Abstract
Introduction
At least half of children with low-grade glioma (LGG) treated with first line chemotherapy experience a relapse/progression and may therefore need a second-line chemotherapy. Irinotecan-bevacizumab has been recommended in this setting in France after encouraging results of pilot studies. We performed a retrospective analysis to define the efficacy, toxicity and predictors for response to the combination on a larger cohort.
Methods
We reviewed the files from children < 19 years of age with progressive or refractory LGG treated between 2009 and 2016 in 7 French centers with this combination.
Results
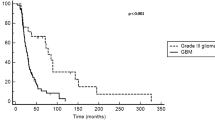
72 patients (median age 7.8 years [range 1–19]) received a median of 16 courses (range 3–30). The median duration of treatment was 9 months (range 1.4–16.2). 96% of patients experienced at least disease stabilization. The 6-month and 2-year progression-free survivals (PFS) were 91.7% [IC 95% 85.5–98.3] and 38.2% [IC 95% 28.2–51.8] respectively. No progression occurred after treatment in 18 patients with a median follow-up of 35.6 months (range 7.6–75.9 months). Younger patients had a worse PFS (p = 0.005). Prior chemoresistance, NF1 status, duration of treatment, histopathology or radiologic response did not predict response. The most frequent toxicities related to bevacizumab included grades 1–2 proteinuria in 21, epistaxis in 10, fatigue in 12 and hypertension in 8 while gastro-intestinal toxicity was the most frequent side effect related to irinotecan.
Conclusions
Bevacizumab-irinotecan has the potential of disease control clinically and radiographically in children with recurrent LGG whatever their previous characteristics; in many cases however these responses are not sustained, especially in younger children.



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- m:
-
Months
- y:
-
Years
- ChemoR:
-
Chemoresistant
- ChemoS:
-
Chemosensitive
- MR:
-
Minor response
- PR:
-
Partial response
- SD:
-
Stable disease
- PD:
-
Progressive disease
- PA/PMA:
-
Pilocytic astrocytoma/pilomyxoid astrocytoma
- GG:
-
Ganglioglioma
- WT:
-
Wild type
- UNDETM:
-
Undetermined
References
Ostrom QT, Gittleman H, Fulop J et al (2015) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2008–2012. Neuro-oncology 17(Suppl 4):iv1–iv62. https://doi.org/10.1093/neuonc/nov189
Gajjar A, Sanford RA, Heideman R et al (1997) Low-grade astrocytoma: a decade of experience at St. Jude Children’s Research Hospital. J Clin Oncol 15:2792–2799
Bowers DC, Gargan L, Kapur P et al (2003) Study of the MIB-1 labeling index as a predictor of tumor progression in pilocytic astrocytomas in children and adolescents. J Clin Oncol 21:2968–2973. https://doi.org/10.1200/JCO.2003.01.017
Merchant TE, Kun LE, Wu S et al (2009) Phase II trial of conformal radiation therapy for pediatric low-grade glioma. J Clin Oncol 27:3598–3604. https://doi.org/10.1200/JCO.2008.20.9494
Massimino M, Spreafico F, Cefalo G et al (2002) High response rate to cisplatin/etoposide regimen in childhood low-grade glioma. J Clin Oncol 20:4209–4216
Gururangan S, Cavazos CM, Ashley D et al (2002) Phase II study of carboplatin in children with progressive low-grade gliomas. J Clin Oncol 20:2951–2958
Bouffet E, Jakacki R, Goldman S et al (2012) Phase II study of weekly vinblastine in recurrent or refractory pediatric low-grade glioma. J Clin Oncol 30:1358–1363. https://doi.org/10.1200/JCO.2011.34.5843
Laithier V, Grill J, Le Deley M-C et al (2003) Progression-free survival in children with optic pathway tumors: dependence on age and the quality of the response to chemotherapy–results of the first French prospective study for the French Society of Pediatric Oncology. J Clin Oncol 21:4572–4578. https://doi.org/10.1200/JCO.2003.03.043
Ater JL, Zhou T, Holmes E et al (2012) Randomized study of two chemotherapy regimens for treatment of low-grade glioma in young children: a report from the Children’s Oncology Group. J Clin Oncol 30:2641–2647. https://doi.org/10.1200/JCO.2011.36.6054
