Abstract
Key message
The genetic linkage map for green ash (Fraxinus pennsylvanica) contains 1201 DNA markers in 23 linkage groups spanning 2008.87cM. The green ash map shows stronger synteny with coffee than tomato.
Abstract
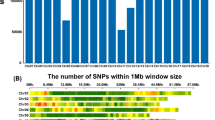
Green ash (Fraxinus pennsylvanica) is an outcrossing, diploid (2n = 46) hardwood tree species, native to North America. Native ash species in North America are being threatened by the rapid spread of the emerald ash borer (EAB, Agrilus planipennis), an invasive pest from Asia. Green ash, the most widely distributed ash species, is severely affected by EAB infestation, yet few genomic resources for genetic studies and improvement of green ash are available. In this study, a total of 5712 high quality single nucleotide polymorphisms (SNPs) were discovered using a minimum allele frequency of 1% across the entire genome through genotyping-by-sequencing. We also screened hundreds of genomic- and EST-based microsatellite markers (SSRs) from previous de novo assemblies (Staton et al., PLoS ONE 10:e0145031, 2015; Lane et al., BMC Genom 17:702, 2016). A first genetic linkage map of green ash was constructed from 90 individuals in a full-sib family, combining 2719 SNP and 84 SSR segregating markers among the parental maps. The consensus SNP and SSR map contains a total of 1201 markers in 23 linkage groups spanning 2008.87 cM, at an average inter-marker distance of 1.67 cM with a minimum logarithm of odds of 6 and maximum recombination fraction of 0.40. Comparisons of the organization the green ash map with the genomes of asterid species coffee and tomato, and genomes of the rosid species poplar and peach, showed areas of conserved gene order, with overall synteny strongest with coffee.



Similar content being viewed by others
References
Arús P, Verde I, Sosinski B, Zhebentyayeva T, Abbott AG (2012) The peach genome. Tree Genet Genomes 8:531–547
Bai X, Rivera-Vega L, Mamidala P, Bonello P, Herms DA, Mittapalli O (2011) Transcriptomic signatures of ash (Fraxinus spp.) phloem. PLoS ONE 6:e16368
Barreneche T, Bodenes C, Lexer C, Trontin J-F, Fluch S, Streiff R, Plomion C, Roussel G, Steinkellner H, Burg K, Favre J-M, Glössl J, Kremer A (1998) A genetic linkage map of Quercus robur L. (pedunculate oak) based on RAPD, SCAR, microsatellite, minisatellite, isozyme and 5S rDNA markers. Theor Appl Genet 97:1090–1103
Bowers JE, Chapman BA, Rong J, Paterson AH (2003) Unravelling angiosperm genome evolution by phylogenetic analysis of chromosomal duplication events. Nature 422:433
Bradshaw HD Jr, Stettler RF (1994) Molecular genetics of growth and development in Populus. II. Segregation distortion due to genetic load. Theor Appl Genet 89:551–558
Burns RM, Honkala BH (1990) Silvics of North America, vol 1. Conifers. Agriculture Handbook, Washington, p 877
Cantino PD, Doyle JA, Graham SW, Judd WS, Olmstead RG, Soltis DE, Soltis PS, Donoghue MJ (2007) Towards a phylogenetic nomenclature of Tracheophyta. Taxon 56:1E–44E
Celik I, Bodur S, Frary A, Doganlar S (2016) Genome-wide SNP discovery and genetic linkage map construction in sunflower (Helianthus annuus L.) using a genotyping by sequencing (GBS) approach. Mol Breed 36:133
Cervera M-T, Storme V, Ivens B, Gusmao J, Liu BH, Hostyn V, Slycken JV, Montagu MV, Boerjan W (2001) Dense genetic linkage maps of three Populus species (Populus deltoides. P. nigra and P. trichocarpa) based on AFLP and microsatellite markers. Genetics 158:787–809
Clarke JD (2009) Cetyltrimethyl ammonium bromide (CTAB) DNA miniprep for plant dna isolation. Cold Spring Harbor Protocols, 2009: pdb.prot5177
Dai B, Guo H, Huang C, Ahmed MM, Lin Z (2016) Identification and characterization of segregation distortion loci on cotton chromosome 18. Front Plant Sci 7:2037
