Abstract
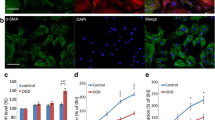
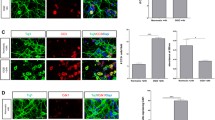
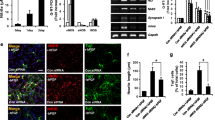
Activation of cyclin dependent kinases (Cdks) contributes to neuronal death following ischemia. We used oxygen–glucose deprivation (OGD) in septal neuronal cultures to test for possible roles of cell cycle proteins in neuronal survival. Increased cdc2-immunoreactive neurons were observed at 24 h after the end of 5 h OGD. Green fluorescent protein (GFP) or GFP along with a wild type or dominant negative form of the retinoblastoma protein (Rb), or cyclin-dependent kinase5 (Cdk5), were overexpressed using plasmid constructs. Following OGD, when compared to controls, neurons expressing both GFP and dominant negative Rb, RbΔK11, showed significantly less damage using microscopy imaging. Overexpression of Rb-wt did not affect survival. Surprisingly, overexpression of Cdk5-wild type significantly protected neurons from process disintegration but Cdk5T33, a dominant negative Cdk5, gave little or no protection. Thus phosphorylation of the cell cycle regulator, Rb, contributes to death in OGD in septal neurons but Cdk5 can have a protective role.




Similar content being viewed by others
References
Heintz N (1993) Cell death and the cell cycle: a relationship between transformation and neurodegeneration? Trends Biochem Sci 18:157–159
Li Y, Chopp M, Powers C (1997) Apoptosis and protein expression after focal cerebral ishemia in rat. Brain Res 765:301–312
Osuga H, Sachiko O, Wang F et al (2000) Cyclin-dependent kinases as a therapeutic target for stroke. Proc Natl Acad Sci 97:10254–10259
Katchanov J, Harms C, Gertz K et al (2001) Mild cerebral ischemia induces loss of cyclin-dependent kinase inhibitors and activation of cell cycle machinery before delayed neuronal cell death. J Neurosci 21:5054–5053
Love S (2003) Neuronal expression of cell cycle-related proteins after brain ischaemia in man. Neurosci Lett 353:29–32
Jin K, Nagayama T, Chen J et al (1999) Molecular cloning of a cell cycle regulation gene cyclin H from ischemic rat brain: expression in neurons after global cerebral ischemia. J Nerochem 73:1598–1608
Van Lookeren Campagne M, Gill R (1998) Increased expression of cyclin G1 and p21WAF1/CIP1 in neurons following transient forebrain ischemia: comparison with early DNA damage. J Neurosci Res 53:279–296
O’Hare M, Wang F, Park DS (2002) Cyclin-dependent kinases as potential targets to improve stroke outcome. Pharmacol Ther 93:135–143
Liu DX, Greene LA (2001) Neuronal apoptosis at the G1/S cell cycle checkpoint. Cell Tissue Res 305:217–228
Lee EY, Hu N, Yuan SS et al (1994) Dual roles of the retinoblastoma protein in cell cycle regulation and neuron differentiation. Genes Dev 8:2008–2021
Kaelin WG (1999) Functions of the retinoblastoma protein. Bioessays 21:950–958
Zhu L, van den Heuvel S, Helin K et al (1993) Inhibition of cell proliferation by p107, a relative of the retiboblastoma protein. Genes Dev 7:1111–1125
Wang J, Liu S, Fu Y et al (2003) Cdk5 activation induces hippocampal CA1 cell death by directly phopshorylating NMDA receptors. Nat Neurosci 6:1039–1047
Weishaupt JH, Kussmaul L, Grotsch P et al (2003a) Inhibition of Cdk5 is protective in necrotic and apoptotic paradigms of neuronal cell death and prevents mitochondrial dysfunction. Mol Cell Neurosci 24:489–502
Weishaupt JH, Neusch C, Bahr M (2003b) Cyclin-dependent kinase 5 (cdk5) and neuronal cell death. Cell Tissue Res 312:1–8
