Abstract
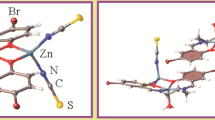
The compound {[Zn(μ1,5-dicyanamide)2(pyrazinamide)2](pyrazinamide)2}n has been synthesized and further characterized by elemental analysis, single-crystal X-ray diffraction, and thermogravimetric analysis. Structural analysis revealed that the dicyanamide anion acted as bridging ligand in a μ-1,5 mode, resulting a polymeric 1-D chain structure. The pairwise N3-H3B···N1 and N9-H9B···N7, N3-H3A···O2 and N9-H9A···O1, N3-H3B···N7 and N9-H9B···N1 multiple hydrogen bonding interactions, impart enhanced stability to supramolecular structures. Hirshfeld surfaces and fingerprint plots were applied to arrive at the same conclusion, which is stabilised mainly by H–H, N–H, O–H interactions. DNA binding studies showed that the compound had interacted with DNA.






Similar content being viewed by others
References
X.Y. Hu, T.X. Xiao, C. Lin, F.H. Huang, L.Y. Wang, Acc. Chem. Res. 47, 2041 (2014)
L. Wang, Y.J. Hu, W.Y. Xu, Y.Y. Pang, F.Q. Liu, Y. Yang, RSC Adv. 4, 56816 (2014)
J.J. Mckinnon, D. Jayatilaka, M.A. Spackman, Chem. Commun. 37, 3814 (2007)
A.D. Martin, J. Britton, T.L. Easun, A.J. Blake, W. Lewis, M. Schroder, Cryst. Growth Des. 15, 1697 (2015)
M.A. Spackman, D. Jayatilaka, CrystEngComm 11, 19 (2009)
M. Owczarek, I. Majerz, R. Jakubas, CrystEngComm 16, 7638 (2014)
Y.-H. Luo, G.-G. Wu, S.-L. Mao, B.-W. Sun, Inorg. Chim. Acta 397, 1 (2013)
A. Somoskovi, M.M. Wades, Z.H. Sun, Y. Zhang, J. Antimicrob. Chemother. 53, 192 (2004)
H. Chiniforoshan, Z.S. Radani, L. Tabrizi, H. Tavakol, M.R. Sabzalian, G. Mohammadnezhad, H. Gorls, W. Plass, J. Mol. Struct. 1081, 237 (2015)
Y.-H. Luo, B.-W. Sun, Cryst. Growth Des. 13, 2098 (2013)
F. Prins, E. Pasca, L.J.D. Jongh, H. Kooijman, A.L. Spek, S. Tanase, Angew. Chem. Int. Ed. 46, 6081 (2007)
A.M. Beatty, Coord. Chem. Rev. 246, 131 (2003)
Z. Ma, B. Moulton, Cryst. Growth Des. 7, 196 (2007)
L.S. Lerman, J. Mol. Biol. 3, 18 (1961)
W. Bauer, J. Vinograd, J. Mol. Biol. 54, 281 (1970)
D.E. Goldberg, V. Sharma, A. Oksman, I.Y. Gluzman, T.E. Wellems, D.P. Worms, J. Biol. Chem. 272, 6567 (1997)
S. Cherukuvada, R. Thakuria, A. Nangia, Cryst. Growth Des. 10, 3931 (2010)
R.A.E. Castro, T.M.R. Maria, A.O.L. Evora, J.C. Feiteira, M.R. Silva, A.M. Beja, J. Canotilho, M.E.S. Eusebio, Cryst. Growth Des. 10, 274 (2010)
V.A. Blatov, Acta Crystallogr. Sect. A Found Crystallogr. 62, 356 (2006)
H. Zabrodsky, S. Peleg, D. Avnir, J. Am. Chem. Soc. 114, 7843 (1992)
M.A. Spackman, P.G. Byrom, Chem. Phys. Lett. 267, 215 (1997)
J.J. McKinnon, M.A. Spackman, A.S. Mitchell, Chem. Eur. J. 4, 2136 (2002)
J.A. Chisholm, S. Motherwell, J. Appl. Crystallogr. 38, 228 (2005)
L. Tabrizi, H. Chiniforoshan, P. Mcardle, Spectrochim. Acta. A 139, 307 (2015)
L. Tabrizi, H. Chiniforoshan, P. Mcardle, J. Coord. Chem. (2015). doi:10.1080/00958972.2015.1036749
Rigaku, CrystalClear, Version 14.0. (Rigaku Corporation, Tokyo, 2005)
G.M. Sheldrick, SHELXS97: Programs for Crystal Structure Analysis (University of Göttingen, Germany, 1997)
K. Brandenburg, DIAMOND: Crystal and Molecular Structure Visualization, Version 3.1b. (Crystal Impact GbR, Bonn, 2006)
Mercury 2.3 Supplied with Cambridge Structural Database, (CCDC, Cambridge, 2003–2004)
Y.-H. Luo, Q.-X. Mao, B.-W. Sun, Inorg. Chim. Acta 412, 60 (2014)
Y. Li, C.-G. Zhang, L.-Y. Cai, Z.-X. Wang, J. Coord. Chem. 66, 3100 (2013)
M.J. Frisch, G.W. Trucks, H.B. Schlegel, Gaussian 09, Revision D.01 (Gaussian, Inc., Wallingford, 2009)
G.-D. Tanga, J.-Y. Zhao, R.-Q. Li, Y. Cao, Z.-C. Zhang, Spectrochim. Acta Part A 78, 449 (2011)
P.E. Elamurugu, S. Muthu, Spectrochim. Acta Part A 134, 453 (2015)
A.M. Pyle, J.P. Rehmann, R. Meshoyrer, C.V. Kumar, N.J. Turro, J.K. Barton, J. Am. Chem. Soc. 111, 3051 (1989)
Acknowledgments
We are very grateful for the financial support from the National Natural Science Foundation of China (No. 21241009, No. 21371031).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, QL., Yang, LJ., Luo, YH. et al. Synthesis, crystal structure, Hirshfeld surface analysis and DNA binding properties of interactions with lattice pyrazinamide and its zinc(II) coordination polymer. Res Chem Intermed 42, 6947–6957 (2016). https://doi.org/10.1007/s11164-016-2506-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2506-y