Abstract
Backgroud and aims
This study was conducted to reveal the genetic diversity of soybean-nodulating rhizobia in Nepal in relation to climate and soil properties.
Method
A total of 102 bradyrhizobial strains were isolated from the root nodules of soybeans cultivated in 12 locations in Nepal varying in climate and soil properties, and their genetic diversity was examined based on 16S rDNA, ITS regions of 16S–23S rDNA, nodC and nifH. In vitro growth properties of some representative strains were examined to elucidate their characteristic distribution in Nepal.
Results
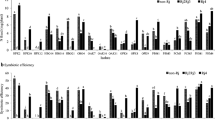
Four species of the genus Bradyrhizobium were isolated, and B. japonicum dominated at temperate locations, while in subtropical locations, B. elkanii, B. yuanmingense, and B. liaoningense dominated at acidic, moderately acidic, and slightly alkaline soils, respectively. The relative nodule occupancies could not be fully explained by their in vitro growth properties. Similar nodC and nifH genes among the strains suggested co-evolution of these genes also in Nepal, probably through horizontal gene transfer.
Conclusions
The influence of climate and soil pH on diversity at the sub-species level was revealed. It is concluded that the highly diverse climate and soils in Nepal might be conducive for the existence of diverse soybean rhizobial strains.







Similar content being viewed by others
References
Alberton O, Kaschuk G, Hungria M (2006) Sampling effects on the assessment of genetic diversity of rhizobia associated with soybean and common bean. Soil Biol Biochem 38:1298–1307
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Ando S, Yokoyama T (1999) Phylogenetic analyses of Bradyrhizobium strains nodulating soybean (Glycine max) in Thailand with reference strains of Bradyrhizobium. Can J Microbiol 45:639–645
Anyango B, Wilson KJ, Beynon JL, Giller KE (1995) Diversity of rhizobia nodulating Phaseolus vulgaris L. in two Kenyan soils with contrasting pHs. Appl Environ Microbiol 61:4016–4021
Appunu C, Dhar B (2006) Symbiotic effectiveness of acid-tolerant Bradyrhizobium strains with soybean in low pH soil. Afr J Biotech 5:842–845
Appunu C, N’Zoue A, Laguerre G (2008) Genetic diversity of native Bradyrhizobia isolated from soybeans (Glycine max L.) in different agricultural-ecological-climatic regions of India. Appl Environ Microbiol 74:5991–5996
Appunu C, Sasirekha N, Prabavathi VR, Nair S (2009) A significant proportion of indigenous rhizobia from India associated with soybean (Glycine max L.) distinctly belong to Bradyrhizobium and Ensifer genera. Biol Fertil Soils 46:57–63
Asanuma S, Ayanaba A (1990) Variation in acid-Al tolerance of Bradyrhizobium japonicum strains from African soils. Soil Sci Plant Nutr 36:309–317
Brockwell J, Pilka A, Holliday RA (1991) Soil pH is a major determinant of the numbers of naturally-occurring Rhizobium meliloti in non-cultivated soils of New South Wales. Aust J Exp Agric 31:211–219
Brockwell J, Bottomley PJ, Thies JE (1995) Manipulation of rhizobia microflora for improving legume productivity and soil fertility: a critical assessment. Plant Soil 174:143–180
Chen WX, Yan GH, Li JL (1988) Numerical taxonomic study of fast-growing soybean rhizobia and a proposal that Rhizobium fredii be assigned to Sinorhizobium gen. nov. Int J Syst Bacteriol 38:392–397
Chen W, Wang E, Wang S, Li Y, Chen X, Li Y (1995) Characteristics of Rhizobium tianshanense sp. nov., a moderately and slowly growing root nodule bacterium isolated from an arid saline environment in Xinjiang, People’s Republic of China. Int J Syst Evol Microbiol 45:153–159