Gnekow AK, Walker DA, Kandels D et al (2017) A European randomised controlled trial of the addition of etoposide to standard vincristine and carboplatin induction as part of an 18-month treatment programme for childhood (≤ 16 years) low grade glioma—a final report. Eur J Cancer 81:206–225. https://doi.org/10.1016/j.ejca.2017.04.019
de Haas V, Grill J, Raquin M-A et al (2009) Relapses of optic pathway tumors after first-line chemotherapy. Pediatr Blood Cancer 52:575–580. https://doi.org/10.1002/pbc.21911
Bartels U, Hawkins C, Jing M et al (2006) Vascularity and angiogenesis as predictors of growth in optic pathway/hypothalamic gliomas. J Neurosurg 104:314–320. https://doi.org/10.3171/ped.2006.104.5.314
Ferrara N, Gerber H-P, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676. https://doi.org/10.1038/nm0603-669
Jain RK (2008) Lessons from multidisciplinary translational trials on anti-angiogenic therapy of cancer. Nat Rev Cancer 8:309–316. https://doi.org/10.1038/nrc2346
Gremonprez F, Descamps B, Izmer A et al (2015) Pretreatment with VEGF(R)-inhibitors reduces interstitial fluid pressure, increases intraperitoneal chemotherapy drug penetration, and impedes tumor growth in a mouse colorectal carcinomatosis model. Oncotarget 6:29889–29900. https://doi.org/10.18632/oncotarget.5092
Friedman HS, Petros WP, Friedman AH et al (1999) Irinotecan therapy in adults with recurrent or progressive malignant glioma. J Clin Oncol 17:1516–1525
Vredenburgh JJ, Desjardins A, Reardon DA, Friedman HS (2009) Experience with irinotecan for the treatment of malignant glioma. Neurooncology 11:80–91. https://doi.org/10.1215/15228517-2008-075
Sapra P, Kraft P, Pastorino F et al (2011) Potent and sustained inhibition of HIF-1α and downstream genes by a polyethyleneglycol-SN38 conjugate, EZN-2208, results in anti-angiogenic effects. Angiogenesis 14:245–253. https://doi.org/10.1007/s10456-011-9209-1
Hurwitz H, Fehrenbacher L, Novotny W et al (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350:2335–2342. https://doi.org/10.1056/NEJMoa032691
Sandler A, Gray R, Perry MC et al (2006) Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 355:2542–2550. https://doi.org/10.1056/NEJMoa061884
Yang JC, Haworth L, Sherry RM et al (2003) A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N Engl J Med 349:427–434. https://doi.org/10.1056/NEJMoa021491
Perren TJ, Swart AM, Pfisterer J et al (2011) A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med 365:2484–2496. https://doi.org/10.1056/NEJMoa1103799
Chisholm JC, Merks JHM, Casanova M et al (2017) Open-label, multicentre, randomised, phase II study of the EpSSG and the ITCC evaluating the addition of bevacizumab to chemotherapy in childhood and adolescent patients with metastatic soft tissue sarcoma (the BERNIE study). Eur J Cancer 83:177–184. https://doi.org/10.1016/j.ejca.2017.06.015
Grill J, Massimino M, Bouffet E et al (2018) Phase II, open-label, randomized, multicenter trial (HERBY) of Bevacizumab in pediatric patients with newly diagnosed high-grade glioma. J Clin Oncol 36:951–958. https://doi.org/10.1200/JCO.2017.76.0611
Packer RJ, Jakacki R, Horn M et al (2009) Objective response of multiply recurrent low-grade gliomas to bevacizumab and irinotecan. Pediatr Blood Cancer 52:791–795. https://doi.org/10.1002/pbc.21935
Couec M-L, André N, Thebaud E et al (2012) Bevacizumab and irinotecan in children with recurrent or refractory brain tumors: toxicity and efficacy trends. Pediatr Blood Cancer 59:34–38. https://doi.org/10.1002/pbc.24066
Hwang EI, Jakacki RI, Fisher MJ et al (2013) Long-term efficacy and toxicity of bevacizumab-based therapy in children with recurrent low-grade gliomas. Pediatr Blood Cancer 60:776–782. https://doi.org/10.1002/pbc.24297
Sikkema AH, de Bont ESJM, Molema G et al (2011) Vascular endothelial growth factor receptor 2 (VEGFR-2) signalling activity in paediatric pilocytic astrocytoma is restricted to tumour endothelial cells. Neuropathol Appl Neurobiol 37:538–548. https://doi.org/10.1111/j.1365-2990.2011.01160.x
Gururangan S, Fangusaro J, Poussaint TY et al (2014) Efficacy of bevacizumab plus irinotecan in children with recurrent low-grade gliomas–a Pediatric Brain Tumor Consortium study. Neurooncology 16:310–317. https://doi.org/10.1093/neuonc/not154