Davey JW, Blaxter ML (2010) RADSeq: next-generation population genetics. Brief Funct Genom 9:416–423
Denoeud F, Carretero-Paulet L, Dereeper A, Droc G, Guyot R, Pietrella M, Zheng C et al (2014) The coffee genome provides insight into the convergent evolution of caffeine biosynthesis. Science 345:1181–1184
Drouaud J, Mercier R, Chelysheva L, Bérard A, Falque M, Martin O, Zanni V, Brunel D, Mézard C (2007) Sex-specific crossover distributions and variations in interference level along Arabidopsis thaliana chromosome 4. PLoS Genet 3(6):e106
Elshire RJ, Glaubitz JC, Sun Q, Poland JA, Kawamoto K, Buckler ES, Mitchell SE (2011) A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6:e19379
Freeman JS, Potts BM, Shepherd M, Vaillancourt RE (2006) Parental and consensus linkage maps of Eucalyptus globulus using AFLP and microsatellite markers. Silvae Genet 55:202–217
Gabriel S, Ziaugra L, Tabbaa D (2001) SNP Genotyping Using the Sequenom MassARRAY iPLEX Platform. In: Haines JL, Korf BR, Morton CC (eds) Current protocols in human genetics. Wiley, New York
Gaur R, Jeena G, Shah N, Gupta S, Pradhan S, Tyagi AK, Jain M, Chattopadhyay D, Bhatia S (2015) High density linkage mapping of genomic and transcriptomic SNPs for synteny analysis and anchoring the genome sequence of chickpea. Sci Rep 5:13387
Giraut L, Falque M, Drouaud J, Pereira L, Martin OC, Mézard C (2011) Genome-wide crossover distribution in Arabidopsis thaliana meiosis reveals sex-specific patterns along chromosomes. PLoS Genet 7:e1002354
Glaubitz JC, Casstevens TM, Lu F, Harriman J, Elshire RJ, Sun Q, Buckler ES (2014) TASSEL-GBS: a high capacity genotyping by sequencing analysis pipeline. PLoS ONE 9:e90346
Henderson IR (2012) Control of meiotic recombination frequency in plant genomes. Curr Opin Plant Biol 15:556–561
Herms DA, McCullough DG (2014) Emerald ash borer invasion of North America: history, biology, ecology, impacts, and management. Annu Rev Entomol 59:13–30
Jaillon OJ. Aury M, Noel B, Policriti A, Clepet C, Casagrande A, Choisne N, Aubourg S et al (2007) The grapevine genome sequence suggests ancestral hexaploidization in major angiosperm phyla. Nature 449:463–467
Jones OR, Wang J (2010) COLONY: a program for parentage and sibship inference from multilocus genotype data. Mol Ecol Res 10:551–555
Knight KS, Herms D, Plumb R, Sawyer E, Spalink D, Pisarczyk E, Wiggin B, Kappler R, Ziegler E, Menard K (2012) Dynamics of surviving ash (Fraxinus spp.) populations in areas long infested by emerald ash borer (Agrilus planipennis). In: Proceedings of the 4th international workshop on genetics host–parasite interactions in forestry, USDA Forest Service Pacific Southwest Research Station, PSW-GTR-240, 143–152
Koch JL, Carey DW, Mason ME, Poland TM, Knight KS (2015) Intraspecific variation in Fraxinus pennsylvanica responses to emerald ash borer (Agrilus planipennis). New For 46:995–1011
Koressaar T, Remm M (2007) Enhancements and modifications of primer design program Primer3. Bioinformatics 23:1289–1291
Kovacs KF, Haight RG, McCullough DG, Mercader RJ, Siegert NW, Liebhold AM (2010) Cost of potential emerald ash borer damage in U.S. communities, 2009–2019. Ecol Econ 69:569–578
Krzywinski M, Schein J, Birol I, Connors J, Gascoyne R, Horsman D, Jones SJ, Marra MA (2009) Circos: an information aesthetic for comparative genomics. Genome Res 19:1639–1645
Lane T, Best T, Zembower N, Davitt J, Henry N, Xu Y, Koch J, Liang H, McGraw J, Schuster S, Shim D, Coggeshall MV, Carlson JE, Staton ME (2016) The green ash transcriptome and identification of genes responding to abiotic and biotic stresses. BMC Genom 17:702
