Abstract
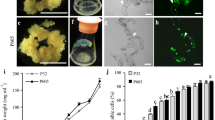
Plant cell suspension cultures are used in basic research and applied biotechnology. In both cases, the transfer and stable integration of heterologous genes is a required technique. This report describes a rapid method for transformation of cell cultures of Medicago truncatula, the model species for the legume family. Accession A17 from the cultivar Jemalong is the reference genotype selected for the sequencing of the genome and therefore most studies on Medicago are carried out on this accession line. However, this line has a low embryogenic capacity and is poorly responsive to transformation protocols that rely on somatic embryogenesis. An alternative method for transformation of suspension cultures of this line, which does not depend on leaf transformation or somatic embryogenesis, was therefore needed. The method described herein uses Agrobacterium tumefaciens mediated gene transfer, allowing the transformation of Medicago callus tissue and the following establishment of liquid suspension cell cultures approximately 2 months after transformation. Kanamycin resistance was used to select for positive transformation events and the screening was facilitated by visualization of a fluorescent marker, which was fused to the gene of interest. This new protocol reduces the time between transformation and cell culture establishment, and allows the generation of transgenic suspension cultures of Medicago reference accession A17.

Similar content being viewed by others
References
An G (1985) High efficiency transformation of cultured tobacco cells. Plant Physiol 79:568–570. https://doi.org/10.1104/pp.79.2.568
Chabaud M, Larsonneau C, Marmouget C, Huguet T (1996) Transformation of barrel medic (Medicago truncatula Gaertn.) by Agrobacterium tumefaciens and regeneration via somatic embryogenesis of transgenic plants with the MtENOD12 nodulin promoter fused to the gus reporter gene. Plant Cell Rep 15:305–310. https://doi.org/10.1007/BF00232361
Chabaud M, Carvalho-Niebel D, Barker DG (2003) Efficient transformation of Medicago truncatula cv. Jemalong using the hypervirulent Agrobacterium tumefaciens strain AGL1. Plant Cell Rep 22:46–51. https://doi.org/10.1007/s00299-003-0649-y
Duque AS, Araújo SS, Cordeiro MA, Santos DM, Fevereiro MP (2007) Use of fused gfp and gus reporters for the recovery of transformed Medicago truncatula somatic embryos without selective pressure. Plant Cell Tissue Organ Cult 90:325–330. https://doi.org/10.1007/s11240-007-9268-6
Iantcheva A, Revalska M, Zehirov G, Vassileva V (2014) Agrobacterium-mediated transformation of Medicago truncatula cell suspension culture provides a system for functional analysis. In Vitro Cell Dev Biol Plant 50:149–157. https://doi.org/10.1007/s11627-013-9554-4
Mayo KJ, Gonzales BJ, Mason HS (2006) Genetic transformation of tobacco NT1 cells with Agrobacterium tumefaciens. Nat Protoc 1:1105–1111. https://doi.org/10.1038/nprot.2006.176
Neves LO, Duque SRL, de Almeida JS, Fevereiro PS (1999) Repetitive somatic embryogenesis in Medicago truncatula ssp. Narbonensis and M. truncatula Gaertn cv. Jemalong. Plant Cell Rep 18:398–405. https://doi.org/10.1007/s002990050593
Pires AS, Cabral MG, Fevereiro P, Stoger E, Abranches R (2008) High levels of stable phytase accumulate in the culture medium of transgenic Medicago truncatula cell suspension cultures. Biotechnol J 3:916–923. https://doi.org/10.1002/biot.200800044
Pires AS, Rosa S, Castanheira S, Fevereiro P, Abranches R (2012) Expression of a recombinant human erythropoietin in suspension cultures of Arabidopsis, tobacco and Medicago. Plant Cell Tissue Organ Cult 110:171–181. https://doi.org/10.1007/s11240-012-0141-x
