Abstract
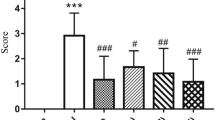
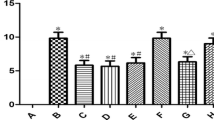
Piceatannol is a natural plant-derived compound with protective effects against cardiovascular diseases. However, its effect on cerebral ischaemia–reperfusion injury (CIRI) induced by oxidative stress remains unclear. This study aimed to investigate piceatannol’s antioxidation in CIRI. An in vitro oxygen–glucose deprivation followed by reoxygenation model was used and cell viability was measured. A middle cerebral artery occlusion followed by reperfusion model was used in vivo. Neurological function, encephalisation quotient, oedema, and volume of the cerebral infarction were then evaluated. The effects of piceatannol on histopathological findings, as well as the ultrastructure of the cortex, were analysed. The activity of superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and lactate dehydrogenase (LDH) and the malondialdehyde (MDA) content was measured both in vitro and in vivo. Finally, the expression of nuclear factor erythroid-2-related factor 2 (Nrf2), hemeoxygenase-1 (HO-1), and nicotinamide adenine dinucleotide phosphate quinone oxidoreductase 1 (NQO1) in cerebral tissue was detected using reverse transcription quantitative polymerase chain reaction (RT-qPCR) and western blotting. Our results demonstrated that cell viability in the piceatannol groups was increased. The SOD, GSH-Px activities were increased as LDH activity and MDA content decreased in the piceatannol groups both in vitro and in vivo, reflecting a decrease in oxidative stress. The neurological severity score and infarction volume in the piceatannol groups at doses of 10 and 20 mg/kg were lower than those of the model group. Furthermore, the damage seen on histopathological examination was partially attenuated by piceatannol. RT-qPCR and western blot analysis indicated that the expression of Nrf2, HO-1, and NQO1 were significantly increased by piceatannol. The results of the study demonstrate that piceatannol exerts a protective effect against CIRI.







Similar content being viewed by others
References
Rebouças Filho PP, Sarmento RM, Holanda GB, de Alencar LD (2017) New approach to detect and classify stroke in skull CT images via analysis of brain tissue densities. Comput Methods Programs Biomed 148:27–43. https://doi.org/10.1016/j.cmpb.2017.06.011
Wiesmann M, Zinnhardt B, Reinhardt D, Eligehausen S, Wachsmuth L, Hermann S, Dederen PJ, Hellwich M, Kuhlmann MT, Broersen LM, Heerschap A, Jacobs AH, Kiliaan AJ (2017) A specific dietary intervention to restore brain structure and function after ischemic stroke. Theranostics 7:493–512. https://doi.org/10.7150/thno.17559
Thorén M, Dixit A, Escudero-Martínez I, Gdovinová Z, Klecka L, Rand V-M, Toni D, Vilionskis A, Wahlgren N, Ahmed N (2020) Effect of recanalization on cerebral edema in ischemic stroke treated with thrombolysis and/or endovascular therapy. Stroke 51:216–223. https://doi.org/10.1161/STROKEAHA.119.026692
Yin P, Wei Y, Wang X, Zhu M, Feng J (2018) Roles of specialized pro-resolving lipid mediators in cerebral ischemia reperfusion injury. Front Neurol 9:1–17. https://doi.org/10.3389/fneur.2018.00617
Sies H (2015) Oxidative stress: a concept in redox biology and medicine. Redox Biol 4:180–183. https://doi.org/10.1016/j.redox.2015.01.002
Carvalho C, Moreira PI (2018) Oxidative stress: a major player in cerebrovascular alterations associated to neurodegenerative events. Front Physiol 9:1–14. https://doi.org/10.3389/fphys.2018.00806
Banerjee S, Ghosh S, Mandal A, Ghosh N, Sil PC (2020) ROS-associated immune response and metabolism: a mechanistic approach with implication of various diseases. Arch Toxicol 94:2293–2317. https://doi.org/10.1007/s00204-020-02801-7
