Abstract
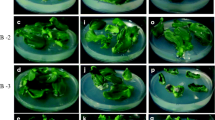
Leaf tissue is widely used for regeneration of many different plants, but an efficient regeneration method using leaf tissue of sunflower is not yet available. In this report, adventitious shoots were obtained from primary leaves of 7-day-old sunflower seedlings. If seeds were germinated on a medium without cytokinin, only 25 % of the leaf explants produced shoots after 3 weeks of culture on a shoot induction medium (SIM) containing 1.5 mg L−1 benzylaminopurine (BA), giving an average of three shoot primordia per leaf. Shoot induction was enhanced up to sixfold when seeds were germinated on media containing BA or 6-(γ, γ-dimethylallylamino)purine at 5 mg L−1 and higher, or kinetin at 15 mg L−1 and above, with an average of 12–19 shoot primordia per leaf. Shoots further developed and elongated following transfer to a medium containing 0.1 mg L−1 gibberellic acid. The highest number of developed shoots was obtained from leaves preconditioned with 5 mg L−1 BA, with 1.9 developed shoots per explant. Histological analysis of leaves from seedlings germinated on a medium containing 5 mg L−1 BA revealed increased cell division primarily in the leaf surface tissue on the adaxial side of the mid vein. After the leaves were excised and plated on SIM, these cells continued to divide and differentiate to form adventitious shoots, suggesting that the cytokinin-containing germination medium preconditioned leaves for shoot organogenesis. An in vitro micrografting approach was developed using the elongated shoots as scions, and 57 % of grafted shoots survived to form plants.



Similar content being viewed by others
References
Abousalim A, Mantell SH (1992) Micrografting of pistachio (Pistacia vera L. cv. Mateur). Plant Cell Tissue Organ Cult 29:231–234
Akasaka-Kennedy Y, Yoshida H, Takahata Y (2005) Efficient plant regeneration from leaves of rapeseed (Brassica napus L.): the influence of AgNO3 and genotype. Plant Cell Rep 24:649–654
Bobák M, Blehová A, Krištín J, Ovečka M, Šamaj J (1995) Direct plant regeneration from leaf explants of Drosera rotundifolia cultured in vitro. Plant Cell, Tissue Organ Cult 43:43–49
Bronner R, Jeannin G, Hahne G (1994) Early cellular events during organogenesis and somatic embryogenesis induced on immature zygotic embryos of sunflower (Helianthus annuus). Can J Bot 72:239–248
Burrus M, Molinier J, Himber C, Hunold R, Bronner R, Rousselin P, Hahne G (1996) Agrobacterium-mediated transformation of sunflower (Helianthus annuus L.) shoot apices: transformation patterns. Mol Breed 2:329–338
Ceriani MF, Hopp HE, Hahne G, Escandón AS (1992) Cotyledons: an explant for routine regeneration of sunflower plants. Plant Cell Physiol 33:157–164
Chevreau E, Skirvin R, Abu-Qaoud H, Korban S, Sullivan J (1989) Adventitious shoot regeneration from leaf tissue of three pear (Pyrus sp.) cultivars in vitro. Plant Cell Rep 7:688–691
de Mendiburu F (2015) Statistical procedures for agricultural research. Package ‘Agricolae,’ version 1.2-1. Comprehensive R Archive Network, Institute for Statistics and Mathematics, Vienna. http://cran.r-project.org/web/packages/agricolae/agricolae.pdf
Escalettes V, Dosba F (1993) In vitro adventitious shoot regeneration from leaves of Prunus spp. Plant Sci 90:201–209
Fasolo F, Zimmerman RH, Fordham I (1989) Adventitious shoot formation on excised leaves of in vitro grown shoots of apple cultivars. Plant Cell Tissue Organ Cult 16:75–87
Finer JJ (1987) Direct somatic embryogenesis and plant regeneration from immature embryos of hybrid sunflower (Helianthus annuus L.) on a high sucrose-containing medium. Plant Cell Rep 6:372–374
Gamborg OL, Miller R, Ojima K (1986) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158
Henderson JHM, Durrell ME, Bonner J (1952) The culture of normal sunflower stem callus. Am J Bot 39:467–473
Hervé P, Jauneau A, Pâques M, Marien JN, Boudet AM, Teulières C (2001) A procedure for shoot organogenesis in vitro from leaves and nodes of an elite Eucalyptus gunnii clone: comparative histology. Plant Sci 161:645–653
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Johnson SJ, Miles CA (2011) Effect of healing chamber design on the survival of grafted eggplant, tomato, and watermelon. Horttechnology 21:752–758
Knittel N, Escandón AS, Hahne G (1991) Plant regeneration at high frequency from mature sunflower cotyledons. Plant Sci 73:219–226
Konov A, Bronner R, Skryabin K, Hahne G (1998) Formation of epiphyllous buds in sunflower (Helianthus annuus L.): induction in vitro and cellular origin. Plant Sci 135:77–86
Koroch A, Juliani H, Kapteyn J, Simon J (2002) In vitro regeneration of Echinacea purpurea from leaf explants. Plant Cell Tissue Organ Cult 69:79–83
Kudo T, Kiba T, Sakakibara H (2010) Metabolism and long-distance translocation of cytokinins. J Integr Plant Biol 52:53–60
Luo J, Gould JH (1999) In vitro shoot-tip grafting improves recovery of cotton plants from culture. Plant Cell Tissue Organ Cult 57:211–213