Chen LS, Figueredo A, Pedrosa FO, Hungria M (2000) Genetic characterization of soybean rhizobia in Paraguay. Appl Environ Microbiol 66:5099–5103
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci USA 103:626–631
Graham PH (1992) Stress tolerance in Rhizobium and Bradyrhizobium, and nodulation under adverse soil conditions. Can J Microbiol 38:475–484
Graham PH, Viteri SE, Mackie F, Vargas AT, Palacios A (1982) Variation in acid soil tolerance among strains of Rhizobium phaseoli. Field Crops Res 5:121–128
Han LL, Wang TT, Han TX, Liu J, Sui XH, Chen WF, Chen WX (2009) Unique community structure and biogeography of soybean rhizobia in the saline-alkaline soils of Xinjiang, China. Plant Soil 324:291–305
Haukka K, Lindstrom K, Young JPW (1998) Three phylogenetic groups of nodA and nifH genes in Sinorhizobium and Mesorhizobium isolates from leguminous trees growing in Africa and Latin America. Appl Environ Microbiol 64:419–426
Jordan DC (1982) Transfer of Rhizobium japonicum Buchanan 1980 to Bradyrhizobium gen. nov., a genus of slow-growing, root nodule bacteria from leguminous plants. Int J Syst Bacteriol 32:136–139
Knudsen D, Peterson GA, Pratt PF (1982) Lithium, sodium and potassium. In: Page et al (eds) Methods of soil analysis, part 2: chemical and microbiological properties - agronomy monograph no. 9, 2nd edn. American Society of Agronomy, Soil Science Society of America, Wisconsin, pp 225–246
Kyukendall LD, Saxena B, Devine TE, Udell SE (1992) Genetic diversity in Bradyrhizobium japonicum Jordan 1982 and a proposal for Bradyrhizobium elkanii sp. nov. Can J Microbiol 38:501–505
LaFavre AK, Eaglesham ARJ (1986) The effects of high temperature on soybean nodulation and growth with different strains of bradyrhizobia. Can J Microbiol 32:22–27
Laguerre G, Nour SM, Macheret V, Sanjuan J, Drouin P, Amarger N (2001) Classification of rhizobia based on nodC and nifH gene analysis reveals a close phylogenetic relationship among Phaseolus vulgaris symbionts. Microbiology 147:981–993
Laranjo M, Rodrigues R, Alho L, Oliveira S (2001) Rhizobia of chickpea from southern Portugal: symbiotic efficiency and genetic diversity. J Appl Microbiol 90:662–667
Li QQ, Wang ET, Zhang YZ, Zhang YM, Tian CF, Sui XH, Chen WF, Chen WX (2011) Diversity and biogeography of rhizobia isolated from root nodules of Glycine max grown in Hebei Province, China. Microb Ecol 61:917–931
Man CX, Wang H, Chen WF, Sui XH, Wang ET, Chen WX (2008) Diverse rhizobia associated with soybean grown in the subtropical and tropical regions of China. Plant Soil 310:77–87
Martinez-Romero E, Caballero-Mellado J (1996) Rhizobium phylogenies and bacterial genetic diversity. Crit Rev Plant Sci 15:113–140
Olsen SR, Cole CV, Watanabe FS, Dean LA (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate. U.S. Department of Agriculture Circ. No. 939
Peng GX, Tan ZY, Wang ET, Reinhold-Hurek B, Chen WF, Chen WX (2002) Identification of isolates from soybean nodules in Xinjiang Region as Sinorhizobium xinjiangense and genetic differentiation of S. xinjiangense from Sinorhizobium fredii. Int J Sys Evol Microbiol 52:457–462
Risal CP, Yokoyama T, Ohkama-Ohtsu N, Djedidi S, Sekimoto H (2010) Genetic diversity of native soybean bradyrhizobia from different topographical regions along the southern slopes of the Himalayan Mountains in Nepal. Syst Appl Microbiol 33:416–425
Saeki Y, Kaneko A, Hara T, Suzuki K, Yamakawa T, Nguyen MT, Nagatomo Y, Akao S (2005) Phylogenetic analysis of soybean-nodulating rhizobia isolated from alkaline soils in Vietnam. Soil Sci Plant Nutr 51:1043–1052
