ABSTRACT
Purpose
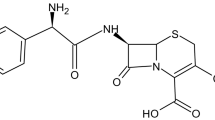
The goal of the study was to isolate and analyze a cetirizine degradation product, formed within a PEG-containing formulation and to elucidate the mechanism of oxidation of cetirizine.
Methods
Cetirizine, formulated in PEG-containing matrix, was subjected to forced degradation conditions in the pH range from 3 to 10, and the product was analyzed by HPLC and LC-MS/MS. Additionally, pure cetirizine was subjected to selective oxidization by hydrogen peroxide and sodium percarbonate. The reaction mixture was purified, and the isolated material was analyzed by 1H NMR.
Results
Oxidation process was investigated in order to model the degradation of cetirizine in PEG-containing formulation. Site of oxidation is proposed based on correlation of the results of forced degradation with ionization scheme of cetirizine. The finding was verified by spiking of cetirizine degradation sample with cetirizine N-oxide reference standard.
Conclusions
Degradation of cetirizine in polyethylene glycol arose from the reaction between the drug and the reactive peroxide intermediates such as peroxyl radicals formed through oxidation of PEG. Selective oxidation of cetirizine and isolation/characterization of the oxidation product allowed the identification of the oxidation product as cetirizine N-oxide. The mechanism of oxidation is proposed.








Similar content being viewed by others
REFERENCES
Jones JW, Francis JJ. Softgels: consumer perceptions and market impact relative to other oral dosage forms. Adv ther. 2000;17:5.
Makhija SN, Vavia PR. Stability indicating HPTLC method for the simultaneous determination of pseudoephedrine and cetirizine in pharmaceutical formulations. J Pharm Biomed Anal. 2001;25:663–7.
Benedetti MS, Plisnier M, Kaise J, Maier L, Baltes E, Arendt C et al. Absorption, distribution, metabolism and excretion of [14C]levocetirizine, the R enantiomer of cetirizine, in healthy volunteers. Eur J Clin Pharmacol. 2001;57:571–82.
V. Kumar, DS, Kalonia. Removal of Peroxides in Polyethylene Glycols by Vacuum Drying: Implications in the stability of Biotech and Parmaceutical Formulations. AAPS PharSciTech. 7(3) 2006; Article 62. DOI; 10 1208 /pt070362.
Hamburger R, Azaz E, Donbrow M. Autoxidation of polyoxyethylenic nonionic surfactants and of polyethylene glycols. Pharm Acta Helv. 1975;50(1–2):10–7.
Donbrow M, Azaz E, Pillersdorf A. Autoxidation of polysorbates. J Pharm Sci. 1978;67(12):1676–81.
Shimizu S, Watanabe N, Kuranishi H. US 5, 668281 (1997).
Pagliara A, Testa B, Carrupt PA, Jolliet P, Morin C, Morin D et al. Molecular properties and pharmacokinetic behaviour of cetirizine, a zwitterionic H1-receptor antagonist. J Med Chem. 1998;41:853–63.
Freed AL, Strohmeyer HE, Mahjour M, Sadineni V, Reid DL, Kingsmill CA. pH control of nucleophilic/electrophilic oxidation. Int J Pharm. 2008;357:180–8.
Tam KY, Quere L. Multiwavelenght spectrophotometric resolution of the micro- equilibria of cetirizine. Anal Sci. 2001;17:1203–8.
Salvador A, Sousa J, Pinto RE. Hydroperoxyl Superoxide and pH Gradients in the Mitochondrial Matrix: A Theoretical Assessment. Free Radic Biol Med. 2001;31(10):1208–15.
Nasri H, Dyakonov T, Toops D. Compatibility of shell ingredients with hydrophilic actives in gelatin-based dosage forms, presented at AAPS, Atlanta (2008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dyakonov, T., Muir, A., Nasri, H. et al. Isolation and Characterization of Cetirizine Degradation Product: Mechanism of Cetirizine Oxidation. Pharm Res 27, 1318–1324 (2010). https://doi.org/10.1007/s11095-010-0114-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-010-0114-x