Hamdane M, Bretteville A, Sambo A-V et al (2005) p25/Cdk5-mediated retinoblastoma phosphorylation is an early event in neuronal cell death. J Cell Sci 118:1291–1298
Tsai LH, Takahashi T, Caviness VS Jr et al (1993) Activity and expression pattern of cyclin-dependent kinase 5 in the embryonic mouse nervous system. Development 119:1029–1040
Lew J, Huang QQ, Qi Z (1994) A brainspecific activator of cyclin-dependent kinase 5. Nature 371:423–426
Zheng M, Leung CL, Liem RK (1998) Region-specific expression of cyclin dependent kinase 5 (cdk5) and its activators, p35 and p39, in the developing and adult rat central nervous system. J Neurobiol 35:141–159
Ohshima T, Ogawa M, Veeranna et al (2001) Synergistic contributions of cyclin-dependent kinase 5/p35 and Reelin/Dab1 to the positioning of cortical neurons in the developing mouse brain. Proc Natl Acad Sci 98:2764–2769
Gilmore EC, Ohshima T, Goffinet AM et al (1998) Cyclin dependent kinase 5-deficient mice demonstrate novel developmental arrest in cerebral cortex. J Neurosci 18:6370–6377
Hayashi T, Warita H, Abe K et al (1999) Expression of cyclin-dependent kinase 5 and its activator p35 in rat brain after middle cerebral artery occlusion. Neurosci Lett 265:37–40
Patzke H, Tsai LH (2002) Calpain-mediated cleavage of the cyclin-dependent kinase5 activator p39 to p29. J Biol Chem 277:8054–8060
Crain BJ, Westerkam WD, Harrison AH et al (1988) Selective neuronal death after transient forebrain ischemia in the mongolian gerbil: a silver impregnation study. Neuroscience 272:387–402
Nonner D, Barrett EF, Barrett JN (1996) Neurotrophin effects on survival and expression of cholinergic properties in cultured rat septal neurons under normal and stress conditions. J Neurosci 16:6665–6675
Kaufman L, Barrett JN (1983) Serum factor supporting long term survival of rat central neurons in culture. Science 220:1394–1396
Yan J, Barrett JN (1998) Purification from bovine serum of a survival-promoting factor for cultured central neurons and its identification as selenoprotein-P. J Neurosci 18:8682–8691
White MG, Emery M, Nonner D et al (2003) Caspase activation contributes to delayed death of heat-stressed striatal neurons. J Neurochem 87:958–68
Gao C, Negash S, Wang HS et al (2001) Cdk5 mediates changes in morphology and promotes apoptosis of astrocytoma cells in response to heat shock. J Cell Sci 114:1145–1153
Jiang Z, Zacksenhaus E (2002) Activation of retinoblastoma protein in mammary gland leads to ductal growth suppression, precocious differentiation, and adenocarcinoma. J Cell Biol 56:185–198
Nikolic M, Dudek H, Kwon YT et al (1996) The cdk5/p35 kinase is essential for neurite outgrowth during neuronal differentiation. Genes Dev 10:816–825
Panickar KS, Nonner D, Barrett JN (2005) Overexpression of Bcl-xl protects septal neurons from prolonged hypoglycemia and from acute ischemia-like stress. Neuroscience 135:73–80
Nonner D, Barrett EF, Kaplan P et al (2001) Bone morphogenetic proteins, BMP6 and BMP7, enhance the protective effect of neurotrophins on cultured septal cholinergic neurons during hypoglycemia. J Neurochem 77:691–699
Knockaert M, Greengard P, Meijer L (2002) Pharmacological inhibitors of cyclin dependent kinases. Trends Pharmacol Sci 23:417–425
Lee KY, Helbing CC, Choi KS et al (1997) Neuronal cdc2 like kinase (Nclk) binds and phosphorylates the retinoblastoma protein. J Biol Chem 272:5622–5626
