Abstract
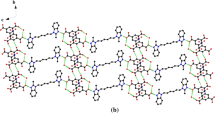
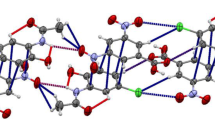
Single crystals of a 1:1 co-crystal of meloxicam [4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-1,1-dioxo-2H-1λ6,2-benzothiazine-3-carboxamide], MXM, with benzoic acid, BZA, were crystalized from a THF solution. The same MXM-BZA co-crystal has been obtained as a fine powder by liquid-assisted co-grinding using as fluid additives solvents with different polarity: benzene, toluene, ortho-xylene, meta-xylene, para-xylene, THF, and water. The latter is especially eco-friendly and can be a good candidate for industrial production. The crystal structures of all the MXM co-crystals deposited in the most recent version of the Cambridge Database were compared, in order to correlate the non-covalent interactions in these structures with the сonclusions from the theoretical analysis of solubility carried out by Cysewski (J. Mol. Model 24:112, 2018).






Similar content being viewed by others
References
Furst DE (1997). Semin Arthritis Rheum 26:21
Schattenkirchner M (1997) Expert. Opin. Investig. Drugs 6:321
Myz SA, Shakhtshneider TP, Fucke K, Fedotov AP, Boldyreva EV, Boldyrev VV, Kuleshova NI (2009). Mendeleev Communications 19:272
Cheney ML, Weyna DR, Shan N, Hanna M, Wojtas L, Zaworotko MJ (2010). Cryst Growth Des 10:4401
Tumanov NA, Myz SA, Shakhtshneider TP, Boldyreva EV (2012). CrystEngComm 14:305–313
Weyna DR, Cheney ML, Shan N, Hanna M, Zaworotko MJ, Sava V, Song S, Sanchez-Ramos JR (2012). Mol Pharm 9:2094
Myz SA, Shakhtshneider TP, Tumanov NA, Boldyreva EV (2012). Russ Chem Bull 9:1782
Fucke K, Myz SA, Shakhtshneider TP, Boldyreva EV, Griesser UJ (2012). New J Chem 36:1969
Bolla G, Sanphui P, Nangia A (2013). Cryst Growth Des 13:1988
Suresh K, Nangia A (2014). Cryst Growth Des 14:2945
Luger P, Daneck K, Engel W, Trummlitz G, Wagner K (1996). Eur J Pharm Sci 4:175
Fabiola GF, Pattabhi V, Manjunatha SG, Rao GV, Nagarajan K (1998) Acta Crystallogr. Sect C 54:2001
Cysewski P (2018). J Mol Model 24:112
Uhlemann T, Seidel S, Müller CW (2017). Phys Chem Chem Phys 19:14625
Rigaku OD (2016). CrysAlis PRO. Rigaku Oxford diffraction Ltd, Yarnton
Sheldrick GM (2015). Acta Cryst. C 71:3
Dolomanov OV, Bourhis LJ, Gildea RJ, Howard JAK, Puschmann H (2009). J Appl Crystallogr 42:339
Macrae CF, Bruno IJ, Chisholm JA, Edgington PR, McCabe P, Pidcock E, Rodriguez-Monge L, Taylor R, van de Streek J, Wood PA (2008). J Appl Crystallogr 41:466
Stoe & Cie (1999) WinXPOW. Stoe & Cie, Darmstadt
Feld R, Lehmann MS, Muir KW, Speakman JC (1981). K Zeitschriftfür Kristallographie 157:215
Arunan E, Desiraju GR, Klein RA, Sadlej J, Scheiner S, Alkorta I, Clary DC, Crabtree RH, Dannenberg JJ, Hobza P, Kjaergaard HG, Legon AC, Mennucci B, Nesbitt DJ (2011) Pure Appl. Chem 83:1637
Bernstein J, Davis RE, Shimoni L, Chang NL (1995). Angew Chem Int Ed Engl 34:1555
Cheney ML, Weyna DR, Shan N, Hanna M, Wojtas L, Zaworotko MJ (2011). J Pharm Sci 100:2172
Tantardini C, Arkhipov SG, Cherkashina KA, Kil’met’ev AS, Boldyreva EV (2016). Acta Cryst. E 72:1856
Cremer D, Pople JA (1975). J Amer Chem Soc 97:1354
Boeyens JCA (1978). J Cryst Mol Struct 8:317
Acknowledgments
The authors received funding from the Russian Ministry of Science and Higher Education.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(CIF 630 kb)
Rights and permissions
About this article
Cite this article
Tantardini, C., Arkipov, S.G., Cherkashina, K.A. et al. Synthesis and crystal structure of a meloxicam co-crystal with benzoic acid. Struct Chem 29, 1867–1874 (2018). https://doi.org/10.1007/s11224-018-1166-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-018-1166-5