Abstract
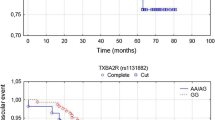
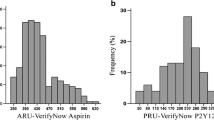
The objective of this study was to use genome-wide association approach and pooled DNA strategy to search for new genomic loci associated with inter-individual differences in platelet reactivity in the diabetic patients during acetylsalicylic acid (ASA) treatment. Study cohort consisted of 297 diabetic patients who had been taking ASA (75 mg daily) for at least 3 months. We tested association of single nucleotide polymorphisms (SNPs) genotyped using high density microarray platform with several platelet reactivity assays, followed by individual genotyping of most significant SNPs identified in the microarray genomic scan. The highest statistical significance (p value of 0.0001–0.008 in individual genotyping) was observed for SNP located within the regulatory G-protein signaling (RGS) 7 gene (rs2502448) using recessive genetic model. The diabetic patients on ASA treatment and homozygotes for its minor allele were characterized by increased odds ratio of at 3.45 (confidence interval: 1.82–6.53) for high on ASA platelet reactivity (i.e. impaired ASA response) when compared with homozygotes for wild-type allele. The genome-wide approach might provide an opportunity to identify novel candidate genes and pathways related to platelet activation in diabetic patients.


Similar content being viewed by others
References
Mortensen SB, Larsen SB, Grove EL et al (2010) Reduced platelet response to aspirin in patients with coronary artery disease and type 2 diabetes mellitus. Thromb Res 126:e318–e322
Davi G, Patrono C (2007) Platelet activation and atherothrombosis. N Engl J Med 357:2482–2494
Postula M, Kaplon-Cieslicka A, Rosiak M et al (2011) Genetic determinants of platelet reactivity during acetylsalicylic acid therapy in diabetic patients: evaluation of 27 polymorphisms within candidate genes. J Thromb Haemost 9:2291–2301
Nielsen HL, Kristensen SD, Thygesen SS et al (2008) Aspirin response evaluated by the VerifyNow aspirin system and light transmission aggregometry. Thromb Res 123:267–273
Kunicki TJ, Williams SA, Salomon DR et al (2009) Genetics of platelet reactivity in normal, healthy individuals. J Thromb Haemost 7:2116–2122
Chakroun T, Addad F, Abderazek F et al (2007) Screening for aspirin resistance in stable coronary artery patients by three different tests. Thromb Res 121:413–418
Gonzalez-Conejero R, Rivera J, Corral J et al (2005) Biological assessment of aspirin efficacy on healthy individuals: heterogeneous response or aspirin failure? Stroke 36:276–280
Janicki PK, Vealey R, Liu J et al (2011) Genome-wide association study using pooled DNA to identify candidate markers mediating susceptibility to postoperative nausea and vomiting. Anesthesiology 115:54–64
Macgregor S, Zhao ZZ, Henders A et al (2008) Highly cost-efficient genome-wide association studies using DNA pools and dense SNP arrays. Nucleic Acids Res 36:e35
Liu JZ, McRae AF, Nyholt DR et al (2010) A versatile gene-based test for genome-wide association studies. Am J Hum Genet 87:139–145
Skol AD, Scott LJ, Abecasis GR et al (2006) Joint analysis is more efficient than replication-based analysis for two-stage genome-wide association studies. Nat Genet 38:209–213
Dupont WD, Plummer WD (1990) Power and sample size calculations. A review and computer program. Control Clin Trials 11:116–128
Haubelt H, Anders C, Vogt A et al (2005) Variables influencing Platelet Function Analyzer-100 closure times in healthy individuals. Br J Haematol 130:759–767
Mathias RA, Kim Y, Sung H et al (2010) A combined genome-wide linkage and association approach to find susceptibility loci for platelet function phenotypes in European American and African American families with coronary artery disease. BMC Med Genomics 3:22
De Vries L, Gist Farquhar M (1999) RGS proteins: more than just GAPs for heterotrimeric G proteins. Trends Cell Biol 9:138–144