Saeki Y, Naoto A, Tsukamoto S, Yamakawa T, Nagatomo Y, Akao S (2006) Diversity and geographical distribution of indigenous soybean-nodulating bradyrhizobia in Japan. Soil Sci Plant Nutr 52:418–426
Smartt J, Hymowitz T (1985) Domestication and evolution of grain legumes. In: Summerfield RJ, Roberts EH (eds) Grain legume crops. Collins, London, pp 32–72
Someya T, Wang X, Gong C, Koshida K, Hakagawa H, Tanaka C, Ishibashi M, Yokobori K, Inoue K (2003) Rapid and specific detection and enumeration of microorganisms in soil and manure. Soil Microorg 57:115–123
Suzuki K, Oguro H, Yamakawa T, Yamamoto A, Akao S, Saeki Y (2008) Diversity and distribution of indigenous soybean-nodulating rhizobia in the Okinawa islands, Japan. Soil Sci Plant Nutr 54:237–246
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
van Berkum P, Fuhrmann JJ (2000) Evolutionary relationships among the soybean bradyrhizobia reconstructed from 16S rRNA gene and internally transcribed spacer region sequence divergence. Int J Syst Evol Microbiol 50:2165–2172
Vincent JM (1970) A manual for the practical study of root nodule bacteria. IBP Handbook No. 15. Blackwell, Oxford
Vinuesa P, Leon-Barrios M, Silva C, Willema A, Jarabo-Lorenzo A, Perez-Galdona R, Warner D, Martinez-Romero E (2005) Bradyrhizobium canariense sp. nov., an acid-tolerant endosymbiont that nodulates endemic genistoid legumes (Papilionoideae:Genisteae) growing in the Canary Islands, along with B. japonicum bv. Genistearum, Bradyrhizobium genospecies α and Bradyrhizobium genospecies β. Int J Syst Evol Microbiol 55:569–575
Vinuesa P, Rojas-Jimenez K, Contreas-Moreira B, Mahna SK, Prasad BN, Moe H, Selvaraju SB, Theirfelder H, Werner D (2008) Multilocus sequence analysis for assessment of the biogeography and evolutionary genetics of four Bradyrhizobium species that nodulate soybeans in the Asiatic Continent. Appl Environ Microbiol 74:6987–6996
Walkley A, Black IA (1934) An examination of Degtjareff method for determining organic carbon in soils: effect of variations in digestion conditions and of inorganic soil constituents. Soil Sci 63:251–263
Wasike VW, Lesueur D, Wachira FN, Mungai NW, Mumera LM, Sanginga N, Mburu HN, Mugadi D, Wango P, Vanlauwe B (2009) Genetic diversity of indigenous Bradyrhizobium nodulating promiscuous soybean [Glycine max (L) Merr.] varieties in Kenya: impact of phosphorus and lime fertilization in two contrasting sites. Plant Soil 322:151–163
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Xu LM, Ge C, Cui Z, Li J, Fan H (1995) Bradyrhizobium liaoningense sp. nov., isolated from the root nodules of soybeans. Int J Syst Bacteriol 45:706–711
Yang JK, Zhou JC (2008) Diversity, phylogeny and host specificity of soybean and peanut bradyrhizobia. Biol Fertil Soils 44:843–851
Yao ZY, Kan FL, Wang ET, Wei GH, Chen WX (2002) Characterization of rhizobia that nodulate legume species of the genus Lespedeza and description of Bradyrhizobium yuanmingense sp. nov. Int J Syst Evol Microbiol 52:2219–2230
Young JPW, Haukka KE (1996) Diversity and phylogeny of rhizobia. New Phytol 133:87–94
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Katharina Pawlowski.
Rights and permissions
About this article
Cite this article
Adhikari, D., Kaneto, M., Itoh, K. et al. Genetic diversity of soybean-nodulating rhizobia in Nepal in relation to climate and soil properties. Plant Soil 357, 131–145 (2012). https://doi.org/10.1007/s11104-012-1134-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1134-6