Abstract
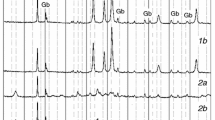
In this study, the solid-state reaction mechanism and kinetics were investigated for production of anhydrous sodium metaborate (NaBO2), an industrially and technologically important boron compound. To assess the kinetics of solid-state production of NaBO2, the chemical reaction between borax (Na2B4O7) and sodium hydroxide (NaOH) was investigated by use of the thermal analysis techniques thermogravimetry (TG) and differential thermal analysis (DTA). DTA curves obtained under non-isothermal conditions at different heating rates (5, 10 and 20 °C/min), revealed five endothermic peaks corresponding to five solid-state reactions occurring at 70, 130, 295, 463, and 595 °C. The stages of the solid-state reaction used for production NaBO2 were also analyzed by XRD, which showed that at 70 and 130 °C, Na2B4O7 and NaOH particles contacted between the grains, and diffusion was initiated at the interface. However, there was not yet any observable formation of NaBO2. Formation of NaBO2 was initiated and sustained from 295 to 463 °C, and then completed at 595 °C; the product was anhydrous NaBO2. Activation energies (E a) of the solid-state reactions were calculated from the weight loss based on the Arrhenius model; it was found that in the initial stages of the solid-state reaction E a values were lower than in the last three steps.








Similar content being viewed by others
References
The Report of Chemical Industry, Turkey State Planning Organization, 2001
E.D. Garret, Borates, Handbook of Deposits, Processing, Properties and Use, vol. 401 (Elsevier, San Diego, 1998)
Y. Kojima, T. ve Haga, Recycling process of sodium metaborate to sodium borohydride. Int. J. Hydrogen Energy 28, 989–993 (2003)
Z.P. Li, B.H. Liu, N. Arai, S. ve Suda, Protide compounds in hydrogen storage systems. J. Alloy. Compd. 356–357, 469–474 (2003)
U.B. Demirci, P. ve Miele, Sodium tetrahydroborate as energy/hydrogen carrier, its history. C. R. Chimie 12, 943–950 (2009)
Z.P. Li, B.H. Liu, J.K. Zhu, N. Morigasaki, S. ve Suda, NaBH4 formation mechanism by reaction of sodium borate with Mg and H2. J. Alloy. Compd. 437, 311–316 (2007)
B.H. Liu, N. Morigasaki, S. Suda, Kinetics characteristics of sodium borohydride formation when sodium meta-borate reacts with magnesium and hydrogen. Int. J. Hydrogen Energy 33, 1323–1328 (2008)
H.B. Liu, Z.P. Li, J.K. ve Zhu, Sodium borohydride formation when mg reacts with hydrous sodium borates under hydrogen. J. Alloy. Compd. 476, L19–L20 (2009)
H. Zhang, S. Zheng, F. Fand, G. Chen, G. Sang, D. ve Sun, Synthesis of NaBH4 based on a solid-state reaction under Ar atmosphere. J. Alloy. Compd. 484, 352–355 (2009)
T. Kemmitt, G.J. ve Gainsford, Regeneration of sodium borohydride from sodium metaborate and isolation of intermediate compounds. Int. J. Hydrogen Energy 34, 5726–5731 (2009)
USP 2.534.533. Methods of Preparing Alkali Metal Borohydrides, United State Patent (1950)
H.I. Schlesinger, H.C. Ve Brown, Uranium (IV) borohydride. J. Am. Chem. Soc. 75, 219–221 (1953)
H.I. Schlesinger, H.C. ve Brown, Metallo borohydrides. Lithium borohydride. J. Am. Chem. Soc 62, 3429–3435 (1940)
H.I. Schlesinger, R.T. Sanderson, A.B. ve Burg, Metallo borohydrides. I. Aluminum borohydride. J. Am. Chem. Soc. 62, 3421–3425 (1940)
H.I. Schlesinger, H.C. Brown, H.R. Hoekstra, L.R. ve Rapp, Reactions of diborane with alkali metal hydrides and their addition compounds. New syntheses of borohydrides. sodium and potassium borohydrides. J. Am. Chem. Soc. 75, 199–204 (1953)
H.I. Schlesinger, H.C. Brown, A.E. ve Finholt, The preparation of sodium borohydride by the high temperature reaction of sodium hydride with borate esters. J. Am. Chem. Soc. 75, 205–209 (1953)
H. Güler, F. Kurtuluş, E. Ay, G. Çelik, I. Doğan. Solid-state and microwave-assisted synthesis and characterization of Mg2B2O5 and Mg3(BO3)2, processing of 4. international boron symposium Eskişehir, (2009), 129–137
Y. Chen, L.T. Chadderton, J.F. Gerald, J.S. Williams, A solid-state process for formation of boron nitride nanotubes. Appl. Phys. Lett. 74, 20 (1999)
A. Kanturk, M. Sari, S. Piskin, Synthesis, crystal structure and dehydration kinetics of NaB(OH)4⋅2H2O. Korean J. Chem. Eng. 25, 1331 (2008)
A. Kanturk, S. Piskin, Parametric investigation on anhydrous sodium metaborate (NaBO2) synthesis from concentrated tincal. Adv. Powder Technol 21, 513–520 (2010)
A. Kanturk, H. Erguven, S. Piskin, in Investigation of solid state reaction mechanism for sodium metaborate (NaBO 2 ) production. Supplemental Proceedings: Materials Processing and Energy Materials, vol 1 (2011), pp. 585–589. doi:10.1002/9781118062111.ch67
S.M. Yılmaz, K.A. Figen, S. Pişkin, Production of sodium metaborate tetrahydrate (NaB(OH)4⋅2H2O) using ultrasonic irradiation P. Powder Technol. 215–216, 166–173 (2012)
H.E. Kissinger, Variation of peak temperature with heating rate in differential thermal analysis. J. Res. Natl. Bur. Stand. 57(4), 217–221 (1956)
TSE 273, Sodium perborates for industrial use determination of bulk density, (ISO 3424) (1975)
O. Buckman Harry, C. Brady Nyle, The Nature and Property of Soils—A College Text of Edaphology, 6th ed. (MacMillan Publishers, New York, 1960)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pişkin, M.B., Figen, A.K. & Ergüven, H. Investigation of the reaction mechanism and kinetics of production of anhydrous sodium metaborate (NaBO2) by a solid-state reaction. Res Chem Intermed 39, 569–583 (2013). https://doi.org/10.1007/s11164-012-0580-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-012-0580-3