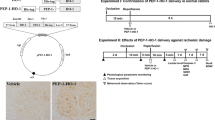
Abstract
A rabbit model of spinal cord ischemia has been introduced as a good model to investigate the pathophysiology of ischemia–reperfusion (I–R)-induced paraplegia. In the present study, we observed the effects of Cu,Zn-superoxide dismutase (SOD1) against ischemic damage in the ventral horn of L5–6 levels in the rabbit spinal cord. For this study, the expression vector PEP-1 was constructed, and this vector was fused with SOD1 to create a PEP-1-SOD1 fusion protein that easily penetrated the blood–brain barrier. Spinal cord ischemia was induced by transient occlusion of the abdominal aorta for 15 min. PEP-1-SOD1 (0.5 mg/kg) was intraperitoneally administered to rabbits 30 min before ischemic surgery. The administration of PEP-1-SOD1 significantly improved neurological scores compared to those in the PEP-1 (vehicle)-treated ischemia group. Also, in this group, the number of cresyl violet-positive cells at 72 h after I–R was much higher than that in the vehicle-treated ischemia group. Malondialdehyde levels were significantly decreased in the ischemic spinal cord of the PEP-1-SOD1-treated ischemia group compared to those in the vehicle-treated ischemia group. In contrast, the administration of PEP-1-SOD1 significantly ameliorated the ischemia–induced reduction of SOD and catalase levels in the ischemic spinal cord. These results suggest that PEP-1-SOD1 protects neurons from spinal ischemic damage by decreasing lipid peroxidation and maintaining SOD and catalase levels in the ischemic rabbit spinal cord.




Similar content being viewed by others
References
Kuniyoshi Y, Koja K, Miyagi K et al (2003) Prevention of postoperative paraplegia during thoracoabdominal aortic surgery. Ann Thorac Surg 76:1477–1484
MacArthur RG, Carter SA, Coselli JS, LeMaire SA (2005) Organ protection during thoracoabdominal aortic surgery: rationale for a multimodality approach. Semin Cardiothorac Vasc Anesth 9:143–149
Zivin JA, DeGirolami U (1980) Spinal cord infarction: a highly reproducible stroke model. Stroke 11:200–202
DeGirolami U, Zivin JA (1982) Neuropathology of experimental spinal cord ischemia in the rabbit. J Neuropathol Exp Neurol 41:129–149
Barut S, Canbolat A, Bilge T, Aydin Y, Cokneşeli B, Kaya U (1993) Lipid peroxidation in experimental spinal cord injury: time-level relationship. Neurosurg Rev 16:53–59
Lin R, Roseborough G, Dong Y, Williams GM, Wei C (2003) DNA damage and repair system in spinal cord ischemia. J Vasc Surg 37:847–858
Chronidou F, Apostolakis E, Papapostolou I et al (2009) Beneficial effect of the oxygen free radical scavenger amifostine (WR-2721) on spinal cord ischemia/reperfusion injury in rabbits. J Cardiothorac Surg 4:50
Shaafi S, Afrooz MR, Hajipour B, Dadadshi A, Hosseinian MM, Khodadadi A (2011) Anti-oxidative effect of lipoic acid in spinal cord ischemia/reperfusion. Med Princ Pract 20:19–22
Riley PA (1994) Free radicals in biology: oxidative stress and the effects of ionizing radiation. Int J Radiat Biol 65:27–33
Crapo JD, Oury T, Rabouille C, Slot JW, Chang LY (1992) Copper, zinc superoxide dismutase is primarily a cytosolic protein in human cells. Proc Natl Acad Sci USA 89:10405–10409
Chan PH, Kawase M, Murakami K et al (1998) Overexpression of SOD1 in transgenic rats protects vulnerable neurons against ischemic damage after global cerebral ischemia and reperfusion. J Neurosci 18:8292–8299
Eum WS, Kim DW, Hwang IK et al (2004) In vivo protein transduction: biologically active intact pep-1-superoxide dismutase fusion protein efficiently protects against ischemic insult. Free Radic Biol Med 37:1656–1669
Hwang IK, Eum WS, Yoo KY et al (2005) Copper chaperone for Cu,Zn-SOD supplement potentiates the Cu,Zn-SOD function of neuroprotective effects against ischemic neuronal damage in the gerbil hippocampus. Free Radic Biol Med 39:392–402
Kofler J, Hurn PD, Traystman RJ (2005) SOD1 overexpression and female sex exhibit region-specific neuroprotection after global cerebral ischemia due to cardiac arrest. J Cereb Blood Flow Metab 25:1130–1137
Davis AS, Zhao H, Sun GH, Sapolsky RM, Steinberg GK (2007) Gene therapy using SOD1 protects striatal neurons from experimental stroke. Neurosci Lett 411:32–36
Deshayes S, Heitz A, Morris MC, Charnet P, Divita G, Heitz F (2004) Insight into the mechanism of internalization of the cell-penetrating carrier peptide Pep-1 through conformational analysis. Biochemistry 43:1449–1457
