Abstract
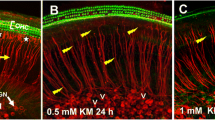
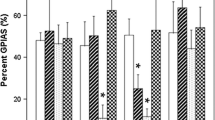
Salicylate-induced ototoxicity leading to sensorineural hearing loss (SNHL) and tinnitus is well documented. However, the exact mechanisms are poorly defined. Caspase-3 is a member of the class of effector caspases and has been activated in nearly every model of apoptosis. To examine its role in salicylate-induced injury, we subjected guinea pigs to treatment with a specific inhibitor zDEVD-FMK via the round window niche (RWN) followed by a systemic injection of salicylate at a dose of 200 mg·kg−1·d−1 i.p. for 10 consecutive days. For those animals administered with salicylate, immunohistochemical studies revealed that caspase-3 was activated in the spiral ganglion neurons (SGNs) and method of terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end-labeling (TUNEL) to identify neuronal apoptosis showed that fragmented nuclei were distributed in Rosenthal’s canal. Topical administration of the zDEVD-FMK at a concentration of 500 mM blocked caspase-3 activation and had an effect in reducing the number of TUNEL-positive auditory neurons. In contrast, the inhibitor at a concentration of 125 or 250 mM caused no variation in the expression of activated caspase-3, or in the ratio of TUNEL-positive neurons. These results indicate that caspase-3 is a crucial mediator of apoptosis induced by salicylate in the primary auditory neuron in vivo, and suggest that the specific inhibitor at a relatively high concentration may be therapeutically beneficial in salicylate-induced apoptosis.




Similar content being viewed by others
Abbreviations
- AI:
-
Apoptosis index
- AOD:
-
Average optical density
- APL:
-
Artificial perilymph
- DPOAEs:
-
Distortion product otoacoustic emission
- IHCs:
-
Inner hair cells
- IOD:
-
Integrated optical density
- OC:
-
The organ of Corti
- OHCs:
-
Outer hair cells
- RWN:
-
Round window niche
- SGNs:
-
Spiral ganglion neurons
- SNHL:
-
Sensorineural hearing loss
- TUNEL:
-
Terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end-labeling
References
Cazals Y (2000) Auditory sensori-neural alterations induced by salicylate. Prog Neurobiol 62:583–631
Escoubet B, Amsallem P, Ferrary E et al (1985) Prostaglandin synthesis by the cochlea of the guinea pig. Influence of aspirin, gentamicin, and acoustic stimulation. Prostaglandins 29:589–599
Jung TTK, Miller SK, Rozehnal S et al (1992) Effect of round window membrane application of salicylate and indomethacin on hearing and levels of arachidonic acid metabolites in perilymph. Acta Otolaryngol Suppl 493:81–87
Shehata WE, Brownell WE, Dieler R (1991) Effects of salicylate on shape, electromotility and membrane characteristics of isolated outer hair cells from guinea pig cochlea. Acta Otolaryngol 111:707–718
Lue AJ, Brownell WE (1999) Salicylate induced changes in outer hair cell lateral wall stiffness. Hear Res 135:163–168
Yu N, Zhu ML, Johnson B et al (2008) Prestin up-regulation in chronic salicylate (aspirin) administration: an implication of functional dependence of prestin expression. Cell Mol Life Sci 65:2407–2418
Janssen T, Boege P, Oestreicher E et al (2000) Tinnitus and 2f1–f2 distortion product otoacoustic emissions following salicylate overdose. J Acoust Soc Am 107:1790–1792
Huang ZW, Luo Y, Wu Z et al (2005) Paradoxical enhancement of active cochlear mechanics in long-term administration of salicylate. J Neurophysiol 93:2053–2061
Dieler R, Shehata-Dieler WE, Brownell WE (1991) Concomitant salicylate-induced alterations of outer hair cell subsurface cisternae and electromotility. J Neurocytol 20:637–653