Li KA, Zhou M, Wenzl P, Huttner E, Mendham N, McIntyre L, Vaillancourt RE (2010) Construction of a high-density composite map and comparative mapping of segregation distortion regions in barley. Mol Genet Genom 284:319–331
Liebhard R, Koller B, Gianfranceschi L, Gessler C (2003) Creating a saturated reference map for the apple (Malus x domestica Borkh.) genome. Theor Appl Genet 106:1497–1508
Lu H, Romero-Severson J, Bernardo R (2002) Chromosomal regions associated with segregation distortion in maize. Theor Appl Genet 105:622–628
Lukens LN, Quijada PA, Udall J, Pires JC, Schranz ME, Osborn TC (2004) Genome redundancy and plasticity within ancient and recent Brassica crop species. Biol J Lin Soc 82:665–674
Luo M-C, You FM, Li P, Wang J-R, Zhu T, Dandekar AM, Leslie CA, Aradhya M, McGuire PA, Dvorak J (2015) Synteny analysis in Rosids with a walnut physical map reveals slow genome evolution in long-lived woody perennials. BMC Genom 16:707
Lysak MA, Koch MA, Pecinka A, Schubert I (2005) Chromosome triplication found across the tribe Brassiceae. Genome Res 15:516–525
Magallón S, Castillo A (2009) Angiosperm diversification through time. Am J Bot 96:349–365
Ming R, Hou S, Feng Y, Yu Q, Dionne-Laporte A, Saw JH, Senin P, Wang W, Ly BV, Lewis KL, Salzberg SL et al (2008) The draft genome of the transgenic tropical fruit tree papaya (Carica papaya Linnaeus)’. Nature 452:991–996
Moumouni KH, Kountche BA, Jean M, Hash CT, Vigouroux Y, Haussmann BIG, Belzile F (2015) Construction of a genetic map for pearl millet, Pennisetum glaucum (L.) R. Br., using a genotyping-by-sequencing (GBS) approach. Mol Breed 35:5
Neves LG, Davis JM, Barbazuk WB, Kirst M (2014) A high-density gene map of loblolly pine (Pinus taeda L.) based on exome sequence capture genotyping. G3: Genes|Genomes|Genetics 4:29–37
Pootakham W, Jomchai N, Ruang-areerate P, Shearman JR, Sonthirod C, Sangsrakru D, Tragoonrung S, Tangphatsornruang S (2015) Genome-wide SNP discovery and identification of QTL associated with agronomic traits in oil palm using genotyping-by-sequencing (GBS). Genomics 105:288–295
Raman H, Dalton-Morgan J, Diffey S, Raman R, Alamery S, Edwards D, Batley J (2014) SNP markers-based map construction and genome-wide linkage analysis in Brassica napus. Plant Biotechnol J 12:851–860
Ren R, Wang H, Guo C, Zhang N, Zeng L, Chen Y, Ma H, Qi J (2018) Widespread whole genome duplications contribute to genome complexity and species diversity in angiosperms. Mol Plant 11:414–428
Sandler L, Novitski E (1957) Meiotic drive as an evolutionary force. Am Nat 91:105–110
Schranz ME, Mitchell-Olds T (2006) Independent ancient polyploidy events in the sister families Brassicaceae and Cleomaceae. Plant Cell 18:1152–1165
Schuelke M (2000) An economic method for the fluorescent labeling of PCR fragments. Nat Biotechnol 18:233–234
Sollars ESA, Harper AL, Kelly LJ, Sambles CM, Ramirez-Gonzalez RH, Swarbreck D, Kaithakottil G, Cooper ED, Uauy C, Havlickova L, Worswick G et al (2017) Genome sequence and genetic diversity of European ash trees. Nature 541:212–216
Soltis DE, Albert VA, Leebens-Mack J, Bell CD, Paterson AH, Zheng C, Sankoff D, Depamphilis CW, Wall PK, Soltis PS (2009) Polyploidy and angiosperm diversification. Am J Bot 96:336–348
Staton M, Best T, Khodwekar S, Owusu S, Xu T, Xu Y, Jennings T, Cronn R, Arumuganathan AK, Coggeshall M, Gailing O, Liang H, Romero-Severson J, Schlarbaum S, Carlson JE (2015) Preliminary genomic characterization of ten hardwood tree species from multiplexed low coverage whole genome sequencing. PLoS ONE 10:e0145031
Subbaiyan GK, Waters DLE, Katiyar SK, Sadananda AR, Vaddadi S, Henry RJ (2012) Genome-wide DNA polymorphisms in elite indica rice inbreds discovered by whole-genome sequencing. Plant Biotechnol J 10:623–634
