Abstract
Key message
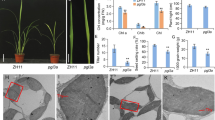
OsSIRP4 is an E3 ligase that acts as a negative regulator in the plant response to salt stress via the 26S proteasomal system regulation of substrate proteins, OsPEX11-1, which it provides important information for adaptation and regulation in rice.
Abstract
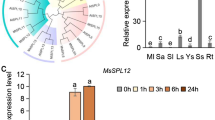
Plants are sessile organisms that can be exposed to environmental stress. Plants alter their cellular processes to survive under potentially unfavorable conditions. Protein ubiquitination is an important post-translational modification that has a crucial role in various cellular signaling processes in abiotic stress response. In this study, we characterized Oryza sativa salt-induced RING finger protein 4, OsSIRP4, a membrane and cytosol-localized RING E3 ligase in rice. OsSIRP4 transcripts were highly induced under salt stress in rice. We found that OsSIRP4 possesses E3 ligase activity; however, no E3 ligase activity was observed with a single amino acid substitution (OsSIRP4C269A). The results of the yeast two hybrid system, in vitro pull-down assay, BiFC analysis, in vitro ubiquitination assay, and in vitro degradation assay indicate that OsSIRP4 regulates degradation of a substrate protein, OsPEX11-1 (Oryza sativa peroxisomal biogenesis factor 11-1) via the 26S proteasomal system. Phenotypic analysis of OsSIRP4-overexpressing plants demonstrated hypersensitivity to salt response compared to that of the wild type and mutated OsSIRP4C269A plants. In addition, OsSIRP4-overexpressing plants exhibited significant low enzyme activities of superoxide dismutase, catalase, and peroxidase, and accumulation of proline and soluble sugar, but a high level of H2O2. Furthermore, qRT data on transgenic plants suggest that OsSIRP4 acted as a negative regulator of salt response by diminishing the expression of genes related to Na+/K+ homeostasis (AtSOS1, AtAKT1, AtNHX1, and AtHKT1;1) in transgenic plants under salt stress. These results suggest that OsSIRP4 plays a negative regulatory role in response to salt stress by modulating the target protein levels.







Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Anonymous (2008) FAO Land and Plant Nutrition Management Service. https://www.fao.org/ag/agl/agll/spush
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress and signal transduction. Annu Rev PlantBiol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Apse MP, Sottosanto JB, Blumwald E (2003) Vacuolar cation/H+ exchange, ion homeostasis, and leaf development are altered in a T-DNA insertional mutant of AtNHX1, the arabidopsis vacuolar Na+/H+ antiporter. Plant J 36(2):229–239
Aung K, Hu J (2011) The arabidopsis tail-anchored protein peroxisomal and mitochondrial division factor1 is involved in the morphogenesis and proliferation of peroxisomes and mitochondria. Plant Cell 23:4446–4461. https://doi.org/10.1105/tpc.111.090142
Bates LS, Waldren RP, Teare ID (2013) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assay and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Berggard T, Linse S, James P (2007) Methods for the detection and analysis of protein-protein interactions. Proteomics 7:2833–2842
Burkhart SE, Lingard MJ, Bartel B (2013) Genetic dissection of peroxisome associated matrix protein degradation in arabidopsis thaliana. Genetics 193:125–141. https://doi.org/10.1534/genetics.112.146100
Callis J (2014) The ubiquitination machinery of the ubiquitin system. Arabidopsis Book 12:e0174. https://doi.org/10.1199/tab.0174
