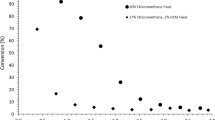
LaCl3 is an active, selective and stable catalyst for oxidative chlorination of methane to methyl chloride. Selective conversion to methyl chloride can be achieved by limiting methane conversion, for example, by using an excess of methane in the feed. Methylene chloride and carbon monoxide are the main side products at higher methane conversion levels. Transient OCl− anion, formed by oxidation of Cl− in LaOCl and LaCl3 with molecular oxygen, is proposed to be the active site for the initial step of methane activation. CO x formation is proposed to proceed through the formation of adsorbed multiply substituted chloromethanes.
Similar content being viewed by others
References
S. Freni G. Calogero S. Cavallaro (2000) J. Power Sources 87 28 Occurrence Handle1:CAS:528:DC%2BD3cXitFyqtbo%3D Occurrence Handle10.1016/S0378-7753(99)00357-2
G.M. Zhidomirov V.I. Avdeev N.U. Zhanpeisov I.I. Zakharov I.V. Yudanov (1995) Catal. Today 24 383 Occurrence Handle1:CAS:528:DyaK2MXlvFKhtLY%3D Occurrence Handle10.1016/0920-5861(95)00063-L
Y. Sun S.M. Campbell J.H. Lunsford G.E. Lewis D. Palke L.M. Tau (1993) J. Catal. 143 32 Occurrence Handle1:CAS:528:DyaK3sXmt1Knt7c%3D Occurrence Handle10.1006/jcat.1993.1251
L.A. Noronha E.F. Souza-Aguiar C.J.A. Mota (2005) Catal. Today 101 9 Occurrence Handle1:CAS:528:DC%2BD2MXjs1Snu7g%3D Occurrence Handle10.1016/j.cattod.2004.12.004
R.A. Periana O. Mironov D. Taube G. Bhalla C.J. Jones (2003) Science 301 814 Occurrence Handle1:CAS:528:DC%2BD3sXmtVGqs78%3D Occurrence Handle10.1126/science.1086466
Eb. Svetlano R.M. Flid Rv. Dzhagats A.M. Taber (1967) Russ. J. Phys. Chem. 41 487
G.A. Olah B. Gupta M. Farina J.D. Felberg W.M. Ip A. Husain R. Karpeles K. Lammertsma A.K. Melhotra N.J. Trivedi (1985) J. Am. Chem. Soc. 107 7097 Occurrence Handle1:CAS:528:DyaL2MXmtFCrs7k%3D Occurrence Handle10.1021/ja00310a057
G.A. Olah (1987) Acc. Chem. Res. 20 422 Occurrence Handle1:CAS:528:DyaL1cXksFCht70%3D Occurrence Handle10.1021/ar00143a006
B.R. Crum, R.F.J. Jarvis, B.M. Naasz and A.I. Toupadakis EP 0720975 A1 (1996)
A.J. Rouco (1995) J. Catal. 157 380 Occurrence Handle1:CAS:528:DyaK2MXhtVSktrfM Occurrence Handle10.1006/jcat.1995.1303
F. Wattimena W.M.H. Sachtler (1982) Stud. Surf. Sci. Catal. 7 816 Occurrence Handle10.1016/S0167-2991(08)64695-9
C. Kenney (1975) Catal. Rev.-Sci. Eng. 11 197 Occurrence Handle1:CAS:528:DyaE2MXlt1WhtLc%3D
C.L. Garcia D.E. Resasco (1989) Appl. Catal. 46 251 Occurrence Handle1:CAS:528:DyaL1MXhvFGjtbw%3D Occurrence Handle10.1016/S0166-9834(00)81121-5
C.L. Garcia D.E. Resasco (1990) J. Catal. 122 151 Occurrence Handle1:CAS:528:DyaK3cXhs1Gqurw%3D Occurrence Handle10.1016/0021-9517(90)90267-N
W.J.M. Pieters W.C. Conner E.J Carlson (1984) Appl. Catal. 11 35 Occurrence Handle1:CAS:528:DyaL2cXksl2hsbs%3D Occurrence Handle10.1016/S0166-9834(00)84038-5
A.E. Schweizer, M.E. Jones and D.A. Hickman US 6452058 B1 (2002)
O.V. Manoilova S.G. Podkolzin B. Tope J.A. Lercher E. Stangland J.M. Goupil B.M. Weckhuysen (2004) J. Phys. Chem. B 108 15770 Occurrence Handle1:CAS:528:DC%2BD2cXnsV2itbY%3D Occurrence Handle10.1021/jp040311m
S.G. Podkolzin O.V. Manoilova B.M. Weckhuysen (2005) J. Phys. Chem. B 109 11634 Occurrence Handle1:CAS:528:DC%2BD2MXkt1aisbo%3D Occurrence Handle10.1021/jp050680y
B. Schoofs J.A. Martens P.A. Jacobs R.A. Schoonheydt (1999) J. Catal. 183 355 Occurrence Handle1:CAS:528:DyaK1MXislelu7w%3D Occurrence Handle10.1006/jcat.1999.2401
P. Avert ParticleVan der B.M. Weckhuysen (2004) PCCP 6 5256
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peringer, E., Podkolzin, S.G., Jones, M.E. et al. LaCl3-based catalysts for oxidative chlorination of CH4 . Top Catal 38, 211–220 (2006). https://doi.org/10.1007/s11244-006-0085-7
Issue Date:
DOI: https://doi.org/10.1007/s11244-006-0085-7