Austin H, De Staercke C, Lally C et al (2011) New gene variants associated with venous thrombosis: a replication study in White and Black Americans. J Thromb Haemost 9:489–495
Bezemer ID, Bare LA, Doggen CJ et al (2008) Gene variants associated with deep vein thrombosis. JAMA 299:1306–1314
Pearson JV, Huentelman MJ, Halperin RF et al (2007) Identification of the genetic basis for complex disorders by use of pooling-based genomewide single-nucleotide-polymorphism association studies. Am J Hum Genet 80:126–139
Signarvic RS, Cierniewska A, Stalker TJ et al (2010) RGS/Gi2alpha interactions modulate platelet accumulation and thrombus formation at sites of vascular injury. Blood 116:6092–6100
Gegenbauer K, Elia G, Blanco-Fernandez A et al (2012) Regulator of G-protein signaling 18 integrates activating and inhibitory signaling in platelets. Blood 119:3799–3807
Rowley JW, Oler AJ, Tolley ND et al (2011) Genome-wide RNA-seq analysis of human and mouse platelet transcriptomes. Blood 118:e101–e111
Ma P, Cierniewska A, Signarvic R et al (2012) A newly identified complex of spinophilin and the tyrosine phosphatase, SHP-1, modulates platelet activation by regulating G protein-dependent signaling. Blood 119:1935–1945
Kim SD, Sung HJ, Park SK et al (2006) The expression patterns of RGS transcripts in platelets. Platelets 17:493–497
Garcia A, Prabhakar S, Hughan S et al (2004) Differential proteome analysis of TRAP-activated platelets: involvement of DOK-2 and phosphorylation of RGS proteins. Blood 103:2088–2095
Johnson AD, Yanek LR, Chen MH et al (2010) Genome-wide meta-analyses identifies seven loci associated with platelet aggregation in response to agonists. Nat Genet 42:608–613
Gagnon AW, Murray DL, Leadley RJ (2002) Cloning and characterization of a novel regulator of G protein signalling in human platelets. Cell Signal 14:595–606
Jedema HP, Gold SJ, Gonzalez-Burgos G et al (2008) Chronic cold exposure increases RGS7 expression and decreases alpha(2)-autoreceptor-mediated inhibition of noradrenergic locus coeruleus neurons. Eur J Neurosci 27:2433–2443
Shi J, Damjanoska KJ, Zemaitaitis B et al (2006) Alterations in 5-HT2A receptor signaling in male and female transgenic rats over-expressing either Gq or RGS-insensitive Gq protein. Neuropharmacology 51:524–535
Niwa N, Nerbonne JM (2010) Molecular determinants of cardiac transient outward potassium current (I(to)) expression and regulation. J Mol Cell Cardiol 48:12–25
Hill TD, White JG, Rao GH (1989) Role of glutathione and glutathione peroxidase in human platelet arachidonic acid metabolism. Prostaglandins 38:21–32
Guerrero JA, Rivera J, Quiroga T et al (2011) Novel loci involved in platelet function and platelet count identified by a genome-wide study performed in children. Haematologica 96:1335–1343
Michelson AD (2009) Methods for the measurement of platelet function. Am J Cardiol 103:20A–26A
Acknowledgments
Dr Postula was supported by a Fulbright Fellowship from USA State Department. The AVOCADO study was supported financially as part of the research grant from the Polish Pharmaceutical Company ADAMED for a Young Scientist 2007 Award [grant number: 1WR DAR1/2007] and Polish Cardiac Society Club 30 grant 2010. The authors thank Thutrang Nguyen, B.A. from the SNP Genotyping Core Facility at Children’s Hospital Boston, Massachusetts, for performing the custom Sequenom iPLEX assay.
Disclosure of conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Marek Postula and Piotr K. Janicki contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Postula, M., Janicki, P.K., Rosiak, M. et al. New single nucleotide polymorphisms associated with differences in platelets reactivity in patients with type 2 diabetes treated with acetylsalicylic acid: genome-wide association approach and pooled DNA strategy. J Thromb Thrombolysis 36, 65–73 (2013). https://doi.org/10.1007/s11239-012-0823-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-012-0823-6