Cassin-Ross G, Hu J (2014) Systematic phenotypic screen of arabidopsis peroxisomal mutants identifies proteins involved in b-oxidation. Plant Physiol 166:1546–1559. https://doi.org/10.1104/pp.114.250183
Chance B, Maehly AC (1955) Assay of catalase and peroxidase. Methods enzymol 2:764–775
Chang L, Zhang Z, Yang J, McLaughlin SH, Barford D (2014) Molecular architecture and mechanism of the anaphase-promoting complex. Nature 513:388–393
Chapagain S, Park YC, Kim JH, Jang CS (2018) Oryza sativa salt-induced RING E3 ligase 2 (OsSIRP2) acts as a positive regulator of transketolase in plant response to salinity and osmotic stress. Planta 247(4):925–939. https://doi.org/10.1007/s00425-017-2838-x
Ci DW, Jiang D, Dai TB, Jing Q, Cao WX (2009) Effects of cadmium on plant growth and physiological traits in contrast wheat recombinant inbred lines differing in cadmium tolerance. Chemosphere 77:1620–1625
Couée I, Sulmon C, Gouesbet G, El Amrani A (2006) Involvement of soluble sugars in reactive oxygen species balance and responses to oxidative stress in plants. J Exp Bot 57(3):449–459
Cui LH, Min HJ, Byun MY, Oh HG, Kim WT (2018) OsDIRP1, a putative RING E3 ligase, plays an opposite role in drought and cold stress responses as a negative and positive factor, respectively, in rice (Oryza sativa L.). Front Plant Sci 9:1797. https://doi.org/10.3389/fpls.2018.01797
Cui P, Liu H, Islam F, Li L, Farooq MA, Ruan S, Zhou W (2016) OsPEX11, a peroxisomal biogenesis factor 11, contributes to salt stress tolerance in Oryza sativa. Front Plant Sci 15(7):1357. https://doi.org/10.3389/fpls.2016.01357
Del Río LA, Corpas FJ, Sandalio LM, Palma JM, Barroso JB (2003) Plant peroxisomes, reactive oxygen metabolism and nitric oxide. IUBMB Life 55:71–81
Del Río LA, Sandalio LM, Corpas FJ, Palma JM, Barroso JB (2006) Barroso, reactive oxygen species and reactive nitrogen species in peroxisomes. Production, scavenging, and role in cell signaling. Plant Physiol 141(2):330–335
Deshaies RJ, Joazeiro CA (2009) RING domain E3 ubiquitin ligases. Annu Rev Biochem 78:399–434. https://doi.org/10.1146/annurev.biochem.78.101807.093809
Dreher K, Callis J (2007) Ubiquitin, hormones and biotic stress in plants. Ann Bot 99:787–822
Fang H, Meng Q, Xu J, Tang H, Tang S, Zhang H, Huang J (2015) Knock-down of stress inducible OsSRFP1 encoding an E3 ubiquitin ligase with transcriptional activation activity confers abiotic stress tolerance through enhancing antioxidant protection in rice. Plant Mol Biol 87(4–5):441–458. https://doi.org/10.1007/s11103-015-0294-1
Pitman MG, Läuchli A (2002) Global impact of salinity and agricultural ecosystems. In: Läuchli A, Lüttge U (eds) Salinity: environment - plants - molecules. Springer, Dordrecht, pp 3–20
Foyer CH, Harbinson J (1994) Oxygen metabolism and the regulation of photosynthetic electron transport. In: Foyer CH, Mullineaux P (eds) Causes of photooxidative stresses and amelioration of defense systems in plants. CRC Press, Boca Raton, Fla, USA, pp 1–42
Foyer CH, Noctor G (2005) Oxidant and antioxidant signaling in plants: are-evaluation of the concept of oxidative stress a physiological context. Plant Cell Environ 28:1056–1071. https://doi.org/10.1111/j.1365-3040.2005.01327.x
García-Cano E, Zaltsman A, Citovsky V (2014) Assaying proteasomal degradation in a cell-free system in plants. J Vis Exp 85:e51293. https://doi.org/10.3791/51293
