Abstract
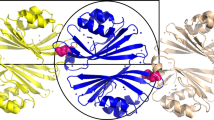
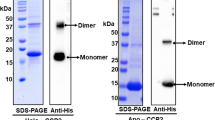
The photoswitchable orange carotenoid protein (OCP) is indispensable for cyanobacterial photoprotection by quenching phycobilisome fluorescence upon photoconversion from the orange OCPO to the red OCPR form. Cyanobacterial genomes frequently harbor, besides genes for orange carotenoid proteins (OCPs), several genes encoding homologs of OCP’s N- or C-terminal domains (NTD, CTD). Unlike the well-studied NTD homologs, called Red Carotenoid Proteins (RCPs), the role of CTD homologs remains elusive. We show how OCP can be reassembled from its functional domains. Expression of Synechocystis OCP-CTD in carotenoid-producing Escherichia coli yielded violet-colored proteins, which, upon mixing with the RCP-apoprotein, produced an orange-like photoswitchable form that further photoconverted into a species that quenches phycobilisome fluorescence and is spectroscopically indistinguishable from RCP, thus demonstrating a unique carotenoid shuttle mechanism. Spontaneous carotenoid transfer also occurs between canthaxanthin-coordinating OCP-CTD and the OCP apoprotein resulting in formation of photoactive OCP. The OCP-CTD itself is a novel, dimeric carotenoid-binding protein, which can coordinate canthaxanthin and zeaxanthin, effectively quenches singlet oxygen and interacts with the Fluorescence Recovery Protein. These findings assign physiological roles to the multitude of CTD homologs in cyanobacteria and explain the evolutionary process of OCP formation.





Similar content being viewed by others
Abbreviations
- OCPO :
-
Orange carotenoid protein—holoprotein, orange form
- OCPR :
-
Orange carotenoid protein—holoprotein, red form
- RCP:
-
Red carotenoid protein—holoprotein
- Apo-RCP:
-
N-terminal domain of Synechocystis OCP (amino acids 1-164)—apoprotein
- COCP:
-
C-terminal OCP-related carotenoid protein (C-terminal domain of Synechocystis OCP, amino acids 165–317)—holoprotein
- Apo-COCP:
-
C-terminal domain of Synechocystis OCP (amino acids 165–317)—apoprotein
- Synechocystis :
-
Synechocystis sp. PCC 6803
- FRP:
-
Fluorescence recovery protein
- HCP:
-
Helical carotenoid protein
- StARD1:
-
Human steroidogenic acute regulatory protein
- ECN:
-
Echinenone (β,β-Carotene-4-one)
- CAN:
-
Canthaxanthin (β,β-Carotene-4,4′-dione)
- ZEA:
-
Zeaxanthin (β,β-Carotene-4,4′-diol)
- NTD:
-
N-terminal domain
- CTD:
-
C-terminal domain
- NPQ:
-
Non-photochemical quenching
- PBs:
-
Phycobilisome
- E. coli :
-
Escherichia coli
- SDS–PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- LC-MS:
-
Liquid chromatography-mass spectrometry
- RR:
-
Resonance Raman
- Mw:
-
Molecular weight
- R H :
-
Hydrodynamic radius
- R g :
-
Radius of gyration
- ROS:
-
Reactive oxygen species
- SOSG:
-
Singlet oxygen sensor green
- 1O2 :
-
Singlet oxygen
References
Abraham MJ, Murtola T, Schulz R, Páll S, Smith JC, Hess B, Lindahl E (2015) GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1–2:19–25. doi:10.1016/j.softx.2015.06.001
Bhosale P, Larson AJ, Frederick JM, Southwick K, Thulin CD, Bernstein PS (2004) Identification and characterization of a Pi isoform of glutathione S-transferase (GSTP1) as a zeaxanthin-binding protein in the macula of the human eye. J Biol Chem 279(47):49447–49454. doi:10.1074/jbc.M405334200
Blankenship RE (2014) Molecular Mechanism of Photosynthesis. 2 edn. Wiley-Blackwell, Oxford, London, Edinburgh
