Abstract
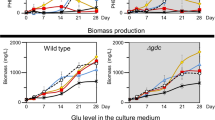
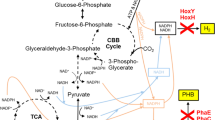
Glycogen synthesis initiated by glucose-1-phosphate adenylyltransferase (glgC) represents a major carbon storage route in cyanobacteria which could divert a significant portion of assimilated carbon. Significant growth retardation in cyanobacteria with glgC knocked out (ΔglgC) has been reported in high light conditions. Here, we knocked out the glgC gene and analyzed its effects on carbon distribution in an isobutanol-producing strain of Synechococcus elongatus PCC7942 and its parental wild-type strain. We showed that isobutanol production was able to partially rescue the growth of ΔglgC mutant where the growth rescue effect positively correlated with the rate of isobutanol production. Using NaH14CO3 incorporation analysis, we observed a 28 % loss of total carbon fixation rate in the ΔglgC mutant compared to the wild-type. Upon expression of the isobutanol production pathway in ΔglgC mutant, the total carbon fixation rate was restored to the wild-type level. Furthermore, we showed that 52 % of the total carbon fixed was redirected into isobutanol biosynthesis in the ΔglgC mutant expressing enzymes for isobutanol production, which is 2.5 times higher than that of the wild-type expressing the same enzymes. These results suggest that biosynthesis of non-native product such as isobutanol can serve as a metabolic sink for replacing glycogen to rescue growth and restore carbon fixation rate. The rescue effect may further serve as a platform for cyanobacteria energy and carbon metabolism study.





Similar content being viewed by others
References
Arp WJ (1991) Effects of source-sink relations on photosynthetic acclimation to elevated CO2. Plant Cell Environ 14(8):869–875. doi:10.1111/j.1365-3040.1991.tb01450.x
Atsumi S, Higashide W, Liao JC (2009) Direct photosynthetic recycling of carbon dioxide to isobutyraldehyde. Nat Biotechnol 27(12):1177–1180
Ballicora MA, Iglesias AA, Preiss J (2003) ADP-glucose pyrophosphorylase, a regulatory enzyme for bacterial glycogen synthesis. Microbiol Mol Biol Rev 67(2):213–225. doi:10.1128/mmbr.67.2.213-225.2003
Carrieri D, Paddock T, Maness P-C, Seibert M, Yu J (2012) Photo-catalytic conversion of carbon dioxide to organic acids by a recombinant cyanobacterium incapable of glycogen storage. Energy Environ Sci 5(11):9457–9461
Dexter J, Fu P (2009) Metabolic engineering of cyanobacteria for ethanol production. Energy Environ Sci 2(8):857–864. doi:10.1039/b811937f
Ducat DC, Sachdeva G, Silver PA (2011) Rewiring hydrogenase-dependent redox circuits in cyanobacteria. Proc Natl Acad Sci 108(10):3941–3946. doi:10.1073/pnas.1016026108
Ducat DC, Avelar-Rivas JA, Way JC, Silver PA (2012) Rerouting carbon flux to enhance photosynthetic productivity. Appl Environ Microbiol 78(8):2660–2668. doi:10.1128/aem.07901-11
Gibson DG, Young L, Chuang R-Y, Venter JC, Hutchison CA, Smith HO (2009) Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat Methods 6(5):343–345
Gómez Casati DF, Aon MA, Iglesias AA (1999) Ultrasensitive glycogen synthesis in Cyanobacteria. FEBS Lett 446(1):117–121
Gründel M, Scheunemann R, Lockau W, Zilliges Y (2012) Impaired glycogen synthesis causes metabolic overflow reactions and affects stress responses in the Cyanobacterium Synechocystis sp. PCC 6803. Microbiology 158(Pt 12):3032–3043. doi:10.1099/mic.0.062950-0
Jacobsen J, Rosgaard L, Sakuragi Y, Frigaard N-U (2011) One-step plasmid construction for generation of knock-out mutants in cyanobacteria: studies of glycogen metabolism in Synechococcus sp. PCC 7002. Photosynth Res 107(2):215–221. doi:10.1007/s11120-010-9613-1
Krapp A, Hofmann B, Schäfer C, Stitt M (1993) Regulation of the expression of rbcS and other photosynthetic genes by carbohydrates: a mechanism for the ‘sink regulation’ of photosynthesis? Plant J 3(6):817–828. doi:10.1111/j.1365-313X.1993.00817.x
Lan EI, Liao JC (2011) Metabolic engineering of cyanobacteria for 1-butanol production from carbon dioxide. Metab Eng 13(4):353–363. doi:10.1016/j.ymben.2011.04.004
