Abstract
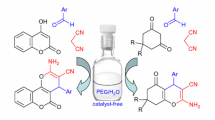
A three-component reaction of benzaldehyde, methyl 5-aminobenzofuran-2-carboxylate, and cyclohexane-1,3-dione in refluxing EtOH under catalyst-free conditions regioselectively gave furo[3,2-a]acridine rather than furo[3,2-b]acridine derivatives. This new procedure has the advantages of one-pot reaction, catalyst-free nature, high yield, and high regioselectivity.
Graphical Abstract







Similar content being viewed by others
References
M.A. Horstmann, W.A. Hassenpflug, U. ZurStadt, G. Escherich, G. Janka, H. Kabisch, Haematologica 90, 1701 (2005)
R. Perrone, E. Butovskaya, D. Daelemans, G. Palu, C. Pannecouque, S.N. Richter, J. Antimicrob. Chemother. 69, 3248 (2014)
X. Li, D. Ma, H. Yang, G. Tan, H. Du, K. Wang, P. Zhang, H. Chen, Gaodeng Xuexiao Huaxue Xuebao 35, 1181 (2014)
C.-J. Lee, C.-H. Yue, Y.-J. Lin, Y.-Y. Lin, S.-H. Kao, J.-Y. Liu, Y.-H. Chen, Anticancer Res. 34, 6467 (2014)
A.H. Al Hamzi, A. Amine, F. Guenoun, N. Moukrad, F.R. Filali, A. Chebaibi, Phys. Chem. News 70, 78(2013)
J.P. Joubert, F.J. Smit, L. du Plessis, P.J. Smith, D.D. N’Da, Eur. J. Pharm. Sci. 56, 16 (2014)
S. Hamulakova, J. Imrich, L. Janovec, P. Kristian, I. Danihel, O. Holas, M. Pohanka, S. Bohm, M. Kozurkova, K. Kuca, Int. J. Biol. Macromol. 70, 435 (2014)
M. Tot, D.M. Opsenica, M. Mitric, J.C. Burnett, L. Gomba, S. Bavari, B.A. Solaja, J. Serb. Chem. Soc. 78, 1847 (2013)
P. Olszewska, E. Mikiciuk-Olasik, K. Blaszczak-Swiatkiewicz, J. Szymanski, P. Szymanski, Biomed. Pharmacother. 68, 959 (2014)
P. Olszewska, J. Szymanski, E. Mikiciuk-Olasik, P. Szymanski, Eur. J. Pharm. 729, 30 (2014)
I. Yesildag, R. Ulus, E. Basar, M. Aslan, M. Kaya, M. Bulbul, Monatsh. Chem. 145, 1027 (2014)
G. Chauvière, B. Bouteille, B. Enanga, C. de Albuquerque, S.L. Croft, M. Dumas, J. Périé, J. Med. Chem. 46, 427 (2003)
M.A. Munawar, P.W. Groundwater, J. Chem. Soc. Pakistan 21, 408 (1999)
C. Jolivet, C. Rivalle, A. Croisy, E. Bisagni, Heterocycles 43, 641 (1996)
J. Reisch, G.M. Kamal, B. Gunaherath, J. Heterocycl. Chem. 26, 1849 (1989)
G.M. Coppola, J. Heterocycl. Chem. 21, 1569 (1984)
J. Reisch, I. Mester, S.K. Kapoor, Z. Rozsa, K. Szendrei, Liebigs. Ann. Chem. 1981, 85 (1981)
K. Takagi, T. Ueda, Chem. Pharm. Bull. 20, 2051 (1972)
K. Takagi, T. Ueda, Chem. Pharm. Bull. 20, 380 (1972)
C. Li, W.T. Zhang, X.S. Wang, Tetrahedron 70, 8919 (2014)
W. Wang, M.M. Zhang, X.S. Wang, J. Heterocycl. Chem. 51, 175 (2014)
C. Li, X.Y. Mu, Y.L. Li, Y. Liu, X.S. Wang, ACS Comb. Sci. 15, 267 (2013)
D.S. Chen, Y.L. Li, Y. Liu, X.S. Wang, Tetrahedron 69, 7045 (2013)
M.M. Zhang, W. Wang, X.S. Wang, J. Heterocycl. Chem. 51, E349 (2014)
X.S. Wang, D.Q. Shi, S.J. Tu, J. Chem. Cryst. 32, 369 (2002)
X.S. Wang, D.Q. Shi, S.J. Tu, C.S. Yao, Chin. J. Struct. Chem. 23, 91 (2004)
H. Jiang, X.S. Wang, M.M. Zhang, C.S. Yao, S.J. Tu, Acta Cryst. E62, o5587 (2006)
Acknowledgments
We are grateful to the Major Natural Science Foundation of Jiangsu Province (14KJA150004), and a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions for financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11164_2015_1998_MOESM1_ESM.pdf
Copies of NMR spectra for compounds 4a–m are available free of charge via the Internet. Crystallographic data for the structure reported in this paper have been deposited with the Cambridge Crystallographic Data Centre as supplementary publication no. CCDC-1051303. Copies of available material can be obtained, free of charge, on application to the Director, CCDC, 12 Union Road, Cambridge CB2 1EZ, UK [fax: +44 (0)1223-336033 or e-mail: deposit@ccdc.cam.ac.uk]. (PDF 472 kb)
Rights and permissions
About this article
Cite this article
Zhang, Y., Xu, J., Zhang, MM. et al. A highly regioselective synthesis of functionalized furo[3,2-a]acridine derivatives via a three-component reaction. Res Chem Intermed 41, 9917–9927 (2015). https://doi.org/10.1007/s11164-015-1998-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-015-1998-1