Boulay C, Wilson A, D’Haene S, Kirilovsky D (2010) Identification of a protein required for recovery of full antenna capacity in OCP-related photoprotective mechanism in cyanobacteria. Proc Natl Acad Sci USA 107(25):11620–11625. doi:10.1073/pnas.1002912107
Britton G (1995) UV/Visible spectroscopy. In: Britton G, Liaaen-Jensen S, Pfander H (eds) Carotenoids, vol 1B. Birkhäuser, Basel
Bryant DA (ed) (1994) The molecular biology of cyanobacteria, vol 1. Advances in photosynthesis and respiration. Springer, Dordrecht
Chabera P, Durchan M, Shih PM, Kerfeld CA, Polivka T (2011) Excited-state properties of the 16 kDa red carotenoid protein from Arthrospira maxima. Biochim Biophys Acta 1807(1):30–35. doi:10.1016/j.bbabio.2010.08.013
Cho H-S, Choi G, Choi KY, Oh B-H (1998) Crystal structure and enzyme mechanism of ∆5-3-ketosteroid isomerase from Pseudomonas testosteroni. Biochemistry 37(23):8325–8330. doi:10.1021/bi9801614
Choi S-K, Nishida Y, Matsuda S, Adachi K, Kasai H, Peng X, Komemushi S, Miki W, Misawa N (2005) Characterization of β-carotene Ketolases, CrtW, from marine bacteria by complementation analysis in Escherichia coli. Marine Biotechnol 7 (5):515–522. doi:10.1007/s10126-004-5100-z
Cianci M, Rizkallah PJ, Olczak A, Raftery J, Chayen NE, Zagalsky PF, Helliwell JR (2002) The molecular basis of the coloration mechanism in lobster shell: β-Crustacyanin at 3.2-Å resolution. Proc Natl Acad Sci USA 99:9795–9800. doi:10.1073/pnas.152088999
de Carbon CB, Thurotte A, Wilson A, Perreau F, Kirilovsky D (2015) Biosynthesis of soluble carotenoid holoproteins in Escherichia coli. Sci Rep 5:9085. doi:10.1038/srep09085
Demmig-Adams B, Adams WW (1996) The role of xanthophyll cycle carotenoids in the protection of photosynthesis. Trends Plant Sci 1(1):21–26. doi:10.1016/S1360-1385(96)80019-7
Demmig-Adams B, Garab G, Adams WW III, Govindjee (eds) (2014) Non-photochemical quenching and energy dissipation in plants, algae and cyanobacteria, vol 40. advances in photosynthesis and respiration. Springer, Dordrecht
Gamiz-Hernandez AP, Angelova IN, Send R, Sundholm D, Kaila VRI (2015) Protein-induced color shift of carotenoids in β-Crustacyanin. Angew Chem Intl Ed 54 (39):11564–11566. doi:10.1002/anie.201501609
Govindjee, Shevela D (2011) Adventures with cyanobacteria: a personal perspective. Front Plant Sci 2(28):1–17. doi:10.3389/fpls.2011.00028
Gupta S, Guttman M, Leverenz RL, Zhumadilova K, Pawlowski EG, Petzold CJ, Lee KK, Ralston CY, Kerfeld CA (2015) Local and global structural drivers for the photoactivation of the orange carotenoid protein. Proc Natl Acad Sci USA 112(41):E5567–E5574. doi:10.1073/pnas.1512240112
Gwizdala M, Wilson A, Kirilovsky D (2011) In vitro reconstitution of the cyanobacterial photoprotective mechanism mediated by the orange carotenoid protein in Synechocystis PCC 6803. Plant Cell 23(7):2631–2643. doi:10.1105/tpc.111.086884
Gwizdala M, Wilson A, Omairi-Nasser A, Kirilovsky D (2013) Characterization of the Synechocystis PCC 6803 fluorescence recovery protein involved in photoprotection. Biochim Biophys Acta 1827(3):348–354. doi:10.1016/j.bbabio.2012.11.001
Kauppi B, Lee K, Carredano E, Parales RE, Gibson DT, Eklund H, Ramaswamy S (1998) Structure of an aromatic-ring-hydroxylating dioxygenase—naphthalene 1,2-dioxygenase. Structure 6(5):571–586. doi:10.1016/S0969-2126(98)00059-8
