Abstract
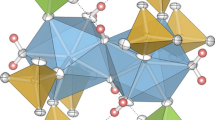
The zirconium nitrate complexes (NO2)[Zr(NO3)3(H2O)3]2(NO3)3 (1), Cs[Zr(NO3)5] ((2), (NH4)[Zr(NO3)5](HNO3) (3), and (NO2)0.23(NO)0.77[Zr(NO3)5] ((4) were prepared by crystallization from nitric acid solutions in the presence of H2SO4 or P2O5. The complexes were characterized by X-ray diffraction. The crystal structure of 1 consists of nitrate anions, nitronium cations, and [Zr(NO3)3(H2O)3]+ complex cations in which the ZrIV atom is coordinated by three water molecules and three bidentate nitrate groups. The coordination polyhedron of the ZrIV atom is a tricapped trigonal prism formed by nine oxygen atoms. The island structures of 2 and 3 contain [Zr(NO3)5]− anions and Cs+ or NH4 + cations, respectively. In addition, complex 3 contains HNO3 molecules. Complex 4 differs from (NO2)[Zr(NO3)5] in that three-fourth of the nitronium cations in 4 are replaced by nitrosonium cations NO+, resulting in a decrease in the unit cell parameters. In the [Zr(NO3)5]− anion involved in complexes 2–4, the ZrIV atom is coordinated by five bidentate nitrate groups and has an unusually high coordination number of 10. The coordination polyhedron is a bicapped square antiprism.
Similar content being viewed by others
References
P. Benard-Rocherulle, J. Rius, and D. Louer, J. Solid State Chem., 1997, 128, 295.
V. E. Plyushchev, L. I. Yuranova, L. N. Komissarova, and V. K. Trunov, Zh. Neorg. Khim., 1968, 13, 956 [J. Inorg. Chem. USSR, 1968, 13 (Engl. Transl.)].
M. Falinsky, Ann. Chimie, 1941, 16, 237.
N. I. Tuseev, A. S. Izmailovich, and L. N. Komissarova, Vestn. Mosk. Univ., Ser. 2 Khim. [Moscow Univ. Bull., Ser. 2 Chem.] 1979, 20, 453 (in Russian).
G. Tikhomirov, I. Morozov, K. Znamenkov, E. Kemnitz, and S. Troyanov, Z. Anorg. Allg. Chem., 2002, 628, 269.
V. Kaiser, S. Ebinal, F. Menzel, and E. Stumpp, Z. Anorg. Allg. Chem., 1997, 623, 449.
T. J. King, N. Logan, A. Morris, and S. C. Wallwork, J. Chem. Soc., Chem. Commun., 1971, 554.
L. J. Blackwell, E. K. Nunn, and S. C. Wallwork, J. Chem. Soc., Chem. Commun., 1975, 2068.
A. A. Fedorova, P. S. Chizhov, I. V. Morozov, and S. I. Troyanov, Zh. Neorg. Khim., 2002, 47, 2007 [Russ. J. Inorg. Chem., 2002, 47, 1845 (Engl. Transl.)].
C. C. Addison, Chem. Rev., 1980, 80, 21.
I. V. Morozov, M. V. Marsova, and S. I. Troyanov, Zh. Neorg. Khim., 2002, 47, 1055 [Russ. J. Inorg. Chem., 2002, 47, 948 (Engl. Transl.)].
C. C. Addison, G. S. Brownlee, and N. Logan, J. Chem. Soc., Dalton Trans., 1972, 1440.
D. Kepert, Inorganic Stereochemistry, Springer-Verlag, Berlin Heidelberg, New York, 1982.
ICSD/RETRIEVE 2.01, Gmelin Istitute/FIZ Karlsruhe, 1997.
E. Grison, K. Eriks, and J. L. de Vries, Acta Crystallogr., 1950, 3, 290.
R. D. Shanon, Acta Crystallogr., 1976, 32, 751.
Handbuch der praparativen anorganischen Chemie, Ed. G. Brauer, Enke, Stuttgart, 1978, 1.
PCPDFWIN Version 1.30, August 1997, JCPDS ICDD.
G. M. Sheldrick, SHELXS-97, Program for Solution of Crystal Structures from Diffraction Data, Universitat Gottingen, 1997.
G. M. Sheldrick, SHELXL-97, Program for Crystal Structure Refinement, Universitat Gottingen, 1997.
Author information
Authors and Affiliations
Additional information
__________
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 92–97, January, 2005.
Rights and permissions
About this article
Cite this article
Morozov, I.V., Fedorova, A.A., Palamarchuk, D.V. et al. Synthesis and crystal structures of zirconium(IV) nitrate complexes (NO2)[Zr(NO3)3(H2O)3]2(NO3) 3, Cs[Zr(NO3)5], and (NH4)[Zr(NO3)5](HNO3). Russ Chem Bull 54, 93–98 (2005). https://doi.org/10.1007/s11172-005-0222-7
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11172-005-0222-7