Abstract
Purpose
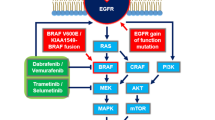
Although uncommon, detection of BRAF V600E mutations in adult patients with glioblastoma has become increasingly relevant given the widespread application of molecular diagnostics and encouraging therapeutic activity of BRAF/MEK inhibitors.
Methods
We performed a retrospective study of adult glioblastoma patients treated at Dana-Farber Cancer Institute/Brigham and Women’s Hospital or Massachusetts General Hospital from January 2011 to July 2019 with an identified BRAF V600E mutation by either immunohistochemistry or molecular testing. Patient characteristics, molecular genomics, and preoperative MRI were analyzed.
Results
Nineteen glioblastoma patients were included, with median age at diagnosis of 41-years-old (range 22–69). Only 1/18 was IDH1/2-mutant; 10/17 had MGMT unmethylated tumors. The most common additional molecular alterations were CDKN2A/2B biallelic loss/loss-of-function (10/13, 76.9%), polysomy 7 (8/12, 66.7%), monosomy 10 (5/12, 41.7%), PTEN biallelic loss/loss-of-function (5/13, 38.5%) and TERT promoter mutations (5/15, 33.3%). Most tumors were well-circumscribed (11/14) and all were contrast-enhancing on MRI. Twelve patients eventually developed subependymal or leptomeningeal dissemination. Six patients were treated with BRAF/MEK inhibition following disease progression after standard of care therapy, with 4/6 patients showing partial response or stable disease as best response. Median time to progression after BRAF/MEK inhibition was 6.0 months (95% CI 1.2–11.8). Grade 1 skin rash was present in 2 patients, but no other adverse events were reported. Median OS for the entire cohort was 24.1 months (95% CI 15.7–38.9).
Conclusion
Understanding the natural history and features of BRAF V600E glioblastoma may help better identify patients for BRAF/MEK inhibition and select therapeutic strategies.



Similar content being viewed by others
References
Ostrom QT, Patil N, Cioffi G, Waite K, Kruchko C, Barnholtz-Sloan JS (2020) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2013–2017. Neuro Oncol 22:iv. https://doi.org/10.1093/neuonc/noaa200
Wen PY, Weller M, Lee EQ et al (2020) Glioblastoma in adults: a Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro Oncol 22(8):1073–1113. https://doi.org/10.1093/neuonc/noaa106
Lamba N, Chukwueke UN, Smith TR et al (2020) Socioeconomic disparities associated with MGMT promoter methylation testing for patients with glioblastoma. JAMA Oncol 6(12):1972–1974. https://doi.org/10.1001/jamaoncol.2020.4937
Brennan CW, Verhaak RGW, McKenna A et al (2013) The somatic genomic landscape of glioblastoma. Cell 155(2):462. https://doi.org/10.1016/j.cell.2013.09.034
Kleinschmidt-Demasters BK, Aisner DL, Birks DK, Foreman NK (2013) Epithelioid GBMs show a high percentage of BRAF V600E mutation. Am J Surg Pathol 37(5):685–698. https://doi.org/10.1097/PAS.0b013e31827f9c5e
Long GV, Stroyakovskiy D, Gogas H et al (2014) Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med 371(20):1877–1888. https://doi.org/10.1056/NEJMoa1406037
Johanns TM, Ferguson CJ, Grierson PM, Dahiya S, Ansstas G (2018) Rapid clinical and radiographic response with combined dabrafenib and trametinib in adults with BRAF-mutated high-grade glioma. JNCCN J Natl Compr Cancer Netw 16(1):4–10. https://doi.org/10.6004/jnccn.2017.7032
Schreck KC, Guajardo A, Lin DDM, Eberhart CG, Grossman SA (2018) Concurrent BRAF/MEK inhibitors in BRAF V600-Mutant high-grade primary brain tumors. JNCCN J Natl Compr Cancer Netw 16(4):343–347. https://doi.org/10.6004/jnccn.2017.7052
Dahiya S, Emnett RJ, Haydon DH et al (2014) BRAF-V600E mutation in pediatric and adult glioblastoma. Neuro Oncol 16(2):318–319. https://doi.org/10.1093/neuonc/not146
Andersen BM, Miranda C, Hatzoglou V, Deangelis LM, Miller AM (2019) Leptomeningeal metastases in glioma: the Memorial Sloan Kettering Cancer Center experience. Neurology 92(21):e2483–e2491. https://doi.org/10.1212/WNL.0000000000007529