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cho JH, Hwang IK, Yoo KY et al (2008) Effective delivery of Pep-1-cargo protein into ischemic neurons and long-term neuroprotection of Pep-1-SOD1 against ischemic injury in the gerbil hippocampus. Neurochem Int 52:659–668
Kiyoshima T, Fukuda S, Matsumoto M et al (2003) Lack of evidence for apoptosis as a cause of delayed onset paraplegia after spinal cord ischemia in rabbits. Anesth Analg 96:839–846
Jacobs TP, Kempski O, McKinley D, Dutka AJ, Hallenbeck JM, Feuerstein G (1992) Blood flow and vascular permeability during motor dysfunction in a rabbit model of spinal cord ischemia. Stroke 23:367–373
Huang Y, Xie K, Li J et al (2011) Beneficial effects of hydrogen gas against spinal cord ischemia-reperfusion injury in rabbits. Brain Res 1378:125–136
Moore WM Jr, Hollier LH (1991) The influence of severity of spinal cord ischemia in the etiology of delayed-onset paraplegia. Ann Surg 213:427–431
Wisselink W, Patetsios P, Panetta TF et al (1998) Medium molecular weight pentastarch reduces reperfusion injury by decreasing capillary leak in an animal model of spinal cord ischemia. J Vasc Surg 27:109–116
Choi HS, An JJ, Kim SY et al (2006) PEP-1-SOD fusion protein efficiently protects against paraquat-induced dopaminergic neuron damage in a Parkinson disease mouse model. Free Radic Biol Med 41:1058–1068
An JJ, Lee YP, Kim SY et al (2008) Transduction of familial amyotrophic lateral sclerosis-related mutant PEP-1-SOD proteins into neuronal cells. Mol Cells 25:55–63
Yune TY, Lee JY, Jiang MH, Kim DW, Choi SY, Oh TH (2008) Systemic administration of PEP-1-SOD1 fusion protein improves functional recovery by inhibition of neuronal cell death after spinal cord injury. Free Radic Biol Med 45:1190–1200
Zhang YE, Fu SZ, Li XQ et al (2011) PEP-1-SOD1 protects brain from ischemic insult following asphyxial cardiac arrest in rats. Resuscitation 82:1081–1086
Zhang YE, Wang JN, Tang JM et al (2009) In vivo protein transduction: delivery of PEP-1-SOD1 fusion protein into myocardium efficiently protects against ischemic insult. Mol Cells 27:159–166
Marnett LJ (2002) Oxy radicals, lipid peroxidation and DNA damage. Toxicology 181–182:219–222
Guven C, Borcek AO, Cemil B et al (2010) Neuroprotective effects of infliximab in experimental spinal cord ischemic injury. J Clin Neurosci 17:1563–1567
Kurt G, Ergün E, Cemil B et al (2009) Neuroprotective effects of infliximab in experimental spinal cord injury. Surg Neurol 71:332–336
Vural M, Arslantaş A, Yazihan N et al (2010) NMDA receptor blockage with 2-amino-5-phosphonovaleric acid improves oxidative stress after spinal cord trauma in rats. Spinal Cord 48:285–289
Ilhan A, Koltuksuz U, Ozen S, Uz E, Ciralik H, Akyol O (1999) The effects of caffeic acid phenethyl ester (CAPE) on spinal cord ischemia/reperfusion injury in rabbits. Eur J Cardiothorac Surg 16:458–463
Ilhan A, Yilmaz HR, Armutcu F, Gurel A, Akyol O (2004) The protective effect of nebivolol on ischemia/reperfusion injury in rabbit spinal cord. Prog Neuropsychopharmacol Biol Psychiatry 28:1153–1160
Khalaj L, Peirovi H, Khodagholi F, Abdi A, Dargahi L, Ahmadiani A (2011) Acute 17β-estradiol pretreatment protects against abdominal aortic occlusion induced spinal cord ischemic-reperfusion injury. Neurochem Res 36:268–280
Savaş S, Delibaş N, Savaş C, Sütçü R, Cindaş A (2002) Pentoxifylline reduces biochemical markers of ischemia-reperfusion induced spinal cord injury in rabbits. Spinal Cord 40:224–229
Acknowledgments
The authors would like to thank Mr. Seok Han, Mr. Seung Uk Lee and Ms. Hyun Sook Kim for their technical help in this study. This work was supported by a grant no. 01-2006-04 from the Hallym University Medical Center Research Fund.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11064_2011_613_MOESM1_ESM.tif
Supplementary material 1 (TIFF 12371 kb). Neurological function scores by modified Tarlov criteria in the sham, PEP-1-ischemia and PEP-1-SOD1-ischemia groups at 24, 48, and 72 h after I-R
Rights and permissions
About this article
Cite this article
Kim, W., Kim, D.W., Yoo, D.Y. et al. Neuroprotective Effects of PEP-1-Cu,Zn-SOD against Ischemic Neuronal Damage in the Rabbit Spinal Cord. Neurochem Res 37, 307–313 (2012). https://doi.org/10.1007/s11064-011-0613-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-011-0613-0