Taylor DR, Ingvarsson PK (2003) Common features of segregation distortion in plants and animals. Genetica 117:27–35
The Tomato Genome, Consortium (2012) The tomato genome sequence provides insights into fleshy fruit evolution. Nature 485:635
Tian M, Li Y, Jing J, Mu C, Du H, Dou J, Mao J, Li X, Jiao W, Wang Y, Hu X, Wang S, Wang R, Bao Z (2015) Construction of a high-density genetic map and quantitative trait locus mapping in the sea cucumber Apostichopus japonicus. Sci Rep 5:14852
Tuskan GA, Difazio S, Jansson S, Bohlmann J, Grigoriev I, Hellsten U, Putnam N, Ralph S, Rombauts S, Salamov A, Schein J, Sterck L, Aerts A, Bhalerao RR, Bhalerao RP,. Blaudez D, Boerjan W, Brun A, Brunner A, Busov V, Campbell M, Carlson J et al (2006) The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 313:1596–1604
Untergasser A, Cutcutache I, Koressaar T, Ye J, Faircloth BC, Remm M, Rozen SG (2012) Primer3—new capabilities and interfaces. Nucleic Acids Res 40:e115–e115
Van Ooijen JW (2006) JoinMap 4: Software for the calculation of genetic linkage maps in experimental populations. Kyazma BV, Wageningen
Van Tassell, CP, Smith TPL, Matukumalli LK, Taylor JF, Schnabel RD, Lawley CT, Haudenschild CD, Moore SS, Warren WC, Sonstegard TS (2008) SNP discovery and allele frequency estimation by deep sequencing of reduced representation libraries. Nat Methods 5:247
Voorrips RE (2002) MapChart: software for the graphical presentation of linkage maps and QTLs. J Hered 93:77–78
Wang K, Wang Z, Li F, Ye W, Wang J, Song G, Yue Z, Cong L, Shang H, Zhu S, Zou C, Li Q, Yuan Y, Lu C, Wei H, Gou C, Zheng Z, Yin Y, Zhang X, Liu K, Wang B, Song C, Shi N, Kohel RJ, Percy RG, Yu JZ, Zhu Y-X, Wang J, Yu S (2012) The draft genome of a diploid cotton Gossypium raimondii. Nat Genet 44:1098
Wikström N, Savolainen V, Chase MW (2001) Evolution of the angiosperms: calibrating the family tree. Proc R Soc Lond B 268:2211–2220
Wu Y, Close TJ, Lonardi S (2008) On the accurate construction of consensus genetic maps. Comput Syst Bioinform Conf 7:285–296
Zhou W, Tang Z, Hou J, Hu N, Yin T (2015) Genetic map construction and detection of genetic loci underlying segregation distortion in an intraspecific cross of Populus deltoides. PLoS ONE 10:e0126077
Acknowledgements
The authors would like to thank Wanyan Wang and Teodora Best for assistance with sample collection. We also thank Lianna Johnson and Byron Bredael for help with DNA extractions and screening SSR primers. Finally, we appreciate assistance from Nicole Zembower and Maureen Mailander on seedling planting and field maintenance. This work was supported by grants from NSF’s Plant Genome Research Program (IOS-1025974) to JEC and the USDA National Institute of Food and Agriculture Federal Appropriations under Project PEN04532 and Accession number 1000326.
Author information
Authors and Affiliations
Contributions
DW conducted all of the research for her dissertation and prepared the first draft of the manuscript. JK conducted the controlled pollinations, reared the seedlings, provided the seedlings for mapping, and provided comments and edits on the manuscript. MC provided nursery space and advice for rearing the seedlings, and provided comments and edits on the manuscript. JC obtained funding, supervised the research, and provided comments and edits on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exit.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, D., Koch, J., Coggeshall, M. et al. The first genetic linkage map for Fraxinus pennsylvanica and syntenic relationships with four related species. Plant Mol Biol 99, 251–264 (2019). https://doi.org/10.1007/s11103-018-0815-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-018-0815-9