No Heading
Purpose.
Experiments were performed to assess the brain distribution of [3H]-verapamil, including the influence of delivery route of inhibitor and substrate (nasal vs. systemic) on brain distribution. The anatomic location of P-glycoprotein (P-gp) at the nose-brain barrier also was investigated.
Methods.
Separate groups of mice were pretreated with rifampin or vehicle nasally or intravenously. [3H]-verapamil was administered either nasally or via in situ brain perfusion, and dose-response profiles were constructed for P-gp inhibition. Localization of P-gp in freshly obtained brain slices and olfactory tissue was evaluated by confocal microscopy.
Results.
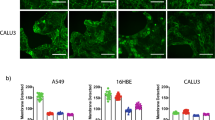
Rifampin inhibited the P-gp–mediated efflux of [3H]-verapamil, regardless of delivery route (Imax = 62 ± 6%). The ED50 for enhancement of [3H]-verapamil uptake by nasal rifampin was ∼400-fold lower than for intravenous rifampin (0.16 vs. 65 mg/kg, respectively). Microscopy showed that P-gp was located in endothelial cells that line the olfactory bulb and within the olfactory epithelium.
Conclusion.
Nasal delivery of rifampin enhanced brain uptake of [3H]-verapamil. The magnitude of transport inhibition was dependent on the dose and route of the inhibitor, the time after administration of the inhibitor, and the specific brain region examined. P-gp is localized to both the olfactory epithelium and the endothelial cells that surround the olfactory bulb.
Similar content being viewed by others
References
1. C. L. Graff and G. M. Pollack. Drug transport at the blood-brain barrier and the choroid plexus. Curr. Drug Metab. 5:95–108 (2004).
2. R. Polt, F. Porreca, L. Z. Szabo, E. J. Bilsky, P. Davis, T. J. Abbruscato, T. P. Davis, R. Horvath, H. I. Yamamura, and V. J. Hruby. Glycopeptide enkephalin analogues produce analgesia in mice: Evidence for penetration of the blood-brain barrier. Proc. Natl. Acad. Sci. USA 91:7114–7118 (1994).
3. C. L. Graff and G. M. Pollack. P-glycoprotein attenuates brain uptake of substrates after nasal instillation. Pharm. Res. 20:1225–1230 (2003).
4. I. Manzini and D. Schild. Multidrug resistance transporters in the olfactory receptor neurons of Xenopus laevis tadpoles. J. Physiol. 546:375–385 (2003).
5. K. K. Kandimalla and M. D. Donovan. Carrier mediated transport of small molecules across bovine olfactory mucosa: implications in nose to brain transport. AAPS PharmSci 5:T3140 (2003).
6. M. A. Wioland, J. Fleury-Feith, P. Corlieu, F. Commo, G. Monceaux, J. Lacau-St-Guily, and J. F. Bernaudin. CFTR, MDR1, and MRP1 immunolocalization in normal human nasal respiratory mucosa. J. Histochem. Cytochem. 48:1215–1222 (2000).
7. P. Kwan, G. J. Sills, E. Butler, T. W. Gant, and M. J. Brodie. Differential expression of multidrug resistance genes in naive rat brain. Neurosci. Lett. 339:33–36 (2003).
8. J. H. Lin. Drug-drug interaction mediated by inhibition and induction of P-glycoprotein. Adv. Drug Deliv. Rev. 55:53–81 (2003).
9. J. W. Smit, M. T. Huisman, O. van Tellingen, H. R. Wiltshire, and A. H. Schinkel. Absence or pharmacological blocking of placental P-glycoprotein profoundly increases fetal drug exposure. J. Clin. Invest. 104:1441–1447 (1999).
10. U. Mayer, E. Wagenaar, B. Dorobek, J. H. Beijnen, P. Borst, and A. H. Schinkel. Full blockade of intestinal P-glycoprotein and extensive inhibition of blood-brain barrier P-glycoprotein by oral treatment of mice with PSC833. J. Clin. Invest. 100:2430–2436 (1997).
11. J. van Asperen, O. van Tellingen, F. Tijssen, A. H. Schinkel, and J. H. Beijnen. Increased accumulation of doxorubicin and doxorubicinol in cardiac tissue of mice lacking mdr1a P-glycoprotein. Br. J. Cancer 79:108–113 (1999).
12. C. Dagenais, C. Rousselle, G. M. Pollack, and J. M. Scherrmann. Development of an in situ mouse brain perfusion model and its application to mdr1a P-glycoprotein-deficient mice. J. Cereb. Blood Flow Metab. 20:381–386 (2000).
13. Q. R. Smith. Brain Perfusion Systems for Studies of Drug Uptake and Metabolism in the Central Nervous System. Plenum Press, New York, 1996.
14. J. Zong and G. M. Pollack. Modulation of P-glycoprotein transport activity in the mouse blood-brain barrier by rifampin. J. Pharmacol. Exp. Ther. 306:556–562 (2003).
15. E. M. Kemper, A. E. van Zandbergen, C. Cleypool, H. A. Mos, W. Boogerd, J. H. Beijnen, and O. van Tellingen. Increased penetration of paclitaxel into the brain by inhibition of P-glycoprotein. Clin. Cancer Res. 9:2849–2855 (2003).
16. C. L. Graff, R. Zhao, and G. M. Pollack. Pharmacokinetics of substrate uptake and distribution in murine brain after nasal instillation. Pharm. Res. (in press).
17. J. Skipor, W. Grzegorzewski, N. Einer-Jensen, and B. Wasowska. Local vascular pathway for progesterone transfer to the brain after nasal administration in gilts. Reprod. Biol. 3:143–159 (2003).
18. L. Illum. Transport of drugs from the nasal cavity to the central nervous system. Eur. J. Pharm. Sci. 11:1–18 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graff, C., Pollack, G. Functional Evidence for P-glycoprotein at the Nose-Brain Barrier. Pharm Res 22, 86–93 (2005). https://doi.org/10.1007/s11095-004-9013-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-004-9013-3