Gururangan S, Chi SN, Young Poussaint T et al (2010) Lack of efficacy of bevacizumab plus irinotecan in children with recurrent malignant glioma and diffuse brainstem glioma: a Pediatric Brain Tumor Consortium study. J Clin Oncol 28:3069–3075. https://doi.org/10.1200/JCO.2009.26.8789
Fangusaro J, Witt O, Hernáiz Driever P et al (2020) Response assessment in paediatric low-grade glioma: recommendations from the Response Assessment in Pediatric Neuro-Oncology (RAPNO) working group. Lancet Oncol 21:e305–e316. https://doi.org/10.1016/S1470-2045(20)30064-4
Stokland T, Liu J-F, Ironside JW et al (2010) A multivariate analysis of factors determining tumor progression in childhood low-grade glioma: a population-based cohort study (CCLG CNS9702). Neurooncology 12:1257–1268. https://doi.org/10.1093/neuonc/noq092
Lassaletta A, Scheinemann K, Zelcer SM et al (2016) Phase II weekly vinblastine for chemotherapy-naïve children with progressive low-grade glioma: a Canadian Pediatric Brain Tumor Consortium Study. J Clin Oncol 34:3537–3543. https://doi.org/10.1200/JCO.2016.68.1585
Ater JL, Xia C, Mazewski CM et al (2016) Nonrandomized comparison of neurofibromatosis type 1 and non-neurofibromatosis type 1 children who received carboplatin and vincristine for progressive low-grade glioma: a report from the Children’s Oncology Group. Cancer 122:1928–1936. https://doi.org/10.1002/cncr.29987
Jost SC, Ackerman JW, Garbow JR et al (2008) Diffusion-weighted and dynamic contrast-enhanced imaging as markers of clinical behavior in children with optic pathway glioma. Pediatr Radiol 38:1293–1299. https://doi.org/10.1007/s00247-008-1003-x
Maloney E, Stanescu AL, Perez FA et al (2018) Surveillance magnetic resonance imaging for isolated optic pathway gliomas: is gadolinium necessary? Pediatr Radiol 48:1472–1484. https://doi.org/10.1007/s00247-018-4154-4
Gressett SM, Shah SR (2009) Intricacies of bevacizumab-induced toxicities and their management. Ann Pharmacother 43:490–501. https://doi.org/10.1345/aph.1L426
Glade Bender JL, Adamson PC, Reid JM et al (2008) Phase I trial and pharmacokinetic study of bevacizumab in pediatric patients with refractory solid tumors: a Children’s Oncology Group Study. J Clin Oncol 26:399–405. https://doi.org/10.1200/JCO.2007.11.9230
Gorsi HS, Khanna PC, Tumblin M et al (2018) Single-agent bevacizumab in the treatment of recurrent or refractory pediatric low-grade glioma: A single institutional experience. Pediatr Blood Cancer 65:e27234. https://doi.org/10.1002/pbc.27234
Author information
Authors and Affiliations
Contributions
JG, ATE, PV and CdM designed and conceptualized the report, acquired and interpreted patient data, drafted the manuscript for intellectual content, reviewed and revised the manuscript. AC, CP, MPR, KB, SP, NB, DFB, EdC, FB, PL, FF, NA, AIB, TB, CD and DVC acquired and interpreted patient data, reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
No funds, grants, or other support was received. The authors have no relevant financial or non-financial interests to disclose. Dr Grill has received grant support from Hoffmann La Roche for a study with bevacizumab in pediatric high-grade gliomas.
Ethical approval
Ethical approval was waived by the local Ethics Committee of Gustave Roussy (CCTIRS no 14.817) in view of the retrospective nature of the study and all the procedures being performed were part of the routine care.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11060_2022_3970_MOESM4_ESM.tif
Supplementary Figure 6: PFS according to radiological response. (MR: minor response, PR: partial response, SD: stable disease, PD: progressive disease) (TIF 8933 kb)
Rights and permissions
About this article
Cite this article
de Marcellus, C., Tauziède-Espariat, A., Cuinet, A. et al. The role of irinotecan-bevacizumab as rescue regimen in children with low-grade gliomas: a retrospective nationwide study in 72 patients. J Neurooncol 157, 355–364 (2022). https://doi.org/10.1007/s11060-022-03970-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-03970-4