Abstract
Purpose
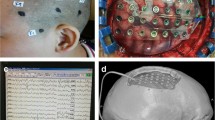
Awake craniotomy with intraoperative functional brain mapping (FBM) bedside neurological testing is an important technique used to optimize resective brain surgeries near eloquent cortex. Awake craniotomy performed with electrocorticography (ECoG) and direct electrical stimulation (DES) for FBM can delineate eloquent cortex from lesions and epileptogenic regions. However, current electrode technology demonstrates spatial limitations. Our group has developed a novel circular grid with the goal of improving spatial recording of ECoG to enhance detection of ictal and interictal activity.
Methods
This retrospective study was approved by the institutional review board at Mayo Clinic Florida. We analyzed patients undergoing awake craniotomy with ECoG and DES and compared ECoG data obtained using the 22 contact circular grid to standard 6 contact strip electrode.
Results
We included 144 cases of awake craniotomy with ECoG, 73 using circular grid and 71 with strip electrode. No significant differences were seen regarding preoperative clinical and demographic data, duration of ECoG recording (p = 0.676) and use of DES (p = 0.926). Circular grid was more sensitive in detecting periodic focal epileptiform discharges (PFEDs) (p = 0.004), PFEDs plus (p = 0.032), afterdischarges (ADs) per case (p = 0.022) at lower minimum (p = 0.012) and maximum (p < 0.0012) intensity stimulation, and seizures (p = 0.048). PFEDs (p < 0.001), PFEDs plus (p < 0.001), and HFOs (p < 0.001) but not ADs (p = 0.255) predicted electrographic seizures.
Conclusion
We demonstrate higher sensitivity in detecting ictal and interictal activity on ECoG during awake craniotomy with a novel circular grid compared to strip electrode, likely due to better spatial sampling during ECoG. We also found association between PFEDs and intraoperative seizures.



Similar content being viewed by others
References
De Benedictis A, Moritz-Gasser S, Duffau H (2010) Awake mapping optimizes the extent of resection for low-grade gliomas in eloquent areas. Neurosurgery 66(6):1074–1084. https://doi.org/10.1227/01.NEU.0000369514.74284.78
Freund BE, Sherman WJ, Sabsevitz DS, et al (2023) Can we improve electrocorticography using a circular grid array in brain tumor surgery? Biomed Phys Eng Express 9(6). https://doi.org/10.1088/2057-1976/ad05dd
Mamani R, Jacobo JA, Mejia S, Nuñez-Velasco S, Aragon-Arreola J, Moreno S (2020) Analysis of intraoperative seizures during bipolar brain mapping in eloquent areas: intraoperative seizures in brain mapping. Clin Neurol Neurosurg. https://doi.org/10.1016/j.clineuro.2020.106304
Nossek E, Matot I, Shahar T et al (2013) Intraoperative seizures during awake craniotomy: incidence and consequences: analysis of 477 patients. Neurosurgery 73(1):135–140. https://doi.org/10.1227/01.neu.0000429847.91707.97
Robertson FC, Ullrich NJ, Al-sayegh H, Goumnerova LC (2018) The impact of intraoperative electrocorticography on seizure outcome after resection of pediatric brain tumors: a cohort study. Neurosurgery 85:375–383. https://doi.org/10.1093/neuros/nyy342
Englot DJ, Berger MS, Barbaro NM, Chang EF (2011) Predictors of seizure freedom after resection of supratentorial low-grade gliomas. J Neurosurg 115(2):240–244. https://doi.org/10.3171/2011.3.JNS1153
Goel K, Pek V, Shlobin NA et al (2022) Clinical utility of intraoperative electrocorticography for epilepsy surgery: a systematic review and meta-analysis. Epilepsia. https://doi.org/10.1111/epi.17472
ReFaey K, Chaichana KL, Feyissa AM et al (2020) A 360° electronic device for recording high-resolution intraoperative electrocorticography of the brain during awake craniotomy. J Neurosurg 133(2):443–450. https://doi.org/10.3171/2019.4.JNS19261
Feyissa AM, Worrell GA, Tatum WO et al (2019) Potential influence of IDH1 mutation and MGMT gene promoter methylation on glioma-related preoperative seizures and postoperative seizure control. Seizure 69(April):283–289. https://doi.org/10.1016/j.seizure.2019.05.018
Domingo RA, Vivas-Buitrago T, De Biase G et al (2021) Intraoperative seizure detection during active resection of glioblastoma through a novel hollow circular electrocorticography array. Oper Neurosurg 21(2):E147–E152. https://doi.org/10.1093/ons/opab110
Tatum WO, McKay JH, ReFaey K et al (2020) Detection of after-discharges during intraoperative functional brain mapping in awake brain tumor surgery using a novel high-density circular grid. Clin Neurophysiol 131(4):828–835. https://doi.org/10.1016/j.clinph.2019.12.416
Tatum WO, Feyissa AM, ReFaey K et al (2019) Periodic focal epileptiform discharges. Clin Neurophysiol 130(8):1320–1328. https://doi.org/10.1016/j.clinph.2019.04.718
van Klink NEC, Zweiphenning WJEM, Ferrier CH et al (2021) Can we use intraoperative high-frequency oscillations to guide tumor-related epilepsy surgery? Epilepsia 62(4):997–1004. https://doi.org/10.1111/epi.16845
Abecassis ZA, Ayer AB, Templer JW, Yerneni K, Murthy NK, Tate MC (2021) Analysis of risk factors and clinical sequelae of direct electrical cortical stimulation–induced seizures and afterdischarges in patients undergoing awake mapping. J Neurosurg 134(5):1610–1617. https://doi.org/10.3171/2020.3.JNS193231
Feyissa AM, Worrell GA, Tatum WO et al (2018) High-frequency oscillations in awake patients undergoing brain tumor-related epilepsy surgery. Neurology 90(13):e1119–e1125. https://doi.org/10.1212/WNL.0000000000005216
Freund B, Gugger JJ, Reynolds A, Tatum WO, Claassen J, Kaplan PW (2018) Clinical and electrographic correlates of bilateral independent periodic discharges. J Clin Neurophysiol 35(3):234. https://doi.org/10.1097/WNP.0000000000000472
Guragain H, Cimbalnik J, Stead M et al (2018) Spatial variation in high-frequency oscillation rates and amplitudes in intracranial EEG. Neurology 90(8):E639–E646. https://doi.org/10.1212/WNL.0000000000004998
Eseonu CI, Rincon-torroella J, Refaey K et al (2017) Awake craniotomy vs craniotomy under general anesthesia for perirolandic gliomas: evaluating perioperative complications and extent of resection. Neurosurgery 81(3):481–490. https://doi.org/10.1093/neuros/nyx023
Freund BE, Feyissa AM, Khan A, et al (2023) Early postoperative seizures following awake craniotomy and functional brain mapping for lesionectomy. World Neurosurg. https://doi.org/10.1016/j.wneu.2023.10.119
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work: BEF, AF, and WOT Drafted the work or revised it critically for important intellectual content: All authors. Approved the version to be published: All authors. Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: All authors.
Corresponding author
Ethics declarations
Competing interests
Dr. Tatum is the designer of the circular grid and Dr. Quinones-Hinojosa is one of the co-developers (Patent Application PCT/US2018/039956).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Freund, B.E., Feyissa, A.M., Khan, A. et al. Enhanced sensitivity of electrocorticography during awake craniotomy using a novel circular grid electrode. J Neurooncol 165, 313–320 (2023). https://doi.org/10.1007/s11060-023-04495-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04495-0