Pires AS, Santos RB, Nogueira AC, Abranches R (2014) Production of human lipocalin-type prostaglandin D synthase in the model plant Medicago truncatula. In Vitro Cell Dev Biol Plant 50:276–281. https://doi.org/10.1007/s11627-013-9584-y
Santos RB, Abranches R, Fischer R, Sack M, Holland T (2016) Putting the spotlight back on plant suspension cultures. Front Plant Sci 7:297. https://doi.org/10.3389/fpls.2016.00297
Santos RB, Pires AS, Abranches R (2017) Addition of a histone deacetylase inhibitor increases recombinant protein expression in Medicago truncatula cell cultures. Sci Rep 7:16756. https://doi.org/10.1038/s41598-017-17006-9
Santos RB, Chandrasekar B, Mandal MK, Kaschani F, Kaiser M, Both L, van der Hoorn RAL, Schiermeyer A, Abranches R (2018) Low protease content in Medicago truncatula cell cultures facilitates recombinant protein production. Biotechnol J 13:1800050. https://doi.org/10.1002/biot.201800050
Shimada TL, Shimada T, Hara-Nishimura I (2010) A rapid and non-destructive screenable marker, FAST, for identifying transformed seeds of Arabidopsis thaliana. Plant J 61:519–528. https://doi.org/10.1111/j.1365-313X.2009.04060.x
Tang H, Krishnakumar V, Bidwell S, Rosen B, Chan A, Zhou S, Gentzbittel L, Childs KL, Yandell M, Gundlach H, Mayer KFX, Schwartz DC, Town CD (2014) An improved genome release (version Mt4.0) for the model legume Medicago truncatula. BMC Genom 15:312. https://doi.org/10.1186/1471-2164-15-312
Thomas MR, Rose RJ, Nolan KE (1992) Genetic transformation of Medicago truncatula using Agrobacterium with genetically modified Ri and disarmed Ti plasmids. Plant Cell Rep 11:113–117. https://doi.org/10.1007/BF00232161
Trieu AT, Harrison MJ (1996) Rapid transformation of Medicago truncatula: regeneration via shoot organogenesis. Plant Cell Rep 16:6–11. https://doi.org/10.1007/BF01275439
Trinh TH, Ratet P, Kondorosi E, Durand P, Kamaté K, Bauer P, Kondorosi A (1998) Rapid and efficient transformation of diploid Medicago truncatula and Medicago sativa ssp. falcata lines improved in somatic embryogenesis. Plant Cell Rep 17: 345–355. https://doi.org/10.1007/s002990050405
Young ND, Debellé F, Oldroyd GE, Geurts R, Cannon SB et al (2011) The Medicago genome provides insight into the evolution of rhizobial symbioses. Nature 480:520–524. https://doi.org/10.1038/nature10625
Acknowledgements
This work has been funded through the ERA-IB project PRODuCE from the Portuguese Fundação para a Ciência e Tecnologia (FCT, ERA-IB/0001/2012 to RA) and the German BMBF (031A219A to AS; 031A219B to RALvdH). Additional funding from FCT was granted through UID/Multi/04551/2013 and PPBI—Portuguese Platform of BioImaging (PPBI-POCI-01-0145-FEDER-022122). RALvdH was recipient of ERC consolidator Grant 616449 ‘GreenProteases’. The authors thank James Yates for revising the manuscript.
Author information
Authors and Affiliations
Contributions
RBS and RA conceived research. RBS and ASP conducted experiments. AS and RALvdH contributed new reagents and tools. RBS and RA wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by S. J. Ochatt.
Rights and permissions
About this article
Cite this article
Santos, R.B., Pires, A.S., van der Hoorn, R.A.L. et al. Generation of transgenic cell suspension cultures of the model legume Medicago truncatula: a rapid method for Agrobacterium mediated gene transfer. Plant Cell Tiss Organ Cult 136, 445–450 (2019). https://doi.org/10.1007/s11240-018-1525-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-018-1525-3