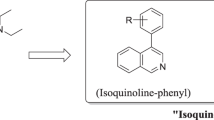
A series of hybrid molecules referred to as “reversed chloroquines” (RCQs) were designed, synthesized, and tested against chloroquine resistant strains of Plasmodium falciparum. The designed compounds contain bulky aromatic groups and chloroquine like nucleus linked with each other via two or three carbon alkyl linkers. Five compounds were designed based on the structure – activity relationship. These five compounds have been successfully synthesized, of which three compounds show significant antimalarial activity in vitro. A modification of the reversal agent (RA) like moiety was designed using substituted biphenyl groups. The aminoalkyl substitution of the heterocycle and tertiary aliphatic aminoalkyl nitrogen atom with two- or three-carbon bridges to the heteroaromatic nitrogen is required for potential “resistance reversal activity”. The synthesized compounds were screened for their in vitro antimalarial activity as characterized by the 50% inhibitory concentration (IC50), cytotoxic concentration (CC50), and selectivity index (SI). These compounds did not show any cytotoxic effect. One of these compounds (5), which contains two-carbon linker chain, unsaturation at position 2, and chlorine substituted biphenyls in RA-like moiety, was found to possess appreciable and promising in vitro antimalarial activity against the chloroquine sensitive 3D7 strain of P. falciparum.




Similar content being viewed by others
References
World Malaria Report, WHO, Geneva, Switzerland (2005).
R. G. Brian, D. A. Fidock, D. E. Kyle, et al., J. Clin. Invest., 118, 1267 – 1271 (2008).
R. G. Ridley, Nature, 415, 686 – 693 (2002).
D. Payne, Parasitol. Today, 3, 241 – 246 (1987).
X. Z. Su, L. A. Kirkman, H. Fujioka and T. E.Wellems, Cell, 91, 593 – 603 (1997).
D. A. Fidock, T. Nomura, A. K. Talley, et al., Mol. Cell, 6, 861 – 871 (2000).
T. J. Egan, R. Hunter, C. H. Kaschula, et al., J. Med. Chem., 43, 283 – 291 (2000).
C. H. Kaschula, T. J. Egan, R. Hunter, et al., J. Med. Chem., 45, 3531 – 3539 (2002).
P. A. Stocks, K. J. Raynes, P. G. Bray, et al., J. Med. Chem., 45, 4975 – 4983 (2002).
R. G. Ridley, W. Hofheinz, H. Matile, et al., Antimicrob. Agents Chemother., 40, 1846 – 54 (1996).
S. J. Burgess, A. Selzer, Kelly J. Xu, et al., J. Med. Chem., 49, 5623 – 5625 (2006).
S. J. Burgess, J. X. Kelly, S. Shomloo, et al., J. Med. Chem., 53, 6477 – 6489 (2010).
A. K. Bhattacharjee, D. E. Kyle, J. L. Vennerstrom and W. K. Milhous, J. Chem. Inf. Comput. Sci., 42, 1212 – 1220 (2002).
Acknowledgments
The authors are thankful to CDRI Lucknow for the biological evaluation of compounds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, R., Tiwari, A. & Parate, A. Synthesis of New Chloroquine Derivatives as Antimalarial Agents. Pharm Chem J 49, 537–542 (2015). https://doi.org/10.1007/s11094-015-1322-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-015-1322-9