Kawasaki S, Mizuguchi K, Sato M, Kono T, Shimizu H (2013) A novel astaxanthin-binding photooxidative stress-inducible aqueous carotenoprotein from a eukaryotic microalga isolated from asphalt in midsummer. Plant Cell Physiol 54(7):1027–1040. doi:10.1093/pcp/pct080
Kerfeld CA, Sawaya MR, Brahmandam V, Cascio D, Ho KK, Trevithick-Sutton CC, Krogmann DW, Yeates TO (2003) The crystal structure of a cyanobacterial water-soluble carotenoid binding protein. Structure 11(1):55–65. doi:10.1016/S0969-2126(02)00936-X
King JD, Liu H, He G, Orf GS, Blankenship RE (2014) Chemical activation of the cyanobacterial orange carotenoid protein. FEBS Lett 588(24):4561–4565. doi:10.1016/j.febslet.2014.10.024
Kirilovsky D, Kerfeld CA (2012) The orange carotenoid protein in photoprotection of photosystem II in cyanobacteria. Biochim Biophys Acta 1817(1):158–166. doi:10.1016/j.bbabio.2011.04.013
Kish E, Pinto MM, Kirilovsky D, Spezia R, Robert B (2015) Echinenone vibrational properties: from solvents to the orange carotenoid protein. Biochim Biophys Acta 1847(10):1044–1054. doi:10.1016/j.bbabio.2015.05.010
Leverenz RL, Jallet D, Li MD, Mathies RA, Kirilovsky D, Kerfeld CA (2014) Structural and functional modularity of the orange carotenoid protein: distinct roles for the N- and C-terminal domains in cyanobacterial photoprotection. Plant Cell 26(1):426–437. doi:10.1105/tpc.113.118588
Leverenz RL, Sutter M, Wilson A, Gupta S, Thurotte A, Bourcier de Carbon C, Petzold CJ, Ralston C, Perreau F, Kirilovsky D, Kerfeld CA (2015) PHOTOSYNTHESIS. A 12 Å carotenoid translocation in a photoswitch associated with cyanobacterial photoprotection. Science 348(6242):1463–1466. doi:10.1126/science.aaa7234
Li B, Vachali P, Frederick JM, Bernstein PS (2011) Identification of StARD3 as a lutein-binding protein in the macula of the primate retina. Biochemistry 50(13):2541–2549. doi:10.1021/bi101906y
López-Igual R, Wilson A, Leverenz RL, Melnicki MR, Bourcier de Carbon C, Sutter M, Turmo A, Perreau F, Kerfeld CA, Kirilovsky D (2016) Different functions of the paralogs to the N-terminal domain of the orange carotenoid protein in the Cyanobacterium Anabaena sp. PCC 7120. Plant Physiol 171(3):1852–1866. doi:10.1104/pp.16.00502
Luecke H, Schobert B, Stagno J, Imasheva ES, Wang JM, Balashov SP, Lanyi JK (2008) Crystallographic structure of xanthorhodopsin, the light-driven proton pump with a dual chromophore. Proc Natl Acad Sci USA 105(43):16561–16565. doi:10.1073/pnas.0807162105
Maksimov EG, Schmitt FJ, Shirshin EA, Svirin MD, Elanskaya IV, Friedrich T, Fadeev VV, Paschenko VZ, Rubin AB (2014) The time course of non-photochemical quenching in phycobilisomes of Synechocystis sp. PCC6803 as revealed by picosecond time-resolved fluorimetry. Biochim Biophys Acta 1837(9):1540–1547. doi:10.1016/j.bbabio.2014.01.010
Maksimov EG, Gvozdev DA, Strakhovskaya MG, Paschenko VZ (2015a) Hybrid structures of polycationic aluminum phthalocyanines and quantum dots. Biochemistry (Moscow) 80(3):323–331. doi:10.1134/S0006297915030074
Maksimov EG, Klementiev KE, Shirshin EA, Tsoraev GV, Elanskaya IV, Paschenko VZ (2015b) Features of temporal behavior of fluorescence recovery in Synechocystis sp. PCC6803. Photosynth Res 125(1–2):167–178. doi:10.1007/s11120-015-0124-y
Maksimov EG, Shirshin EA, Sluchanko NN, Zlenko DV, Parshina EY, Tsoraev GV, Klementiev KE, Budylin GS, Schmitt FJ, Friedrich T, Fadeev VV, Paschenko VZ, Rubin AB (2015c) The signaling state of orange carotenoid protein. Biophys J 109(3):595–607. doi:10.1016/j.bpj.2015.06.052
