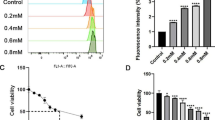
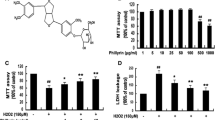
Abstract
Aldehyde stress contributes to molecular mechanisms of cell death and the pathogenesis of Parkinson’s disease (PD). The neurotoxin 1-Methy-4-Phenylpyridinium Ion (MPP+) is commonly used to model PD. Aldehyde dehydrogenase 2 (ALDH2) is an important enzyme detoxifying aldehydes. The aim of this study is to evaluate whether MPP+-induced neurotoxicity is involved in aldehyde stress by modulation of ALDH2. Our results demonstrated that treatment of PC12 cells with MPP+ leads to aldehyde stress by increasing in loads of malondialdehyde and 4-hydroxynonenal, which indicated that MPP+-induced aldehyde stress contributes to its cytotoxicity in PC12 cells. We also showed that MPP+ up-regulates the expression and activity of ALDH2 in PC12 cells and that inhibition of ALDH2 by its specific inhibitor daidzin prevents MPP+-induced decrease in cell viability and increases in apoptosis, oxidative stress and aldehyde stress in PC12 cells. These findings suggest that aldehyde stress contributes to MPP+-induced toxicity in PC12 cells by upregulation of ALDH2. This study provides a novel insight into the role of ALDH2 in the neurotoxicity of MPP+.










Similar content being viewed by others
References
Dawson TM, Dawson VL (2002) Neuroprotective and neurorestorative strategies for Parkinson’s disease. Nat Neurosci 5(Suppl):1058–1061
Wood PL, Khan MA, Kulow SR, Mahmood SA, Moskal JR (2006) Neurotoxicity of reactive aldehydes: the concept of “aldehyde load” as demonstrated by neuroprotection with hydroxylamines. Brain Res 1095:190–199
Wood PL, Khan MA, Moskal JR (2007) The concept of “aldehyde load” in neurodegenerative mechanisms: cytotoxicity of the polyamine degradation products hydrogen peroxide, acrolein, 3-aminopropanal, 3-acetamidopropanal and 4-aminobutanal in a retinal ganglion cell line. Brain Res 1145:150–156
Wood PL (2006) Neurodegeneration and aldehyde load: from concept to therapeutics. J Psychiatry Neurosci 31:296–297
Ellis EM (2007) Reactive carbonyls and oxidative stress: potential for therapeutic intervention. Pharmacol Ther 115:13–24
Uchida K (2000) Role of reactive aldehyde in cardiovascular diseases. Free Radic Biol Med 28:1685–1696
Aldini G, Dalle-Donne I, Facino RM, Milzani A, Carini M (2007) Intervention strategies to inhibit protein carbonylation by lipoxidation-derived reactive carbonyls. Med Res Rev 27:817–868
Jinsmaa Y, Florang VR, Rees JN, Mexas LM, Eckert LL et al (2011) Dopamine-derived biological reactive intermediates and protein modifications: implications for Parkinson’s disease. Chem Biol Interact 192:118–121
Qin Z, Hu D, Han S, Reaney SH, Di Monte DA et al (2007) Effect of 4-hydroxy-2-nonenal modification on alpha-synuclein aggregation. J Biol Chem 282:5862–5870
Yoritaka A, Hattori N, Uchida K, Tanaka M, Stadtman ER et al (1996) Immunohistochemical detection of 4-hydroxynonenal protein adducts in Parkinson disease. Proc Natl Acad Sci USA 93:2696–2701
Selley ML (1998) (E)-4-hydroxy-2-nonenal may be involved in the pathogenesis of Parkinson’s disease. Free Radic Biol Med 25:169–174
Farooqui T, Farooqui AA (2011) Lipid-mediated oxidative stress and inflammation in the pathogenesis of Parkinson’s disease. Parkinsons Dis 2011:247467
Xiang W, Schlachetzki JC, Helling S, Bussmann JC, Berlinghof M et al (2013) Oxidative stress-induced posttranslational modifications of alpha-synuclein: specific modification of alpha-synuclein by 4-hydroxy-2-nonenal increases dopaminergic toxicity. Mol Cell Neurosci 54:71–83
Przedborski S, Tieu K, Perier C, Vila M (2004) MPTP as a mitochondrial neurotoxic model of Parkinson’s disease. J Bioenerg Biomembr 36:375–379
Abou-Sleiman PM, Muqit MM, Wood NW (2006) Expanding insights of mitochondrial dysfunction in Parkinson’s disease. Nat Rev Neurosci 7:207–219
