Abstract
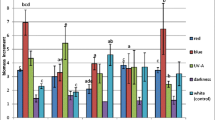
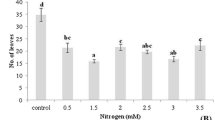
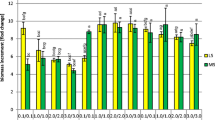
A system for growing in liquid medium whole plants of Valeriana glechomifolia, endemic to southern Brazil and capable of accumulating bioactive valepotriates, is described. Murashige and Skoog (MS) and Gamborg B5 (B5) media (1.0×, 0.3× and 0.1× strength) without phytohormones were evaluated after four weeks of culture in relation to growth and valepotriate yield. Plants grown in 1.0× MS displayed greatest growth and valepotriate yields and the study of the light condition showed that plants grown under light and dark had similar weight increase and maximum valepotriate yield, 27.2 mg/g DW and 25.0 mg/g DW, respectively. Valtrate was the most abundant valepotriate, followed by acevaltrate and didrovaltrate.
Similar content being viewed by others
Abbreviations
- ACE:
-
Acevaltrate
- DID:
-
Didrovaltrate
- B5:
-
Gamborg B5 medium
- GI:
-
Growth Index
- MS:
-
Murashige and Skoog medium
- VAL:
-
Valtrate
References
Backlund A, Moritz T (1998) Phylogenetic implications of an expanded valepotriate distribution in the Valerianaceae. Biochem Syst Ecol 26:309–335
Banerjee S, Rahman L, Uniyal GC, Ahuja PS (1998) Enhanced production of valepotriates by Agrobacterium rhizogenes induced hairy roots cultures of Valeriana wallichii DC. Plant Sci 131:203–208
Bondarev N, Reshetnyak O, Nosov A (2003) Effects of nutrient medium composition on development of Stevia rebaudiana shoots cultivated in the roller bioreactor and their production of steviol glycosides. Plant Sci 165:845–850
Bounthanh C, Bergmann C, Beck JP, Haag-Berrurier M, Anton R (1981) Valepotriates, a new class of cytotoxic and antitumor agents. Planta Med 41:21–28
Bos R, Hendriks H, Scheffer JJC, Woerdenbag HJ (1998a) Cytotoxic potential of valerian constituents and valerian tinctures. Phytomedicine 5:219–225
Bos R, Woerdenbag HJ, van Putten FMS, Hendriks H, Scheffer JC (1998b) Seasonal variation of the essential oil, valerenic acid and derivatives, and valepotriates in Valeriana officinalis roots and rhizomes, and the selection of plants suitable for phytomedicines. Planta Med 64:143–147
Castillo P, Zamilpa A, Márquez J, Hernández G, Lara M, Alvarez L (2002) Comparative study of differentiation levels and valepotriate content of in vitro cultures and regenerated and wild plants of Valeriana edulis ssp. procera. J Nat Prod 65:573–575
Chen Y-Q, Fei Y, Cai M, Lou J-X (2003) Effects of amino acids, nitrate, and ammonium on the growth and taxol production in cell cultures of Taxus yunnanensis. Plant Growth Reg 41:265–268
De Carvalho CMB, Maurmann N, Luz DI, Fett-Neto AG, Rech SB (2004) Control of development and valepotriate production by auxins in micropropagated Valeriana glechomifolia. Plant Cell Rep 23:251–255
Etienne H, Berthouly M (2001) Temporary immersion systems in plant micropropagation. Plant Cell Tiss Org Cult 69:215–231
Fuzzati N, Wolfender JL, Hostettmann K, Msonthi JD, Mavi S, Molleyres LP (1996) Isolation and antifungal valepotriates from Valeriana capense and the search for valepotriates in crude Valerianaceae extracts. Phytochem Anal 7:76–85
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158
Gao XQ, Björk L (2000) Valerenic acids and valepotriates among individuals, varieties and species of Valeriana. Fitoterapia 71:19–24