Maksimov EG, Moldenhauer M, Shirshin EA, Parshina EA, Sluchanko NN, Klementiev KE, Tsoraev GV, Tavraz NN, Willoweit M, Schmitt FJ, Breitenbach J, Sandmann G, Paschenko VZ, Friedrich T, Rubin AB (2016) A comparative study of three signaling forms of the orange carotenoid protein. Photosynth Res 130(1–3):389–401. doi:10.1007/s11120-016-0272-8
Maksimov EG, Sluchanko NN, Mironov KS, Shirshin EA, Klementiev KE, Tsoraev GV, Moldenhauer M, Friedrich T, Los DA, Allakhverdiev SI, Paschenko VZ, Rubin AB (2017) Fluorescent labeling preserving OCP photoactivity reveals its reorganization during the photocycle. Biophys J 112:46–56. doi:10.1016/j.bpj.2016.11.3193
Mamedov M, Govindjee, Nadtochenko V, Semenov A (2015) Primary electron transfer processes in photosynthetic reaction centers from oxygenic organisms. Photosynth Res 125(1):51–63. doi:10.1007/s11120-015-0088-y
Melnicki MR, Leverenz RL, Sutter M, Lopez-Igual R, Wilson A, Pawlowski EG, Perreau F, Kirilovsky D, Kerfeld CA (2016) Structure, diversity, and evolution of a new family of soluble carotenoid-binding proteins in cyanobacteria. Mol Plant 9 (10):1379–1394. doi:10.1016/j.molp.2016.06.009
Mendes-Pinto MM, Sansiaume E, Hashimoto H, Pascal AA, Gall A, Robert B (2013) Electronic absorption and ground state structure of carotenoid molecules. J Phys Chem B 117(38):11015–11021. doi:10.1021/jp309908r
Mirkovic T, Ostroumov EE, Anna JM, van Grondelle R, Govindjee, Scholes GD (2016) Light absorption and energy transfer in the antenna complexes of photosynthetic organisms. Chem Rev 117(2):249–293. doi:10.1021/acs.chemrev.6b00002
Misawa N, Satomi Y, Kondo K, Yokoyama A, Kajiwara S, Saito T, Ohtani T, Miki W (1995) Structure and functional analysis of a marine bacterial carotenoid biosynthesis gene cluster and astaxanthin biosynthetic pathway proposed at the gene level. J Bacteriol 177(22):6575–6584. doi:10.1128/jb.177.22.6575-6584.1995
Mori Y (2016) Computational study on the color change of 3′-hydroxyechinenone in the orange carotenoid protein. Chem Phys Lett 652:184–189. doi:10.1016/j.cplett.2016.04.062
Niyogi KK, Truong TB (2013) Evolution of flexible non-photochemical quenching mechanisms that regulate light harvesting in oxygenic photosynthesis. Curr Opin Plant Biol 16(3):307–314. doi:10.1016/j.pbi.2013.03.011
Schlücker S, Szeghalmi A, Schmitt M, Popp J, Kiefer W (2003) Density functional and vibrational spectroscopic analysis of β-carotene. J Raman Spectr 34(6):413–419. doi:10.1002/jrs.1013
Schmitt F-J, Renger G, Friedrich T, Kreslavski VD, Zharmukhamedov SK, Los DA, Kuznetsov VV, Allakhverdiev SI (2014) Reactive oxygen species: re-evaluation of generation, monitoring and role in stress-signaling in phototrophic organisms. Biochim Biophys Acta 1837(6):835–848. doi:10.1016/j.bbabio.2014.02.005
Sedoud A, Lopez-Igual R, Ur Rehman A, Wilson A, Perreau F, Boulay C, Vass I, Krieger-Liszkay A, Kirilovsky D (2014) The cyanobacterial photoactive orange carotenoid protein is an excellent singlet oxygen quencher. Plant Cell 26(4):1781–1791. doi:10.1105/tpc.114.123802
Shevela D, Pishchalinikov RY, Eichacker LA, Govindjee (2013) Oxygenic photosynthesis in cyanobacteria. In: Srivastava AK, Rai AN, Neilan BA (eds) Stress biology of cyanobacteria. Taylor&Francis, Boca Raton, London, New York
