Abstract
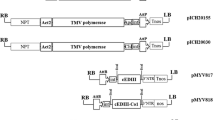
The dengue virus envelope glycoprotein (DV-E) has been identified as a promising candidate for the development of a subunit vaccine and to provide an antigen for diagnostic kits. In this study, cell suspension cultures of Nicotiana tabacum and Morinda citrifolia were transformed and evaluated for production of the DV-E serotype 2 protein. The expression cassette consisted of the DV-E gene along with a signal peptide at its 5′ end. In addition, a KDEL endoplasmic retention sequence was included in a second construct to evaluate its influence on accumulation levels of the recombinant protein. Expression cassettes were sub-cloned into a binary vector pCAMBIA 1305.2, and transformation was carried out using Agrobacterium tumefaciens. Transformed cell lines of both N. tabacum and M. citrifolia accumulated DV-E protein, although those carrying the construct containing the KDEL sequence showed higher accumulation levels than those without. Overall, cell suspension cultures of N. tabacum were more suitable for dengue antigen production than those of M. citrifolia. The highest levels of protein accumulation (0.71 ± 0.06 mg DV-E/L) were observed in tobacco cell lines at 5 days of culture, corresponding to 0.3% of total soluble protein. The recombinant protein was reactive with anti-E antibodies.




Similar content being viewed by others
References
Bisht H, Chugh D, Raje M, Swaminathan S, Khanna N (2002) Recombinant dengue virus type 2 envelope/hepatitis B surface antigen hybrid protein expressed in Pichia pastoris can function as a bivalent immunogen. J Biotechnol 99:97–110. doi:10.1016/S0168-1656(02)00181-5
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
De Sousa Sampaio P, Neto H, Poejo P, Serrazina S, Soares Pais M (2010) Overexpression and characterization of cyprosin B in transformed suspension cells of Cynara cardunculus. Plant Cell Tissue Organ Cult 101:311–321. doi:10.1007/s11240-010-9690-z
Delenda C, Staropoli I, Frenkiel M, Cabani L, Deubel V (1994) Analysis of C-terminally truncated dengue 2 and dengue 3 virus envelope glycoproteins: processing in insect cells and immunogenic properties in mice. J Gen Virol 75:1569–1578. doi:10.1099/0022-1317-75-7-1569
Emanuelsson O, Nielsen H, Brunak S, von Heijne G (2000) Predicting subcellular localization of proteins based on their N-terminal amino acid sequence. J Mol Biol 300:1005–1016. doi:10.1006/jmbi.2000.3903
Faye L, Boulaflous A, Benchabane M, Gomord V, Michaud D (2005) Protein modifications in the plant secretory pathway: current status and practical implications in molecular pharming. Vaccine 23:1770–1778. doi:10.1016/j.vaccine.2004.11.003
Fiedler U, Phillips J, Artsaenko O, Conrad U (1997) Optimization of scFv antibody production in transgenic plants. Immunotechnology 3(3):205–216. doi:10.1016/S1380-2933(97)00014-6
Fischer R, Stoger E, Schillberg S, Christou P, Twyman R (2004) Plant-based production of biopharmaceuticals. Curr Opin Plant Biol 7(2):152–158. doi:10.1016/j.pbi.2004.01.007
Franconi R, Demurtas O, Massa S (2010) Plant-derived vaccines and other therapeutics produced in contained systems. Expert Rev Vacc 9(8):877–892. doi:10.1586/erv.10.91
Gamborg O, Miller R, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50(1):151–158. doi:10.1016/0014-4827(68)90403-5
Girard L, Bastin M, Courtois D (2004) Expression of the human milk protein sCD14 in tobacco plant cell culture. Plant Cell Tissue Organ Cult 78:253–260. doi:10.1023/B:TICU.0000025667.46429.4d
Gómez E, Chimeno Zoth S, Asurmendi Vázquez, Rovere C, Berinstein A (2009) Expression of hemagglutinin–neuraminidase glycoprotein of newcastle disease virus in agroinfiltrated Nicotiana benthamiana plants. J Biotechnol 144:337–340. doi:10.1016/j.jbiotec.2009.09.015
Hellwig S, Drossar J, Twyman R, Fischer R (2004) Plant cell cultures for the production of recombinant proteins. Nat Biotechnol 22(11):1415–1422. doi:10.1038/nbt1027
Hsieh S, Liu I, King C, Chang G, Wang W (2008) A strong endoplasmic reticulum retention signal in the stem–anchor region of envelope glycoprotein of dengue virus type 2 affects the production of virus-like particles. Virology 374(2):338–350. doi:10.1016/j.virol.2007.12.041