Lan EI, Liao JC (2012) ATP drives direct photosynthetic production of 1-butanol in cyanobacteria. Proc Natl Acad Sci 109(16):6018–6023. doi:10.1073/pnas.1200074109
Lan EI, Liao JC (2013) Microbial synthesis of n-butanol, isobutanol, and other higher alcohols from diverse resources. Bioresour Technol 135:339–349. doi:10.1016/j.biortech.2012.09.104
Lee C, Kim J, Shin SG, Hwang S (2006) Absolute and relative QPCR quantification of plasmid copy number in Escherichia coli. J Biotechnol 123(3):273–280
Lindberg P, Park S, Melis A (2010) Engineering a platform for photosynthetic isoprene production in cyanobacteria, using Synechocystis as the model organism. Metab Eng 12(1):70–79. doi:10.1016/j.ymben.2009.10.001
Liu X, Sheng J, Curtiss R III (2011) Fatty acid production in genetically modified cyanobacteria. Proc Natl Acad Sci 108(17):6899–6904. doi:10.1073/pnas.1103014108
Miao X, Wu Q, Wu G, Zhao N (2003a) Changes in photosynthesis and pigmentation in an agp deletion mutant of the cyanobacterium Synechocystis sp. Biotechnol Lett 25(5):391–396. doi:10.1023/a:1022446330284
Miao X, Wu Q, Wu G, Zhao N (2003b) Sucrose accumulation in salt-stressed cells of agp gene deletion-mutant in cyanobacterium Synechocystis sp. PCC 6803. FEMS Microbiol Lett 218(1):71–77. doi:10.1111/j.1574-6968.2003.tb11500.x
Ohnishi N, Mukherjee B, Tsujikawa T, Yanase M, Nakano H, Moroney JV, Fukuzawa H (2010) Expression of a Low CO2–inducible protein, LCI1, increases inorganic carbon uptake in the green alga Chlamydomonas reinhardtii. Plant Cell 22:3105–3117. doi:10.1105/tpc.109.071811
Oliver JWK, Machado IMP, Yoneda H, Atsumi S (2013) Cyanobacterial conversion of carbon dioxide to 2,3-butanediol. Proc Natl Acad Sci 110(4):1249–1254. doi:10.1073/pnas.1213024110
Preiss J, Romeo T (1989) Physiology, biochemistry and genetics of bacterial glycogen synthesis. Adv Microb Physiol 30:183–238
Shen CR, Liao JC (2012) Photosynthetic production of 2-methyl-1-butanol from CO2 in cyanobacterium Synechococcus elongatus PCC7942 and characterization of the native acetohydroxyacid synthase. Energy Environ Sci 5(11):9574–9583. doi:10.1039/c2ee23148d
Stal LJ, Moezelaar R (1997) Fermentation in cyanobacteria. FEMS Microbiol Rev 21(2):179–211. doi:10.1016/S0168-6445(97)00056-9
Suzuki E, Ohkawa H, Moriya K, Matsubara T, Nagaike Y, Iwasaki I, Fujiwara S, Tsuzuki M, Nakamura Y (2010) Carbohydrate metabolism in mutants of the cyanobacterium Synechococcus elongatus PCC 7942 defective in glycogen synthesis. Appl Environ Microbiol 76(10):3153–3159. doi:10.1128/aem.00397-08
Tan X, Yao L, Gao Q, Wang W, Qi F, Lu X (2011) Photosynthesis driven conversion of carbon dioxide to fatty alcohols and hydrocarbons in cyanobacteria. Metab Eng 13(2):169–176. doi:10.1016/j.ymben.2011.01.001
Vonshak A, Chanawongse L, Bunnag B, Tanticharoen M (1996) Light acclimation and photoinhibition in three Spirulina platensis (cyanobacteria) isolates. J Appl Phycol 8(1):35–40. doi:10.1007/bf02186220
Zhou J, Zhang H, Zhang Y, Li Y, Ma Y (2012) Designing and creating a modularized synthetic pathway in cyanobacterium Synechocystis enables production of acetone from carbon dioxide. Metab Eng 14(4):394–400. doi:10.1016/j.ymben.2012.03.005
Acknowledgments
The authors would like to thank Ethan I. Lan for the plasmids and valuable discussions. This work is supported by PETRO program of the Advanced Research Projects Agency–Energy (ARPA-E) of DOE (Award number DE-AR0000201). This project is also partially supported by NSF MCB-1139318 and UCLA-DOE Institute for Genomics and Proteomics.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, X., Shen, C.R. & Liao, J.C. Isobutanol production as an alternative metabolic sink to rescue the growth deficiency of the glycogen mutant of Synechococcus elongatus PCC 7942. Photosynth Res 120, 301–310 (2014). https://doi.org/10.1007/s11120-014-9987-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-014-9987-6