Gierth M, Mäser P, Schroeder JI (2005) The potassium transporter AtHAK5 functions in K+ deprivation-induced high-affinity K+ uptake and AKT1 K+ channel contribution to K+ uptake kinetics in arabidopsis roots. Plant Physiol 137:1105–1114
Habib SH, Kausar H, Saud HM (2016) Plant growth- promoting rhizobacteria enhance salinity stress tolerance In okra through ROS-scavenging enzymes. Biomed Res Int 2016:6284547. https://doi.org/10.1155/2016/6284547
Hakes L, Pinney JW, Robertson DL, Lovell SC (2008) Protein-protein interaction networks and biology–what’s the connection? Nat Biotechnol 26:69–72
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7(11):1456–1466. https://doi.org/10.4161/psb.21949
Hirsch RE, Lewis BD, Spalding EP, Sussman MR (1998) A role for the AKT1 potassium channel in plant nutrition. Science 280:918–921
Horie T, Kaneko T, Sugimoto G, Sasano S, Panda SK, Shibasaka M, Katsuhara M (2011) Mechanisms of water transport mediated by PIP aquaporins and their regulation via phosphorylation events under salinity stress in barley roots. Plant Cell Physiol 52(4):663–675. https://doi.org/10.1093/pcp/pcr027
Horie T, Karahara I, Katsuhara M (2012) Salinity tolerance mechanisms in glycophytes: an overview with the central focus on rice plants. Rice 5:11. https://doi.org/10.1186/1939-8433-5-11
Hossain MS, Dietz K-J (2016) Tuning of redox regulatory mechanisms, reactive oxygen species and redox homeostasis under salinity stress. Front Plant Sci 7:548. https://doi.org/10.3389/fpls.2016.00548
Hwang SG, Kim JJ, Lim SD, Park YC, Moon JC, Jang CS (2016) Molecular dissection of Oryza sativa salt-induced RING finger protein 1 (OsSIRP1): possible involvement in the sensitivity response to salinity stress. Physiol Plant 158(2):168–179. https://doi.org/10.1111/ppl.12459
Islam F, Ali B, Wang J, Farooq MA, Gill RA, Ali S, Wang D, Zhou W (2016) Combined herbicide and saline stress differentially modulates hormonal regulation and antioxidant defense system in Oryza Sativa cultivars. Plant Physiol Biochem 107:82–95. https://doi.org/10.1016/j.plaphy.2016.05.027
Islam F, Yasmeen T, Ali S, Ali B, Farooq MA, Gill RA (2015) Priming- induced antioxidative responses into wheat cultivars under saline stress. Acta Physiol Plant 37:1–12. https://doi.org/10.1007/s11738-015-1897-5
Jaleel CA, Gopi R, Manivannan P, Gomathinayagam M, Riadh K, Zhao JI, Chang-Xing Z, Hong-Bo S, Panneerselvam R (2009) Antioxidant defense responses: physiological plasticity in higher plants under abiotic constraints. Acta Physiolog Plant 31(3):427–436
Jamil A, Riaz S, Ashraf M, Foolad MR (2011) Gene expression profiling of plants under salt stress. Crit Rev Plant Sci 30(5):435–458
Kao YT, Gonzalez KL, Bartel B (2018) Peroxisome function, biogenesis, and dynamics in plants. Plant Physiol 176(1):162–177. https://doi.org/10.1104/pp.17.01050
Kim JH, Lim SD, Jang CS (2020) Oryza sativa drought-, heat-, and salt-induced RING finger protein 1 (OsDHSRP1) negatively regulates abiotic stress-responsive gene expression. Plant Mol Biol 103(3):235–252. https://doi.org/10.1007/s11103-020-00989-x
Lee DH, Choi HW, Hwang BK (2011) The pepper E3 ubiquitin ligase RING1 gene, CaRING1, is required for cell death and the salicylic acid-dependent defense response. Plant Physiol 156:2011–2025
Li J, Han Y, Zhao Q, Li C, Xie Q, Chong K, Xu Y (2013) The E3 ligase AtRDUF1 positively regulates salt stress responses in arabidopsis thaliana. PLoS ONE 8(8):e71078. https://doi.org/10.1371/journal.pone.0071078