Huang T, Plesha M, Falk B, Dandekar A, McDonald K (2009) Bioreactor strategies for improving production yield and functionality of a recombinant human protein in transgenic tobacco cell cultures. Biotechnol Bioeng 102:508–520. doi:10.1002/bit.22061
James E, Wang C, Wang Z, Reeves R, Shin J, Magnuson N, Lee J (2000) Production and characterization of biologically active human GM-CSF secreted by genetically modified plant cells. Protein Expr Purif 19:131–138. doi:10.1006/prep.2000.1232
Karg S, Kallio P (2009) The production of biopharmaceuticals in plant systems. Biotechnol Adv 27(6):879–894. doi:10.1016/j.biotechadv.2009.07.002
Kelly E, Greene J, King A, Innis B (2000) Purified dengue 2 virus envelope glycoprotein aggregates produced by baculovirus are immunogenic in mice. Vaccine 18(23):2549–2559. doi:10.1016/S0264-410X(00)00032-3
Kim Y, Kwon T, Sik Y (2004) Direct transfer and expression of human GM-CSF in tobacco suspension cell using Agrobacterium-mediated transfer system. Plant Cell Tissue Organ Cult 78(2):133–138. doi:10.1023/B:TICU.0000022555.50772.09
Kim Y, Kim B, Kim T, Kang T, Yang M (2006) Expression of a cholera toxin B subunit in transgenic lettuce (Lactuca sativa L.) using Agrobacterium-mediated transformation system. Plant Cell Tissue Organ Cult 87:203–210
Kim M, Kim T, Yoo H, Yang M (2010a) Expression and assembly of ApxIIA toxin of Actinobacillus pleuropneumoniae fused with the enterotoxigenic E. coli heat-labile toxin B subunit in transgenic tobacco. Plant Cell Tissue Organ Cult. Online First. doi:10.1007/s11240-010-9877-3
Kim T, Kim M, Yang M (2010b) Cholera toxin B subunit-domain III of dengue virus envelope glycoprotein E fusion protein production in transgenic plants. Protein Expr Purif 74(2):236–241. doi:10.1016/j.pep.2010.07.013
Kyle J, Harris E (2008) Global spread and persistence of dengue. Annu Rev Microbiol 62:71–92. doi:10.1146/annurev.micro.62.081307.163005
Lerouge P, Cabanes-Macheteau M, Rayon C, Fischette-Lainé A, Gomord V, Faye L (1998) N-Glycoprotein biosynthesis in plants: recent developments and future trends. Plant Mol Biol 38:31–48. doi:10.1023/A:1006012005654
Lienard D, Tran Dinh O, van Oort E, Van Overtvelt L, Bonneau C, Wambre E, Bardor M, Cosette P, Didier-Laurent A, Dorlhac de Borne F, Delon R, van Ree R, Moingeon P, Faye L, Gomord V (2007) Suspension-cultured BY-2 tobacco cells produce and mature immunologically active house dust mite allergens. Plant Biotechnol J 5:93–108. doi:10.1111/j.1467-7652.2006.00221.x
Loc N, Song N, Tien N, Minh T, Nga P, Kim T, Yang M (2010) Expression of the Escherichia coli heat-labile enterotoxin B subunit in transgenic watercress (Nasturtium officinale L.). Plant Cell Tissue Organ Cult 105(1):39–45. doi:10.1007/s11240-010-9835-0
Martínez C, Petruccelli S, Giulietti A, Álvarez M (2005) Expression of the antibody 14D9 in Nicotiana tabacum hairy roots. Electronic Journal of Biotechnology 8(2): 170-176. doi:10.2225/vol8-issue2-fulltext-10. http://www.ejbiotechnology.info/content/vol8/issue2/full/10/index.html
Martínez C, Topal E, Giulietti A, Rodríguez Talou J, Mason H (2010) Exploring different strategies to express dengue virus envelope protein in a plant system. Biotechnol Lett 32:867–875. doi:10.1007/s10529-010-0236-6
Muné M, Rodríguez R, Ramírez R, Soto Y, Sierra B, Rodríguez Roche R, Marquez G, Garcia J, Guillén G, Guzmán G (2003) Carboxy-terminally truncated Dengue 4 virus envelope glycoprotein expressed in Pichia pastoris induced neutralizing antibodies and resistance to Dengue 4 virus challenge in mice. Arch Virol 148:2267–2273. doi:10.1007/s00705-003-0167-9
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Murrell S, Wu S, Butler M (2011) Review of dengue virus and the development of a vaccine. Biotechnol Adv 29:239–247. doi:10.1016/j.biotechadv.2010.11.008
Nielsen H, Engelbrecht J, Brunak S, von Heijne G (1997) Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng 10:1–6. doi:10.1093/protein/10.1.1
Plas L, Eijkelboom C, Hagendoorn MJM (1995) Relation between primary and secondary metabolism in plant cell suspensions. Plant Cell Tissue Organ Cult 43(2):111–116. doi:10.1007/BF00052164
Pugachev K, Guirakhoo F, Trent D, Monath T (2003) Traditional and novel approaches to flavivirus vaccines. Int J Parasitol 33:567–582. doi:10.1016/S0020-7519(03)00063-8
