 452–459
Schelin, J., Wallin-Carlquist, N., Cohn, M.T., Lindqvist, R., Barker, G.C., and Radstrom, P., 2011. The formation of Staphylococcus aureus enterotoxin in food environments and advances in risk assessment, Virulence, 2, 580–592
Schuberth, H.J., Krueger, C., Zerbe, H., Bleckmann, E., and Leibold, W., 2001. Characterization of leukocytotoxic and superantigen-like factors produced by Staphylococcus aureus isolates from milk of cows with mastitis, Veterinary Microbiology, 82, 187–199
Stewart, P.S., Davison, W.M., and Steenbergen, J.N., 2009. Daptomycin rapidly penetrates a Staphylococcus epidermidis biofilm, Antimicrobial Agents and Chemotherapy, 53, 3505–3507
Thompson-Crispi, K., Atalla, H., Miglior, F., and Mallard, B.A., 2014. Bovine mastitis: frontiers in immunogenetics, Frontiers in Immunology, 5, 493
Vicosa, G.N., Moraes, P.M., Yamazi, A.K., and Nero, L.A., 2010. Enumeration of coagulase and thermonuclease-positive Staphylococcus spp. in raw milk and fresh soft cheese: an evaluation of Baird-Parker agar, Rabbit Plasma Fibrinogen agar and the Petrifilm Staph Express count system, Food Microbiology, 27, 447–452
Viguier, C., Arora, S., Gilmartin, N., Welbeck, K., and O’Kennedy, R., 2009. Mastitis detection: current trends and future perspectives, Trends in Biotechnology, 27, 486–493
Wang, D., Wang, Z., Yan, Z., Wu, J., Ali, T., Li, J., Lv, Y., and Han, B., 2015. Bovine mastitis Staphylococcus aureus: antibiotic susceptibility profile, resistance genes and molecular typing of methicillin-resistant and methicillin-sensitive strains in China, Infection, Genetics and Evolution, 31, 9–16
Zschock, M., Risse, K., and Sommerhauser, J., 2004. Occurrence and clonal relatedness of sec/tst-gene positive Staphylococcus aureus isolates of quartermilk samples of cows suffering from mastitis, Letters in Applied Microbiology, 38, 493–498
Funding
This study was supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil), FAPEMIG (Fundação de Amparo a Pesquisa do Estado de Minas Gerais, Brazil), FAPEMA (Fundação de Amparo a Pesquisa e ao Desenvolvimento Científico e Tecnológico do Estado do Maranhão, Brazil), CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brazil), and UEMA (Universidade Estadual do Maranhão, Brazil). FNC had a scholarship from CAPES (PROCAD-NF Program). RLS has a fellowship from CNPq. IAC has a fellowship from UEMA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The manuscript does not contain clinical studies or patient data.
This experimental protocol has been approved by the Ethics Committee on Animal Use of the Universidade Estadual do Maranhão (CEEA-UEMA, Protocol 039/2011).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Costa, F.N., Belo, N.O., Costa, E.A. et al. Frequency of enterotoxins, toxic shock syndrome toxin-1, and biofilm formation genes in Staphylococcus aureus isolates from cows with mastitis in the Northeast of Brazil. Trop Anim Health Prod 50, 1089–1097 (2018). https://doi.org/10.1007/s11250-018-1534-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-018-1534-6