Zhu Q, Wang J, Zhang Y, Sun S (2012) Mechanisms of MPP(+)-induced PC12 cell apoptosis via reactive oxygen species. J Huazhong Univ Sci Technolog Med Sci 32:861–866
Zawada WM, Banninger GP, Thornton J, Marriott B, Cantu D et al (2011) Generation of reactive oxygen species in 1-methyl-4-phenylpyridinium (MPP+) treated dopaminergic neurons occurs as an NADPH oxidase-dependent two-wave cascade. J Neuroinflammation 8:129
Tang XQ, Fan LL, Li YJ, Shen XT, Zhuan YY et al (2011) Inhibition of hydrogen sulfide generation contributes to 1-methy-4-phenylpyridinium ion-induced neurotoxicity. Neurotox Res 19:403–411
Qian JJ, Cheng YB, Yang YP, Mao CJ, Qin ZH et al (2008) Differential effects of overexpression of wild-type and mutant human alpha-synuclein on MPP+ -induced neurotoxicity in PC12 cells. Neurosci Lett 435:142–146
Wu Y, Shang Y, Sun S, Liang H, Liu R (2007) Erythropoietin prevents PC12 cells from 1-methyl-4-phenylpyridinium ion-induced apoptosis via the Akt/GSK-3beta/caspase-3 mediated signaling pathway. Apoptosis 12:1365–1375
Yamamoto N, Sawada H, Izumi Y, Kume T, Katsuki H et al (2007) Proteasome inhibition induces glutathione synthesis and protects cells from oxidative stress: relevance to Parkinson disease. J Biol Chem 282:4364–4372
Tang XQ, Li YJ, Zhao J, Shen XT, Yang CT et al (2010) Neuroprotective effect of asymmetric dimethylarginine against 1-methyl-4-phenylpyridinium ion-induced damage in PC12 cells. Clin Exp Pharmacol Physiol 37:530–535
Siddiqui MA, Kumar V, Kashyap MP, Agarwal M, Singh AK et al (2012) Short-term exposure of 4-hydroxynonenal induces mitochondria-mediated apoptosis in PC12 cells. Hum Exp Toxicol 31:336–345
Chen CH, Budas GR, Churchill EN, Disatnik MH, Hurley TD et al (2008) Activation of aldehyde dehydrogenase-2 reduces ischemic damage to the heart. Science 321:1493–1495
Hyun DH, Lee MH, Halliwell B, Jenner P (2002) Proteasomal dysfunction induced by 4-hydroxy-2,3-trans-nonenal, an end-product of lipid peroxidation: a mechanism contributing to neurodegeneration? J Neurochem 83:360–370
Lee WC, Wong HY, Chai YY, Shi CW, Amino N et al (2012) Lipid peroxidation dysregulation in ischemic stroke: plasma 4-HNE as a potential biomarker? Biochem Biophys Res Commun 425:842–847
Ohsawa I, Nishimaki K, Yasuda C, Kamino K, Ohta S (2003) Deficiency in a mitochondrial aldehyde dehydrogenase increases vulnerability to oxidative stress in PC12 cells. J Neurochem 84:1110–1117
Keung WM (2001) Biogenic aldehyde(s) derived from the action of monoamine oxidase may mediate the antidipsotropic effect of daidzin. Chem Biol Interact 130–132:919–930
Keung WM, Vallee BL (1993) Daidzin: a potent, selective inhibitor of human mitochondrial aldehyde dehydrogenase. Proc Natl Acad Sci USA 90:1247–1251
Sai T, Uchida K, Nakayama H (2013) Involvement of monoamine oxidase-B in the acute neurotoxicity of MPTP in embryonic and newborn mice. Exp Toxicol Pathol 65:365–373
Hsieh YC, Mounsey RB, Teismann P (2011) MPP(+)-induced toxicity in the presence of dopamine is mediated by COX-2 through oxidative stress. Naunyn Schmiedebergs Arch Pharmacol 384:157–167
Jin BK, Shin DY, Jeong MY, Gwag MR, Baik HW et al (1998) Melatonin protects nigral dopaminergic neurons from 1-methyl-4-phenylpyridinium (MPP+) neurotoxicity in rats. Neurosci Lett 245:61–64
Jang YJ, Kim J, Shim J, Byun S, Oak MH et al (2011) Kaempferol attenuates 4-hydroxynonenal-induced apoptosis in PC12 cells by directly inhibiting NADPH oxidase. J Pharmacol Exp Ther 337:747–754
Mattammal MB, Haring JH, Chung HD, Raghu G, Strong R (1995) An endogenous dopaminergic neurotoxin: implication for Parkinson’s disease. Neurodegeneration 4:271–281