Vertosick FT, Selker RG (1990) Brain stem and spinal metastases of supratentorial glioblastoma multiforme. Neurosurgery 27(4):516. https://doi.org/10.1097/00006123-199010000-00002
Dias-Santagata D, Akhavanfard S, David SS et al (2010) Rapid targeted mutational analysis of human tumours: a clinical platform to guide personalized cancer medicine. EMBO Mol Med 2(5):146–158. https://doi.org/10.1002/emmm.201000070
Garcia EP, Minkovsky A, Jia Y et al (2017) Validation of oncopanel a targeted next-generation sequencing assay for the detection of somatic variants in cancer. Arch Pathol Lab Med 141(6):751–758. https://doi.org/10.5858/arpa.2016-0527-OA
Brat DJ, Aldape K, Colman H et al (2020) cIMPACT-NOW update 5: recommended grading criteria and terminologies for IDH-mutant astrocytomas. Acta Neuropathol 139(3):603–608. https://doi.org/10.1007/s00401-020-02127-9
Behling F, Barrantes-Freer A, Skardelly M et al (2016) Frequency of BRAF V600E mutations in 969 central nervous system neoplasms. Diagn Pathol. https://doi.org/10.1186/s13000-016-0506-2
Ryall S, Zapotocky M, Fukuoka K et al (2020) Integrated molecular and clinical analysis of 1,000 pediatric low-grade gliomas. Cancer Cell 37(4):569-583.e5. https://doi.org/10.1016/j.ccell.2020.03.011
Korshunov A, Chavez L, Sharma T et al (2018) Epithelioid glioblastomas stratify into established diagnostic subsets upon integrated molecular analysis. Brain Pathol 28(5):656–662. https://doi.org/10.1111/bpa.12566
Lu VM, George ND, Brown DA et al (2019) Confirming diagnosis and effective treatment for rare epithelioid glioblastoma variant: an integrated survival analysis of the literature. World Neuroncol. https://doi.org/10.1016/j.wneu.2019.08.007
Mistry M, Zhukova N, Merico D et al (2015) BRAF mutation and CDKN2A deletion define a clinically distinct subgroup of childhood secondary high-grade glioma. J Clin Oncol 9:1015–1022. https://doi.org/10.1200/JCO.2014.58.3922
Ellison DW, Hawkins C, Jones DTW et al (2019) cIMPACT-NOW update 4: diffuse gliomas characterized by MYB, MYBL1, or FGFR1 alterations or BRAFV600E mutation. Acta Neuropathol 137(4):683–687. https://doi.org/10.1007/s00401-019-01987-0
Ryall S, Zapotocky M, Fukuoka K, Ellison DW, Tabori U, Hawkins Correspondence C (2020) Integrated molecular and clinical analysis of 1,000 pediatric low-grade gliomas. Cancer Cell. https://doi.org/10.1016/j.ccell.2020.03.011
Broniscer A, Tatevossian RG, Sabin ND et al (2014) Clinical, radiological, histological and molecular characteristics of paediatric epithelioid glioblastoma. Neuropathol Appl Neurobiol 40(3):327–336. https://doi.org/10.1111/nan.12093
Arita N, Taneda M, Hayakawa T (1994) Leptomeningeal dissemination of malignant gliomas. Incidence, diagnosis and outcome. Acta Neurochir (Wien) 126(24):84–92. https://doi.org/10.1007/BF01476415
Burger MC, Ronellenfitsch MW, Lorenz NI et al (2017) Dabrafenib in patients with recurrent, BRAF V600E mutated malignant glioma and leptomeningeal disease. Oncol Rep 38(6):3291–3296. https://doi.org/10.3892/or.2017.6013
Hauschild A, Grob JJ, Demidov LV et al (2012) Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet 380(9839):358–365. https://doi.org/10.1016/S0140-6736(12)60868-X
Robert C, Grob JJ, Stroyakovskiy D et al (2019) Five-year outcomes with dabrafenib plus trametinib in metastatic melanoma. N Engl J Med 381(7):626–636. https://doi.org/10.1056/NEJMoa1904059
Kaley T, Touat M, Subbiah V et al (2018) BRAF inhibition in BRAFV600-mutant gliomas: results from the VE-BASKET study. J Clin Oncol 36:3477–3484. https://doi.org/10.1200/JCO.2018.78.9990
Kushnirsky M, Feun LG, Gultekin SH, de la Fuente MI (2020) Prolonged complete response with combined dabrafenib and trametinib after BRAF inhibitor failure in BRAF-mutant glioblastoma. JCO Precis Oncol 4:44–50. https://doi.org/10.1200/po.19.00272
Wen P, Alexander S, Yung-Jue B et al (2018) Rare-09. Efficacy and safety of dabrafenib + trametinib in patients with recurrent/refractory braf v600e–mutated high-grade gliomA (HGG). Neuro Oncol 20:238. https://doi.org/10.1093/neuonc/noy148.986
Funding
No funding was received in support of this study.