Tunstall MJ, Gale JE, Ashmore JF (1995) Action of salicylate on membrane capacitance of outer hair cells from the guinea-pig cochlea. J Physiol 485:739–752
Kakehata S, Santos-Sacchi J (1996) Effects of salicylate and lanthanides on outer hair cell motility and associated gating charge. J Neurosci 16:4881–4889
Takahashi ST, Ohyama KO, Ikeda KI et al. (1999) Salicylate effects on the distortion products in live guinea pig cochlea. In: Association for Research in Otolaryngology, vol B. p 131
Schuknecht HF (1994) Auditory and cytocochlear correlates of inner ear disorders. Otolaryngol Head Neck Surg 110:530–538
Starr A, Picton TW, Sininger Y et al (1996) Auditory neuropathy. Brain 119:741–753
Incesulu A, Nadol JB Jr (1998) Correlation of acoustic threshold measures and spiral ganglion cell survival in severe to profound sensorineural hearing loss: implications for cochlear implantation. Ann Otol Rhinol Laryngol 107:906–911
Liu Y, Li X, Ma C et al (2005) Salicylate blocks l-type calcium channels in rat inferior colliculus neurons. Hear Res 205:271–276
Liu Y, Zhang H, Li X et al (2007) Inhibition of voltage-gated channel currents in rat auditory cortex neurons by salicylate. Neuropharmacology 53:870–880
Liu Y, Li X (2004) Effects of salicylate on transient outward and delayed rectifier potassium channels in rat inferior colliculus neurons. Neurosci Lett 369:115–120
Morishita Y, Sakube Y, Sasaki S et al (2004) Molecular mechanisms and drug development in aquaporin water channel diseases: aquaporin superfamily (superaquaporins): expansion of aquaporins restricted to multicellular organisms. J Pharmacol Sci 96:276–279
Liu Y, Li X (2004) Effects of salicylate on voltage-gated sodium channels in rat inferior colliculus neurons. Hear Res 193:68–74
Niedzielski AS, Wenthold RJ (1995) Expression of AMPA, kainate and NMDA receptor subunits in cochlear and vestibular ganglia. J Neurosci 15:2338–2353
Guitton MJ, Caston J, Ruel J et al (2003) Salicylate induces tinnitus through activation of cochlear NMDA receptors. J Neurosci 23:3944–3952
Peng BG, Chen S, Lin X (2003) Aspirin selectively augmented N-methyl-D-aspartate types of glutamate responses in cultured spiral ganglion neurons of mice. Neurosci Lett 343:21–24
Bauer CA, Brozoski TJ, Holder TM et al (2000) Effects of chronic salicylate on GABAergic activity in rat inferior colliculus. Hear Res 147:175–182
Nakagawa T, Yamashita M, Hisashi K et al (2005) GABA-induced response in spiral ganglion cells acutely isolated from guinea pig. Neurosci Res 53:396–403
Xu H, Gong N, Chen L et al (2005) Sodium salicylate reduces gamma aminobutyric acid—induced current in rat spinal dorsal horn neurons. Neuroreport 16:813–816
Wei L, Ding D, Salvi R (2010) Salicylate-induced degeneration of cochlea spiral ganglion neurons-apoptosis signaling. Neuroscience 168:288–299
Liu W, Staecker H, Stupak H et al (1998) Caspase inhibitors prevent cisplatin-induced apoptosis of auditory sensory cells. Neuroreport 9:2609–2614
Matsui JI, Ogilvie JM, Warchol ME (2002) Inhibition of caspases prevents ototoxic and ongoing hair cell death. J Neurosci 22:1218–1227
Feng H, Yin SH, Tang AZ et al (2010) Caspase-3 activation in the guinea pig cochlea exposed to salicylate. Neurosci Lett 479:34–39
Bobbin RP, Konishi T (1974) Action of cholinergic and anticholinergic drugs at the crossed olive-cochlear bundle-hair cell junction. Acta Otolaryngol 77:55–65
Jung TT, Kim JP, Bunn J et al (1997) Effect of leukotriene inhibitor on salicylate induced morphologic changes of isolated cochlear outer hair cells. Acta Otolaryngol 117:258–264