Burke WJ, Li SW, Williams EA, Nonneman R, Zahm DS (2003) 3,4-Dihydroxyphenylacetaldehyde is the toxic dopamine metabolite in vivo: implications for Parkinson’s disease pathogenesis. Brain Res 989:205–213
Rojas P, Serrano-Garcia N, Medina-Campos ON, Pedraza-Chaverri J, Maldonado PD et al (2011) S-Allylcysteine, a garlic compound, protects against oxidative stress in 1-methyl-4-phenylpyridinium-induced parkinsonism in mice. J Nutr Biochem 22:937–944
Chiueh CC, Andoh T, Lai AR, Lai E, Krishna G (2000) Neuroprotective strategies in Parkinson’s disease: protection against progressive nigral damage induced by free radicals. Neurotox Res 2:293–310
Chan KC, Mong MC, Yin MC (2009) Antioxidative and anti-inflammatory neuroprotective effects of astaxanthin and canthaxanthin in nerve growth factor differentiated PC12 cells. J Food Sci 74:H225–H231
Cheng B, Yang X, Chen C, Cheng D, Xu X et al (2010) D-beta-hydroxybutyrate prevents MPP+ -induced neurotoxicity in PC12 cells. Neurochem Res 35:444–451
Conklin D, Prough R, Bhatanagar A (2007) Aldehyde metabolism in the cardiovascular system. Mol BioSyst 3:136–150
Vasiliou V, Pappa A, Estey T (2004) Role of human aldehyde dehydrogenases in endobiotic and xenobiotic metabolism. Drug Metab Rev 36:279–299
Vasiliou V, Nebert DW (2005) Analysis and update of the human aldehyde dehydrogenase (ALDH) gene family. Hum Genomics 2:138–143
Yoval-Sanchez B, Rodriguez-Zavala JS (2012) Differences in susceptibility to inactivation of human aldehyde dehydrogenases by lipid peroxidation byproducts. Chem Res Toxicol 25:722–729
Solito R, Corti F, Chen CH, Mochly-Rosen D, Giachetti A et al (2013) Mitochondrial aldehyde dehydrogenase-2 activation prevents beta-amyloid-induced endothelial cell dysfunction and restores angiogenesis. J Cell Sci 126:1952–1961
Ohta S, Ohsawa I, Kamino K, Ando F, Shimokata H (2004) Mitochondrial ALDH2 deficiency as an oxidative stress. Ann N Y Acad Sci 1011:36–44
Ma H, Guo R, Yu L, Zhang Y, Ren J (2011) Aldehyde dehydrogenase 2 (ALDH2) rescues myocardial ischaemia/reperfusion injury: role of autophagy paradox and toxic aldehyde. Eur Heart J 32:1025–1038
He L, Liu B, Dai Z, Zhang HF, Zhang YS et al (2012) Alpha lipoic acid protects heart against myocardial ischemia-reperfusion injury through a mechanism involving aldehyde dehydrogenase 2 activation. Eur J Pharmacol 678:32–38
Kong D, Kotraiah V (2012) Modulation of aldehyde dehydrogenase activity affects (±)-4-hydroxy-2E-nonenal (HNE) toxicity and HNE-protein adduct levels in PC12 cells. J Mol Neurosci 47:595–603
Wey MC, Fernandez E, Martinez PA, Sullivan P, Goldstein DS et al (2012) Neurodegeneration and motor dysfunction in mice lacking cytosolic and mitochondrial aldehyde dehydrogenases: implications for Parkinson’s disease. PLoS ONE 7:e31522
Fitzmaurice AG, Rhodes SL, Lulla A, Murphy NP, Lam HA et al (2013) Aldehyde dehydrogenase inhibition as a pathogenic mechanism in Parkinson disease. Proc Natl Acad Sci USA 110:636–641
Klaidman LK, Adams JD Jr, Leung AC, Kim SS, Cadenas E (1993) Redox cycling of MPP+: evidence for a new mechanism involving hydride transfer with xanthine oxidase, aldehyde dehydrogenase, and lipoamide dehydrogenase. Free Radic Biol Med 15:169–179
Acknowledgments
This study was supported by Natural Science Foundation of China (81200986, 81202518), Natural Science Foundation of Hunan Province, China (11JJ3117) and the construct program of the key discipline in Hunan province.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Ai-Hua Chen and Ping Zhang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, AH., Zhang, P., Yin, WL. et al. Role of Aldehyde Dehydrogenase 2 in 1-Methy-4-Phenylpyridinium Ion-Induced Aldehyde Stress and Cytotoxicity in PC12 Cells. Neurochem Res 39, 1767–1775 (2014). https://doi.org/10.1007/s11064-014-1376-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-014-1376-1