Pugachev K, Guirakhoo F, Monath T (2005) New developments in flavivirus vaccines with special attention to yellow fever. Curr Opin Infect Dis 18:387–394. doi:00001432-200510000-00004
Rodríguez Talou J, Verberne MC, Budi Muljono RA, van Tegelen L, Gonsalvez Bernal B, Linthorst H, Wullems G, Bol J, Verpoorte R (2001) Isochorismate synthase transgenic expression in Catharanthus roseus cell suspensions. Plant Physiol Biochem 39:595–602. doi:10.1016/S0981-9428(01)01279-7
Saejung W, Fujiyama K, Takasaki T, Ito M, Hori K, Malasit P, Watanabe Y, Kurane I, Seki T (2007) Production of dengue 2 envelope domain III in plant using TMV-based vector system. Vaccine 25(36):6646–6654. doi:10.1016/j.vaccine.2007.06.029
Scheller J, Gührs K, Grosse F, Conrad U (2001) Production of spider silk proteins in tobacco and potato. Nat Biotechnol 19(6):573–577. doi:10.1038/89335
Shih S, Doran P (2009) Foreign protein production using plant cell and organ cultures: advantages and limitations. Biotechnol Adv 27:1036–1042. doi:10.1016/j.biotechadv.2009.05.009
Sojikul P, Buehner N, Mason H (2003) A plant signal peptide–hepatitis B surface antigen fusion protein with enhanced stability and immunogenicity expressed in plant cells. PNAS 100(5):2209–2214. doi:10.1073_pnas.0438037100
Streatfield S (2007) Approaches to achieve high-level heterologous protein production in plants. Plant Biotechnol J 5:2–15. doi:10.1111/j.1467-7652.2006.00216.x
Sugrue R, Cui T, Xu Q, Fu J, Chan Y (1997) The production of recombinant dengue virus E protein using Escherichia coli and Pichia pastoris. J Virol Methods 69:159–169. doi:10.1016/S0166-0934(97)00151-1
Teli N, Timko M (2004) Recent developments in the use of transgenic plants for the production of human therapeutics and biopharmaceuticals. Plant Cell Tissue Organ Cult 79(2):125–145. doi:10.1007/s11240-004-0653-0
Teoh P, Ooi A, AbuBakar S, Othman R (2009) Virus-specific read-through codon preference affects infectivity of chimeric cucumber green mottle mosaic viruses displaying a dengue virus epitope. J Biomed Biotechnol, Volume 2009, Article ID 781712. doi:10.1155/2009/781712
Torres E, Vaquero C, Nicholson L, Sack M, Stöger E, Drossard J, Christou P, Fischer R, Perrin Y (1999) Rice cell culture as an alternative production system for functional diagnostic and therapeutic antibodies. Transgenic Res 8(6):441–449. doi:10.1023/A:1008969031219
Triguero A, Cabrera G, Cremata J, Yuen C, Wheeler J, Ramírez N (2005) Plant-derived mouse IgG monoclonal antibody fused to KDEL endoplasmic reticulum-retention signal is N-glycosylated homogeneously throughout the plant with mostly high-mannose-type N-glycans. Plant Biotechnol J 3(4):449–457. doi:10.1111/j.1467-7652.2005.00137.x
Twyman R, Stoger E, Schillberg S, Christou P, Fischer R (2003) Molecular farming in plants: host systems and expression technology. Trends Biotechnol 21(12):570–578. doi:10.1016/j.tibtech.2003.10.002
Webster D, Farrar J, Rowland-Jones S (2009) Progress towards a dengue vaccine. Lancet Infect Dis 9:678–687. doi:10.1016/S1473-3099(09)70254-3
WHO (World Health Organization) (2008) Guidelines for the clinical evaluation of dengue vaccines in endemic areas. Inmunization, vaccines and biologicals. WHO/IVB/08.12. http://whqlibdoc.who.int/hq/2008/WHO_IVB_08.12_eng.pdf
Acknowledgments
CAM is supported by Universidad de Buenos Aires. AMG and JRT are supported by CONICET (Consejo Nacional de Investigaciones Científicas y Tecnológicas). CAM, AMG and JRT thank Dr. Andrea Gamarnik for giving us the DNA genetic sequences and her assistance and to CAMBIA research organization for supplying pCAMBIA vector. This work was granted by PICT 14-15112 and 14-33166, Agencia Nacional de Promoción Científica y Tecnológica, Argentina (ANPCyT), and UBACyT B111, Universidad de Buenos Aires.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martínez, C.A., Giulietti, A.M. & Rodríguez Talou, J. Expression of a KDEL-tagged dengue virus protein in cell suspension cultures of Nicotiana tabacum and Morinda citrifolia . Plant Cell Tiss Organ Cult 107, 91–100 (2011). https://doi.org/10.1007/s11240-011-9960-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-011-9960-4