Jung TT, Hwang AL, Miller SK et al (1995) Effect of leukotriene inhibitor on cochlear blood flow in salicylate ototoxicity. Acta Otolaryngol Suppl 115:251–254
Jeong SW, Kim LS, Hur D et al (2010) Gentamicin-induced spiral ganglion cell death: apoptosis mediated by ROS and the JNK signaling pathway. Acta Otolaryngol 130:670–678
Karnes HE, Scaletty PN, Durham D (2010) Histochemical and fluorescent analyses of mitochondrial integrity in chick auditory neurons following deafferentation. J Am Acad Audiol 21:204–218
Porter AG, Jänicke RU (1999) Emerging roles of caspase-3 in apoptosis. Cell Death Differ 6:99–104
Karnes HE, Kaiser CL, Durham D (2009) Deafferentation-induced caspase-3 activation and DNA fragmentation in chick cochlear nucleus neurons. Neuroscience 159:804–818
Sha SH, Chen FQ, Schacht J (2009) Activation of cell death pathways in the inner ear of the aging CBA/J mouse. Hear Res 254:92–99
Liu X, Zou H, Slaughter C et al (1997) DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 89:175–184
Kothakota S, Azuma T, Reinhard C et al (1997) Caspase-3-generated fragment of gelsolin: effector of morphological change in apoptosis. Science 278:294–298
Matsui JI, Haque A, Huss D et al (2003) Caspase inhibitors promote vestibular hair cell survival and function after aminoglycoside treatment in vivo. J Neurosci 23:6111–6122
Mizutari K, Matsunaga T, Kamiya K et al (2008) Caspase inhibitor facilitates recovery of hearing by protecting the cochlear lateral wall from acute cochlear mitochondrial dysfunction. J Neurosci Res 86:215–222
Steinbach S, Lutz J (2007) Glutamate induces apoptosis in cultured spiral ganglion explants. Biochem Biophys Res Commun 357:14–19
Kaiser CL, Chapman BJ, Guidi JL et al (2008) Comparison of activated caspase detection methods in the gentamicin-treated chick cochlea. Hear Res 240:1–11
Wang J, Ladrech S, Pujol R et al (2004) Caspase inhibitors, but not c-Jun NH2-terminal kinase inhibitor treatment, prevent cisplatin-induced hearing loss. Cancer Res 64:9217–9224
Atar O, Avraham KB (2010) Anti-apoptotic factor z-Val-Ala-Asp-fluoromethylketone promotes the survival of cochlear hair cells in a mouse model for human deafness. Neuroscience 168:851–857
Yamashita D, Miller JM, Jiang HY et al (2004) AIF and EndoG in noise-induced hearing loss. Neuroreport 15:2719–2722
Jiang H, Sha SH, Forge A et al (2006) Caspase-independent pathways of hair cell death induced by kanamycin in vivo. Cell Death Differ 13:20–30
Zine A, Van de Water TR (2004) The MAPK/JNK signalling pathway offers potential therapeutic targets for the prevention of acquired deafness. Curr Drug Targets CNS Neurol Disord 3:325–332
Tabuchi K, Oikawa K, Hoshino T et al (2010) Cochlear protection from acoustic injury by inhibitors of p38 mitogen-activated protein kinase and sequestosome 1 stress protein. Neuroscience 166:665–670
Meltser I, Tahera Y, Canlon B et al (2010) Differential activation of mitogen-activated protein kinases and brain-derived neurotrophic factor after temporary or permanent damage to a sensory system. Neuroscience 165:1439–1446
Acknowledgments
This study was supported by National Natural Science Foundation of China (No. 30860312) and China Postdoctoral Science Foundation (No. 20080440819). We thank Mr. Daniel J. Clark for his careful proofreading.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Feng, H., Yin, SH., Tang, AZ. et al. Salicylate Initiates Apoptosis in the Spiral Ganglion Neuron of Guinea Pig Cochlea by Activating Caspase-3. Neurochem Res 36, 1108–1115 (2011). https://doi.org/10.1007/s11064-011-0455-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-011-0455-9