Wen Y, Yang S, Liu R et al (2005) Cell-cycle regulators are involved in transient cerebral ischemia-induced neuronal apoptosis in female rats. FEBS Lett 579:4591–4599
Small DL, Monette R, Fournier MC et al (2001) Characterization of cyclin D1 expression in a rat global model of cerebral ischemia. Brain Res 900:26–37
Hayashi T, Sakai K, Sasaki C et al (2000) Phosphorylation of retinoblastoma protein in rat brain after transient middle cerebral artery occlusion. Neuropathol Appl Neurobiol 26:390–397
Guegan C, Levy V, David JP et al (1997) c-Jun and cyclin D1 proteins as mediators of neuronal death after a focal ischemic insult. Neuroreport 8:1003–1007
McManus JP, Koch CJ, Jian M (1999) Decreased brain infarct following focal ischemia in mice lacking the transcription factor E2F1. Neuroreport 10:2711–2714
Smith DS, Tsai LH (2002) Cdk5 behind the wheel: a role in trafficking and transport? Trends Cell Biol 12:28–36
Green SL, Kulp KS, Vulliet R (1997) Cyclin-dependent protein kinase 5 activity increases in rat brain following ischemia. Neurochem Int 31:617–623
Nath R, Davis M, Probert AW et al (2000) Processing of a Cdk5 activator p35 to its truncated form (p25) by calpain in acutely injured neuronal cells. Biochem Biophys Res Commun 274:16–21
Cruz JC, Tsai LH (2004) A Jekyll and Hyde kinase: roles for Cdk5 in brain development and disease. Curr Opin Neurobiol 14:390–394
O’Hare MJ, Kushwaha N, Zhang Y et al (2005) Differential roles of nuclear and cytoplasmic cyclin-dependent kinase 5 in apoptotic and excitotoxic neuronal death. J Neurosci 25:8954–8966
Patrick GN, Zhou P, Kwon YT et al (1998) p35, the neuronal specific activator of cyclin-dependent kinase 5 (Cdk5) is degraded by the ubiquitinproteasome pathway. J Biol Chem 273:24057–24064
Kusakawa G, Saito T, Onuki R et al (2000) Calpaindependent proteolytic cleavage of the p35 cyclin-dependent kinase 5 activator to p25. J Biol Chem 275:17166–17172
Saito T, Onuki R, Fujita Y et al (2003) Developmental regulation of the proteolysis of the p35 cyclin dependent kinase 5 activator by phosphorylation. J Neurosci 23:1189–1197
Vartiainen N, Keksa-Goldsteine V, Goldsteins G et al (2002) Aspirin provides cyclin-dependent kinase 5-dependent protection against subsequent hypoxia/reoxygenation damage in culture. J Neurochem 82:329–335
Sharma P, Veeranna, Sharma M et al (2002) Phosphorylation of MEK1 by cdk5/p35 down-regulates the mitogen-activated protein kinase pathway. J Biol Chem 277:528–534
Li BS, Zhang L, Takahashi S (2002) Cyclindependent kinase 5 prevents neuronal apoptosis by negative regulation of c-Jun N-terminal kinase 3. EMBO J 21:324–333
Li BS, Ma W, Jaffe H et al (2003) Cdk5 is involved in neuregulin-dependent activation of phosphatidylinositol 3-kinase and Akt activity mediating neuronal survival. J Biol Chem 278(37):35702–35709
Zhang S, Edelmann L, Liu J et al (2008) Cdk5 regulates the phosphorylation of tyrosine 1472 NR2B and the surface expression of NMDA receptors. J Neurosci 28:415–424
Acknowledgements
This work was supported by NIH grant NS 12207 and a grant from the Florida Heart Association to John Barrett.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panickar, K.S., Nonner, D., White, M.G. et al. Overexpression of Cdk5 or Non-phosphorylatable Retinoblastoma Protein Protects Septal Neurons from Oxygen–Glucose Deprivation. Neurochem Res 33, 1852–1858 (2008). https://doi.org/10.1007/s11064-008-9647-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9647-3