Gontier E, Clément A, Gravot TA, Lièvre A, Guckert F, Bourgaud F (2002) Hydroponic combined with natural or forced root permeabilization: a promising technique for plant secondary metabolite production. Plant Sci 163:723–732
Gränicher F, Christen P, Kapetanidis I (1992) High-yield production of valepotriates by hairy roots cultures of Valeriana officinalis L. var. sambucifolia Mikan. Plant Cell Rep 11:339–342
Herrera-Arellano A, Luna-Villegas G, Cuevas-Uriostegui ML, Alvarez L, Vargas-Pineda G, Zamilpa-Alvarez A, Tortoriello J (2001) Polysomnographic evaluation of the hypnotic effect of Valeriana edulis standardized extract in patients suffering from insomnia. Planta Med 67:695–699
Houghton PJ (1999) The scientific basis for the reputed activity of Valerian. J Pharm Pharmacol 51:505–512
Kittipongpatana N, Davis DL, Porter JR (2002) Methyl jasmonate increases the production of valepotriates by transformed root cultures of Valerianella locusta. Plant Cell Tiss Org Cult 71:65–75
Lange BM, Croteau R (1999) Isoprenoid biosynthesis via a mevalonate-independent pathway in plants: Cloning and heterologous expression of 1-deoxy-D-xylulose-5-phosphate reductoisomerase from peppermint. Arch Biochem Biophys 365:170–174
Liu CZ, Murch SJ, El-Demerdash M, Saxena PK (2004) Artemisia judaica: Micropropagation and antioxidant activity. J Biotechnol 110:63–71
Liu CZ, Honda KH, Kobayashi T (2001) In situ regeneration of rice (Oryza sativa L.) callus immobilized in polyurethane foam. J Biosci Bioeng 91:76–80
Mathur J, Ahuja PS, Mathur A, Kukreja AK, Shah NC (1988) In vitro propagation of Valeriana wallichii. Planta Med 54:82–83
Maurmann N, De Carvalho CMB, Silva AL, Fett-Neto AG, von Poser GL, Rech SB (2006) Valepotriates accumulation in callus, suspended cells and untransformed root cultures of Valeriana glechomifolia. In Vitro Cell Dev Biol Plant 42:50–53
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Murch SJ, Liu C, Romero R, Saxena PK (2003) In vitro culture and temporary immersion bioreactor production of Crescentia cujete L. Plant Cell Tiss Org Cult 73:63–68
Rout GR, Samantary S, Das P (2000) In vitro manipulation and propagation of medicinal plants. Biotechnol Adv 18:91–120
Salles LA, Silva AL, Rech SB, Zanatta N, von Poser GL (2000) Constituents of Valeriana glechomifolia Meyer. Biochem Syst Ecol 28:907–910
Salles LA, Silva AL, Fett-Neto AG, von Poser GL, Rech SB (2002) Valeriana glechomifolia: In vitro propagation and production of valepotriates. Plant Sci 163:165–168
Silva AL, Rech SB, von Poser GL (2002) Quantitative determination of valepotriates from Valeriana native to south Brazil. Planta Med 68:570–572
Sokal RR, Rohlf FJ (1981) Biometry. W. H. Freeman, San Francisco
Violon C, Dekegel D, Vercruysse A (1984) Relation between valepotriate content and differentiation level in various tissues of Valerianeae. J Nat Prod 47:934–940
Acknowledgements
The authors are grateful to the Brazilian agencies CAPES, CNPq and FAPERGS for financial support. We would also like to thank Prof. Dr. Pedro R. Petrovick (Faculdade de Farmácia, UFRGS) for lending the HPLC equipment.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Russowski, D., Maurmann, N., Rech, S.B. et al. Role of Light and Medium Composition on Growth and Valepotriate Contents in Valeriana glechomifolia Whole Plant Liquid Cultures. Plant Cell Tiss Organ Cult 86, 211–218 (2006). https://doi.org/10.1007/s11240-006-9109-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-006-9109-z