Liang W, Ma X, Wan P, Liu L (2018) Plant salt-tolerance mechanism: a review. Biochem Biophys Res Commun 495(1):286–291. https://doi.org/10.1016/j.bbrc.2017.11.043
Lim SD, Hwang JG, Jung CG, Hwang SG, Moon JC, Jang CS (2013) Comprehensive analysis of the rice RING E3 ligase family reveals their functional diversity in response to abiotic stress. DNA Res 20(3):299–314
Liu Q, Ning Y, Zhang Y, Yu N, Zhao C, Zhan X, Wu W, Chen D, Wei X, Wang GL, Cheng S, Cao L (2017) OsCUL3a negatively regulates cell death and immunity by degrading OsNPR1 in rice. Plant Cell 29:345–359
Lu S, Wang J, Chitsaz F, Derbyshire MK, Geer RC, Gonzales NR, Gwadz M, Hurwitz DI, Marchler GH, Song JS, Thanki N, Yamashita RA, Yang M, Zhang D, Zheng C, Lanczycki CJ, Marchler BA (2020) CDD/SPARCLE: the conserved domain database in 2020. Nucleic Acids Res 8(48):265–268. https://doi.org/10.1093/nar/gkz991
McClellan AJ, Laugesen SH, Ellgaard L (2019) Cellular functions and molecular mechanisms of non-lysine ubiquitination. Open Biol 9(9):190147. https://doi.org/10.1098/rsob.190147
Metzger MB, Pruneda JN, Klevit RE, Weissman AM (1843) RING-type E3 ligases: master manipulators of E2 ubiquitin-conjugating enzymes and ubiquitination. Biochim Biophys Acta 1:47–60. https://doi.org/10.1016/j.bbamcr.2013.05.026
Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 33(4):453–467. https://doi.org/10.1111/j.1365-3040.2009.02041.x
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Møller IM, Jensen PE, Hansson A (2007) Oxidative modifications to cellular components in plants. Annu Rev PlantBiol 58:459–481. https://doi.org/10.1146/annurev.arplant.58.032806.103946
Moon J, Parry G, Estelle M (2004) The ubiquitin-proteasome pathway and plant development. Plant Cell 16:3181–3195
Munns R, Gilliham M (2015) Salinity tolerance of crops-what is the cost? New Phytol 208:668–673. https://doi.org/10.1111/nph.13519
Munns R, James RA, Läuchli A (2006) Approaches to increasing the salt tolerance of wheat and other cereals. J Exp Bot 57(5):1025–1043
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
Munns R, andTester, M. (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
Nayidu NK, Wang L, Xie W, Zhang C, Fan C, Lian X, Zhang Q, Xiong L (2008) Comprehensive sequence and expression profile analysis of PEX11 gene family in rice. Gene 412(1–2):59–70. https://doi.org/10.1016/j.gene.2008.01.006
Nelson BK, Cai X, Nebenführ A (2007) A multicolored set of in vivo organelle markers for co-localization studies in Arabidopsis and other plants. Plant J 51(6):1126–1136
Nito K, Kamigaki A, Kondo M, Hayashi M, Nishimura M (2007) Functional classification of arabidopsis peroxisome biogenesis factors proposed from analyses of knockdown mutants. Plant Cell Physiol 48(6):763–774
Noreen Z, Ashraf M, Akram N (2010) Salt-induced regulation of some key antioxidant enzymes and physio-biochemical phenomena in five diverse cultivars of turnip (Brassicarapa L.). J Agro Crop Sci 196:273–285
Park YC, Chapagain S, Jang CS (2018) A negative regulator in response to salinity in rice: Oryza sativa salt-, ABA- and drought-induced RING finger protein 1 (OsSADR1). Plant Cell Physiol 59(3):575–589. https://doi.org/10.1093/pcp/pcy009
Pazhouhandeh M, Molinier J, Berr A, Genschik P (2011) MSI4/FVE interacts with CUL4-DDB1 and a PRC2-like complex to control epigenetic regulation of flowering time in arabidopsis. Proc Natl Acad Sci USA 108:3430–3435
Qi S, Lin Q, Zhu H, Gao F, Zhang W, Hua X (2016) The RING finger E3 ligase SpRing is a positive regulator of salt stress signaling in salt-tolerant wild tomato species. Plant Cell Physiol 57(3):528–539. https://doi.org/10.1093/pcp/pcw006