Sluchanko NN, Tugaeva KV, Faletrov YV, Levitsky DI (2016) High-yield soluble expression, purification and characterization of human steroidogenic acute regulatory protein (StAR) fused to a cleavable Maltose-Binding Protein (MBP). Prot Expr Purif 119:27–35. doi:10.1016/j.pep.2015.11.002
Sluchanko NN, Klementiev KE, Shirshin EA, Tsoraev GV, Friedrich T, Maksimov EG (2017) The purple Trp288Ala mutant of Synechocystis OCP persistently quenches phycobilisome fluorescence and tightly interacts with FRP. Biochim Biophys Acta 1858:1–11. doi:10.1016/j.bbabio.2016.10.005
Sutter M, Wilson A, Leverenz RL, Lopez-Igual R, Thurotte A, Salmeen AE, Kirilovsky D, Kerfeld CA (2013) Crystal structure of the FRP and identification of the active site for modulation of OCP-mediated photoprotection in cyanobacteria. Proc Natl Acad Sci USA 110(24):10022–10027. doi:10.1073/pnas.1303673110
Thurotte A, López-Igual R, Wilson A, Comolet L, Bourcier de Carbon C, Xiao F, Kirilovsky D (2015) Regulation of orange carotenoid protein activity in cyanobacterial photoprotection. Plant Physiol 169(1):737–747. doi:10.1104/pp.15.00843
Velazquez Escobar F, Hildebrandt T, Utesch T, Schmitt FJ, Seuffert I, Michael N, Schulz C, Mroginski MA, Friedrich T, Hildebrandt P (2013) Structural parameters controlling the fluorescence properties of phytochromes. Biochemistry 53(1):20–29. doi:10.1021/bi401287u
Whitmore L, Wallace BA (2004) DICHROWEB, an online server for protein secondary structure analyses from circular dichroism spectroscopic data. Nucleic Acid Res 32 (Web Server issue):W668–W673. doi:10.1093/nar/gkh371
Wilson A, Punginelli C, Gall A, Bonetti C, Alexandre M, Routaboul J-M, Kerfeld CA, van Grondelle R, Robert B, Kennis JTM, Kirilovsky D (2008) A photoactive carotenoid protein acting as light intensity sensor. Proc Natl Acad Sci USA 105(33):12075–12080. doi:10.1073/pnas.0804636105
Wilson A, Kinney JN, Zwart PH, Punginelli C, D’Haene S, Perreau F, Klein MG, Kirilovsky D, Kerfeld CA (2010) Structural determinants underlying photoprotection in the photoactive orange carotenoid protein of cyanobacteria. J Biol Chem 285(24):18364–18375. doi:10.1074/jbc.M110.115709
Acknowledgements
This work was supported by the German Ministry of Education and Research (WTZ-RUS Grant 01DJ15007 to M.M., N.N.T., F.-J.S, and T.F.), the German Research Foundation (Cluster of Excellence “Unifying Concepts in Catalysis” to T.F., D.B., and P.H), the Russian Foundation for Basic Research (Grant 15-04-01930a to E.G.M.), the Russian Science Foundation (no. 14-17-00451 to E.G.M), and the Russian Ministry of Education and Science (Grant MK-5949.2015.4 to E.G.M.). E.G.M. was supported by Dynasty Foundation Fellowship. The study was funded by RFBR and Moscow City Government (research project № 15-34-70007 «mol_а_mos»). The authors thank Prof. N. Budisa and Dr. Tobias Baumann for providing access to CD spectroscopy equipment and Dr. M. Schlangen-Ahl for technical support during LC-MS experiments.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Marcus Moldenhauer and Nikolai N. Sluchanko have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moldenhauer, M., Sluchanko, N.N., Buhrke, D. et al. Assembly of photoactive orange carotenoid protein from its domains unravels a carotenoid shuttle mechanism. Photosynth Res 133, 327–341 (2017). https://doi.org/10.1007/s11120-017-0353-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-017-0353-3