Author information
Authors and Affiliations
Contributions
MJL-F, KWS and IA-R contributed to the experimental design, implementation, analysis and interpretation of data, including statistical analysis, writing and revision of the manuscript; JBI and BMA, DAF, ERG, JTJ, DAR and PYW contributed to the analysis and interpretation of data, writing and revision the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
M.J. Lim-Fat; K.W. Song; B.M. Andersen; D.A. Forst; E.R. Gerstner and I. Arrillaga-Romany report no disclosures relevant to the manuscript; J.B. Iorgulescu received support from the NIH (NCI K12CA090354) and Conquer Cancer Foundation/Sontag Foundation Young Investigator Award; J.T. Jordan received honoraria from Elsevier and Consulting for Navio Theragnostics, Recursion Pharmaceuticals, CereXis, Inc, and Health2047, Inc; D.A. Reardon received Research support (paid to DFCI) fromAcerta Phamaceuticals; Agenus; Celldex; EMD Serono; Incyte; Inovio; Midatech; Omniox; Tragara, advisory/consultation (paid to Dr. Reardon): Abbvie; Advantagene; Agenus; Amgen; Bayer; Bristol-Myers Squibb; Celldex; DelMar; EMD Serono; Genentech/Roche; Imvax; Inovio; Merck; Merck KGaA; Monteris; Novocure; Oncorus; Oxigene; Regeneron; Stemline; Taiho Oncology, Inc., Honoraria (paid to Dr. Reardon): Abbvie; Advantagene; Agenus; Bristol-Myers Squibb; Celldex; EMD Serono; Genentech/Roche; Imvax; Inovio; Merck; Merck KGaA; Monteris; Novocure; Oncorus; Oxigene; Regeneron; Stemline; Taiho Oncology, Inc; P.Y. Wen received honoraria from Agios, Astra Zeneca/Medimmune, Beigene, Celgene, Eli Lily, Genentech/Roche, Kazia, MediciNova, Merck, Novartis, Oncoceutics, Vascular Biogenics, VBI Vaccines (research support); Agios, Astra Zeneca, Bayer, Blue Earth Diagnostics, CNS Pharmaceuticals, Immunomic Therapeutics, Karyopharm, Kiyatec, Puma, Taiho, Vascular Biogenics, Deciphera, VBI Vaccines, Tocagen, Voyager, QED, Imvax, Elevate Bio (advisory board) and Merck, Prime Oncology (speaker honoraria).
Ethical approval
This study was conducted under a Dana-Farber/Harvard Cancer Center Institutional Review Board protocol. A waiver was granted for informed consent due to its retrospective nature.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (PDF 247 KB)
Representative BRAF V600E immunohistochemistry micrographs of the initial, pre-treatment pathology from 2 GBM patients, which demonstrate variable positive staining (brown) – with (B) 19.8% and (C) 24.3% variant allele frequencies of BRAF V600E on molecular testing. Microvascular proliferation served as internal negative controls. Total magnification 200x.
Rights and permissions
About this article
Cite this article
Lim-Fat, M.J., Song, K.W., Iorgulescu, J.B. et al. Clinical, radiological and genomic features and targeted therapy in BRAF V600E mutant adult glioblastoma. J Neurooncol 152, 515–522 (2021). https://doi.org/10.1007/s11060-021-03719-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-021-03719-5