Rahman A, Nahar K, Mahmud JA, Hasanuzzaman M, Hossain S, Fujita M (2017) Salt stress tolerance in rice: emerging role of exogenous phytoprotectants. In: Li J (ed) Advances in international rice research. InTech, Rijeka, pp 139–174
Rakshit A, Singh H (2018) Advances in seed priming. Springer, Singapore. https://doi.org/10.1007/978-981-13-0032-5
Rathinasabapathi B (2000) Metabolic engineering for stress tolerance: installing osmoprotectant synthesis pathways. Ann Bot 86(4):709–716
Rejeb KB, Abdelly C, Savouré A (2014) How reactive oxygen species and proline face stress together. Plant Physiol Biochem 80:278–284. https://doi.org/10.1016/j.plaphy.2014.04.007
Roy SJ, Negrao S, Tester M (2014) Salt resistant crop plants. Curr Opin Biotechnol 26:115–124. https://doi.org/10.1016/j.copbio.2013.12.004
Rubio F, Nieves-Cordones M, Alemán F, Martínez V (2008) Relative contribution of AtHAK5 and AtAKT1 to K+ uptake in the high-affinity range of concentrations. Physiol Plant 134(4):598–608. https://doi.org/10.1111/j.1399-3054.2008.01168.x
Sadanandom A, Bailey M, Ewan R, Lee J, Nelis S (2012) The ubiquitin-proteasome system: central modifier of plant signaling. New Phytol 196:13–28
Saunders DN, Iconomou M (2016) Systematic approaches to identify E3 ligase substrates. Biochem J 473(22):4083–4101
Shrivastava P, Kumar R (2015) Soil salinity: a serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J Biol Sci 22:123–131
Singh T, Pun KB, Saikia K, Satapathy BS, Bhagat K, Das A, Lal B (2015) In book: Integrated soil and water resource management for livelihood and environmental security, chapter: abiotic stress management in rice. 219–258
Sottosanto JB, Saranga Y, Blumwald E (2007) Impact of AtNHX1, a vacuolar Na+/H+ antiporter, upon gene expression during short- and long-term salt stress in Arabidopsis thaliana. BMC Plant Biol 7:18. https://doi.org/10.1186/1471-2229-7-18
Wang Q, Guan C, Wang S (2014) Coordination of AtHKT1;1 and AtSOS1 facilitates Na+ and K+ homeostasis in Arabidopsis thaliana under salt stress. J Plant Biol 57:282–290. https://doi.org/10.1007/s12374-014-0222-y
Wang R, Ning Y, Shi X, He F, Zhang C, Fan J, Jiang N, Zhang Y, Zhang T, Hu Y, Bellizzi M, Wang GL (2016) Immunity to rice blast disease by suppression of effector-triggered necrosis. Curr Biol 26:2399–2411
Zeng D, Hou P, Xiao F, Liu Y (2014) Overexpressing a novel RING-H2 finger protein gene, OsRHP1, enhances drought and salt tolerance in rice (Oryza sativa L.). J Plant Biol 57:357–365. https://doi.org/10.1007/s12374-013-0481-z
Zhang Y, Su J, Duan S, Ao Y, Dai J, Liu J, Wang P, Li Y, Liu B, Feng D, Wang J, Wang H (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7(1):30. https://doi.org/10.1186/1746-4811-7-30
Zheng N, Schulman BA, Song L, Miller JJ, Jeffrey PD, Wang P, Chu C, Koepp DM, Elledge SJ, Pagano M, Conaway RC, Conaway JW, Harper JW, Pavletich NP (2002) Structure of the Cul1-Rbx1-Skp1-F boxSkp2 SCF ubiquitin ligase complex. Nature 416:703–709
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (Grant Number NRF-2019R1A2C1009840).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, J.H., Jang, C.S. E3 ligase, the Oryza sativa salt-induced RING finger protein 4 (OsSIRP4), negatively regulates salt stress responses via degradation of the OsPEX11-1 protein. Plant Mol Biol 105, 231–245 (2021). https://doi.org/10.1007/s11103-020-01084-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-01084-x