Wang L, Pu Z, Li M, Wang K, Deng L, Chen W (2020) Antioxidative and antiapoptosis: neuroprotective effects of dauricine in Alzheimer’s disease models. Life Sci 243:117237. https://doi.org/10.1016/j.lfs.2019.117237
Dai X, Yan X, Wintergerst KA, Cai L, Keller BB, Tan Y (2020) Nrf2: redox and metabolic regulator of stem cell state and function. Trends Mol Med 26:185–200. https://doi.org/10.1016/j.molmed.2019.09.007
Chang C, Zhao Y, Song G, She K (2018) Resveratrol protects hippocampal neurons against cerebral ischemia–reperfusion injury via modulating JAK/ERK/STAT signaling pathway in rats. J Neuroimmunol 315:9–14. https://doi.org/10.1016/j.jneuroim.2017.11.015
Hosoda R, Hamada H, Uesugi D, Iwahara N, Nojima I, Horio Y, Kuno A (2021) Different antioxidative and antiapoptotic effects of piceatannol and resveratrol. J Pharmacol Exp Ther 376:385–396. https://doi.org/10.1124/jpet.120.000096
Hu WH, Dai DK, Zheng BZ-Y, Duan R, Dong TT-X, Qin Q-W, Tsim KW-K (2020) Piceatannol, a natural analog of resveratrol, exerts anti-angiogenic efficiencies by blockage of vascular endothelial growth factor binding to its receptor. Molecules 25:3769. https://doi.org/10.3390/molecules25173769
Lee HJ, Kang M-G, Cha HY, Kim YM, Lim Y, Yang SJ (2019) Effects of piceatannol and resveratrol on sirtuins and hepatic inflammation in high-fat diet-fed mice. J Med Food 22:833–840. https://doi.org/10.1089/jmf.2018.4261
Madreiter-Sokolowski CT, Gottschalk B, Parichatikanond W, Eroglu E, Klec C, Waldeck-Weiermair M, Malli R, Graier WF (2016) Resveratrol specifically kills cancer cells by a devastating increase in the Ca2+ coupling between the greatly tethered endoplasmic reticulum and mitochondria. Cell Physiol Biochem 39:1404–1420. https://doi.org/10.1159/000447844
Boutron I, Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl U, Emerson M, Garner P, Holgate ST, Howells DW, Karp NA, Lazic SE, Lidster K, MacCallum CJ, Macleod M, Pearl EJ, Petersen OH, Rawle F, Reynolds P, Rooney K, Sena ES, Silberberg SD, Steckler T, Würbel H (2020) The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. PLoS Biol 18:e3000410. https://doi.org/10.1113/JP280389
Song Y, Bei Y, Xiao Y, Tong HD, Wu XQ, Chen MT (2018) Edaravone, a free radical scavenger, protects neuronal cells’ mitochondria from ischemia by inactivating another new critical factor of the 5-lipoxygenase pathway affecting the arachidonic acid metabolism. Brain Res 1690:96–104. https://doi.org/10.1016/j.brainres.2018.03.006
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91. https://doi.org/10.1161/01.STR.20.1.84
Bieber M, Gronewold J, Scharf A-C, Schuhmann MK, Langhauser F, Hopp S, Mencl S, Geuss E, Leinweber J, Guthmann J, Doeppner TR, Kleinschnitz C, Stoll G, Kraft P, Hermann DM (2019) Validity and reliability of neurological scores in mice exposed to middle cerebral artery occlusion. Stroke 50:2875–2882. https://doi.org/10.1161/STROKEAHA.119.026652
Liu L, Ding J, Leng X, Pu Y, Huang L, Xu A, Wong K, Wang X, Wang Y (2018) Guidelines for evaluation and management of cerebral collateral circulation in ischaemic stroke 2017. Stroke Vasc Neurol 3:117–130. https://doi.org/10.1136/svn-2017-000135
Kim JS (2019) tPA helpers in the treatment of acute ischemic stroke: are they ready for clinical use? J Stroke 21:160–174. https://doi.org/10.5853/jos.2019.00584
Li C, Zhang L, Wang C, Teng H, Fan B, Chopp M, Zhang ZG (2019) N-acetyl-seryl-aspartyl-lysyl-proline augments thrombolysis of tPA (tissue-type plasminogen activator) in aged rats after stroke. Stroke 50:2547–2554. https://doi.org/10.1161/STROKEAHA.119.026212