Malik KA, Saxena PK (1991) Regeneration in Phaseolus vulgaris L. Promotive role of N6-benzylaminopurine in cultures from juvenile leaves. Planta 184:148–150
Malik KA, Khan STA, Saxena PK (1992) Direct organogenesis and plant regeneration in preconditioned tissue cultures of Lathyrus cicera L., L. ochrus (L.)DC. and L. sativus L. Ann Bot 70:301–304
McClean P, Grafton KF (1989) Regeneration of dry bean (Phaseolus vulgaris L.) via organogenesis. Plant Sci 60:117–122
McCormick S, Niedermeyer J, Fry J, Barnason A, Horsch R, Fraley R (1986) Leaf disc transformation of cultivated tomato (L. esculentum) using Agrobacterium tumefaciens. Plant Cell Rep 5:81–84
Mneney EE, Mantell SH (2001) In vitro micrografting of cashew. Plant Cell Tissue Organ Cult 66:49–58
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Navarro L, Roistacher C, Murashige T (1975) Improvement of shoot-tip grafting in vitro for virus-free citrus. J Am Soc Hort Sci 100:471–479
Paterson KE (1984) Shoot tip culture of Helianthus annuus-flowering and development of adventitious and multiple shoots. Am J Bot 71:925–931
Peña L, Cervera M, Juárez J, Navarro A, Pina JA, Durán-Vila N, Navarro L (1995a) Agrobacterium-mediated transformation of sweet orange and regeneration of transgenic plants. Plant Cell Rep 14:616–619
Peña L, Cervera M, Juárez J, Ortega C, Pina J, Durán-Vila N, Navarro L (1995b) High efficiency Agrobacterium-mediated transformation and regeneration of citrus. Plant Sci 104:183–191
Pieron S, Boxus P, Dekegel D (1998) Histological study of nodule morphogenesis from Cichorium intybus L. leaves cultivated in vitro. In Vitro Cell Dev Biol Plant 34:87–93
Power CJ (1987) Organogenesis from Helianthus annuus inbreds and hybrids from the cotyledons of zygotic embryos. Am J Bot 74:497–503
Puthra GT, Anil SR (2002) In vitro grafting of cashew (Anacardium occidentale L.). Sci Hortic 92:177–182
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Sarrafi A, Roustan JP, Fallot J, Alibert G (1996) Genetic analysis of organogenesis in the cotyledons of zygotic embryos of sunflower (Helianthus annuus L.). Theor Appl Genet 92:225–229
Saxena PK, Malik KA, Gill R (1992) Induction by thidiazuron of somatic embryogenesis in intact seedlings of peanut. Planta 187:421–424
Shiva Prakash N, Pental D, Bhalla-Sarin N (1994) Regeneration of pigeonpea (Cajanus cajan) from cotyledonary node via multiple shoot formation. Plant Cell Rep 13:623–627
Stamp JA, Colby SM, Meredith CP (1990) Direct shoot organogenesis and plant regeneration from leaves of grape (Vitis spp.). Plant Cell Tissue Organ Cult 22:127–133
Sujatha M, Vijay S, Vasavi S, Reddy PV, Rao SC (2012a) Agrobacterium-mediated transformation of cotyledons of mature seeds of multiple genotypes of sunflower (Helianthus annuus L.). Plant Cell Tissue Organ Cult 110:275–287
Sujatha M, Vijay S, Vasavi S, Sivaraj N, Rao SC (2012b) Combination of thidiazuron and 2-isopentenyladenine promotes highly efficient adventitious shoot regeneration from cotyledons of mature sunflower (Helianthus annuus L.) seeds. Plant Cell Tissue Organ Cult 111:359–372
Weber S, Friedt W, Landes N, Molinier J, Himber C, Rousselin P, Horn R (2003) Improved Agrobacterium-mediated transformation of sunflower (Helianthus annuus L.): assessment of macerating enzymes and sonication. Plant Cell Rep 21:475–482
Wright M, Koehler S, Hinchee M, Carnes M (1986) Plant regeneration by organogenesis in Glycine max. Plant Cell Rep 5:150–154
Wright M, Williams M, Pierson P, Carnes M (1987) Initiation and propagation of Glycine max L. Merr.: plants from tissue-cultured epicotyls. Plant Cell Tissue Organ Cult 8:83–90
Wu H, Du Toit ES, Reinhardt C (2007) Micrografting of Protea cynaroides. Plant Cell Tissue Organ Cult 89:23–28
Acknowledgments
We thank Tea Meulia and Andrea Kaszas in the Molecular and Cellular Imaging Center at The Ohio State University for assistance with the histological analysis. Salaries and research support were provided by the United States Department of Agriculture-Agricultural Research Service program National Sclerotinia Initiative and by State and Federal funds appropriated to The Ohio State University/Ohio Agricultural Research and Development Center. We would also like to thank Brent Hulke at the Sunflower Research Unit of the Red River Valley Agricultural Research Center for providing RHA280 germplasm and the United States Department of Agriculture North Central Regional Plant Introduction Station at Iowa State University for providing other sunflower germplasm. Mention of trademark or proprietary products does not constitute a guarantee or warranty of the product by OSU/OARDC and also does not imply approval to the exclusion of other products that may also be suitable.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Z., Finer, J.J. Sunflower (Helianthus annuus L.) organogenesis from primary leaves of young seedlings preconditioned by cytokinin. Plant Cell Tiss Organ Cult 123, 645–655 (2015). https://doi.org/10.1007/s11240-015-0867-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-015-0867-3