Xue R, Du M, Zhou TY, Ai WZ, Zhang ZS, Xiang XW, Zhou YF, Wen ZS (2020) Polysaccharides from hemp seed protect against cyclophosphamide-induced intestinal oxidative damage via Nrf2-Keap1 signaling pathway in mice. Oxid Med Cell Longev 2020:1–13. https://doi.org/10.1155/2020/1813798
Xiao X, Lu Z, Lin V, May A, Shaw DH, Wang Z, Che B, Tran K, Du H, Shaw PX (2018) MicroRNA miR-24-3p reduces apoptosis and regulates Keap1-Nrf2 pathway in mouse cardiomyocytes responding to ischemia/reperfusion injury. Oxid Med Cell Longev 2018:1–9. https://doi.org/10.1155/2018/7042105
Fei D, Wang Y, Zhai Q, Zhang X, Zhang Y, Wang Y, Li B, Wang Q (2021) KAT6A regulates stemness of aging bone marrow-derived mesenchymal stem cells through Nrf2/ARE signaling pathway. Stem Cell Res Ther 12:104–117. https://doi.org/10.1186/s13287-021-02164-5
Kaspar JW, Niture SK, Jaiswal AK (2009) Nrf 2:INrf2 (Keap1) signaling in oxidative stress. Free Radic Biol Med 47:1304–1309. https://doi.org/10.1016/j.freeradbiomed.2009.07.035
Lv Y, Jiang H, Li S, Han B, Liu Y, Yang D, Li J, Yang Q, Wu P, Zhang Z (2020) Sulforaphane prevents chromium-induced lung injury in rats via activation of the Akt/GSK-3β/Fyn pathway. Environ Pollut 259:113812. https://doi.org/10.1016/j.envpol.2019.113812
Hao Y, Liu J, Wang Z, Yu L, Wang J (2019) Piceatannol protects human retinal pigment epithelial cells against hydrogen peroxide induced oxidative stress and apoptosis through modulating PI3K/Akt signaling pathway. Nutrients 11:1515. https://doi.org/10.3390/nu11071515
Narayanan SV, Dave KR, Saul I, Perez-Pinzon MA (2015) Resveratrol preconditioning protects against cerebral ischemic injury via nuclear erythroid 2–related factor 2. Stroke 46:1626–1632. https://doi.org/10.1161/STROKEAHA.115.008921
Sato D, Shimizu N, Shimizu Y, Akagi M, Eshita Y, Ozaki S-i, Nakajima N, Ishihara K, Masuoka N, Hamada H, Shimoda K, Kubota N (2014) Synthesis of glycosides of resveratrol, pterostilbene, and piceatannol, and their anti-oxidant, anti-allergic, and neuroprotective activities. Biosci Biotechnol Biochem 78:1123–1128. https://doi.org/10.1080/09168451.2014.921551
Sueishi Y, Nii R, Kakizaki N (2017) Resveratrol analogues like piceatannol are potent antioxidants as quantitatively demonstrated through the high scavenging ability against reactive oxygen species and methyl radical. Bioorg Med Chem Lett 27:5203–5206. https://doi.org/10.1016/j.bmcl.2017.10.045
Wen H, Fu Z, Wei Y, Zhang X, Ma L, Gu L, Li J (2018) Antioxidant activity and neuroprotective activity of stilbenoids in rat primary cortex neurons via the PI3K/Akt signalling pathway. Molecules 23:2328. https://doi.org/10.3390/molecules23092328
Funding
This work was funded by the National Natural Science Foundation of China (81573643), the Natural Science Foundation of ZheJiang Province (Y20H280057).
Author information
Authors and Affiliations
Contributions
LFW, XZ and ZH designed the study. LFW conducted the most of the experiments. YG provided valued guidance for the article writing and major revision. JY, ZP and PH assisted biochemical analysis. FQ and DZ provided technical support. LFW, XZ and ZH wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical Approval
All experiments were performed according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Animal Care Committee of Zhejiang Chinese Medical University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, L., Guo, Y., Ye, J. et al. Protective Effect of Piceatannol Against Cerebral Ischaemia–Reperfusion Injury Via Regulating Nrf2/HO-1 Pathway In Vivo and Vitro. Neurochem Res 46, 1869–1880 (2021). https://doi.org/10.1007/s11064-021-03328-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-021-03328-8