Abstract
Purpose
Primary central nervous system lymphoma (PCNSL) is a subtype of non-Hodgkin’s lymphoma that involves the brain, spinal cord, or leptomeninges, without evidence of systemic disease. This rare disease accounts for ~ 3% of all primary central nervous system (CNS) tumors. Methotrexate-based regimens are the standard of care for this disease with overall survival rates ranging from 14 to 55 months. Relapse after apparent complete remission can occur. We sought to understand the outcomes of patients who relapsed.
Methods
This is an IRB-approved investigation of patients treated at our institution between 12/31/2004 and 10/12/2016. We retrospectively identified all cases of PCNSL as part of a database registry and evaluated these cases for demographic information, absence or presence of relapse, location of relapse, treatment regimens, and median relapse-free survival.
Results
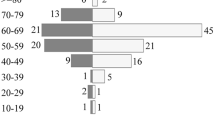
This analysis identified 44 patients with a pathologically confirmed diagnosis of PCNSL. Mean age at diagnosis was 63.1 years (range 20–86, SD = 13.2 years). Of the 44 patients, 28 patients successfully completed an initial treatment regimen without recurrence or toxicity that required a change in therapy. Relapse occurred in 11 patients with the location of relapse being in the CNS only (n = 5), vitreous fluid only (n = 1), outside CNS only (n = 3), or a combination of CNS and outside of the CNS (n = 2). Sites of relapse outside of the CNS included testes (n = 1), lung (n = 1), adrenal gland (n = 1), kidney/adrenal gland (n = 1), and retroperitoneum (n = 1). Median relapse-free survival after successful completion of therapy was 6.7 years (95% CI 1.1, 12.6).
Conclusion
After successful initial treatment, PCNSL has a propensity to relapse, and this relapse can occur both inside and outside of the CNS. Vigilant monitoring of off-treatment patients with a history of PCNSL is necessary to guide early diagnosis of relapse and to initiate aggressive treatment.


Similar content being viewed by others
References
DeAngelis LM, Yahalom J, Thaler HT, Kher U (1992) Combined modality therapy for primary CNS lymphoma. J Clin Oncol 10:635–643. https://doi.org/10.1200/JCO.1992.10.4.635
Villano JL, Koshy M, Shaikh H, Dolecek TA, McCarthy BJ (2011) Age, gender, and racial differences in incidence and survival in primary CNS lymphoma. Br J Cancer 105:1414–1418. https://doi.org/10.1038/bjc.2011.357
Graham MS, DeAngelis LM (2018) Improving outcomes in primary CNS lymphoma. Best Pract Res Clin Haematol 31:262–269. https://doi.org/10.1016/j.beha.2018.07.006
Provencher S, Ferlay C, Alaoui-Slimani K, Devidas A, Lepretre S, de Prijck B, Sebban C, de la Fouchardiere A, Chassagne-Clement C, Ketterer N, Thyss A, Delannoy A, Tilly H, Biron P, Blay JY, Ghesquieres H (2011) Clinical characteristics and outcome of isolated extracerebral relapses of primary central nervous system lymphoma: a case series. Hematol Oncol 29:10–16. https://doi.org/10.1002/hon.944
Abrey L, Yahalom J Relapse and late neurotoxicity in primary central nervous system lymphoma (PCNSL). American Academy of Neurology, Boston, p A18
Nelson DF, Martz KL, Bonner H, Nelson JS, Newall J, Kerman HD, Thomson JW, Murray KJ (1992) Non-Hodgkin's lymphoma of the brain: can high dose, large volume radiation therapy improve survival? Report on a prospective trial by the Radiation Therapy Oncology Group (RTOG): RTOG 8315. Int J Radiat Oncol Biol Phys 23:9–17. https://doi.org/10.1016/0360-3016(92)90538-s
NCCN (2019) Network NCC Central Nervous System Cancers, Version 3.2019. https://www.nccn.org/professionals/physician_gls/recently_updated.aspx
Hoang-Xuan K, Bessell E, Bromberg J, Hottinger AF, Preusser M, Ruda R, Schlegel U, Siegal T, Soussain C, Abacioglu U, Cassoux N, Deckert M, Dirven CM, Ferreri AJ, Graus F, Henriksson R, Herrlinger U, Taphoorn M, Soffietti R, Weller M, European Association for Neuro-Oncology Task Force on Primary CNSL (2015) Diagnosis and treatment of primary CNS lymphoma in immunocompetent patients: guidelines from the European Association for Neuro-Oncology. Lancet Oncol 16:e322–e332. https://doi.org/10.1016/S1470-2045(15)00076-5
Jahnke K, Thiel E, Martus P, Herrlinger U, Weller M, Fischer L, Korfel A, on behalf of the German Primary Central Nervous System Lymphoma Study Group (G-PCNSL-SG) (2006) Relapse of primary central nervous system lymphoma: clinical features, outcome and prognostic factors. J Neurooncol 80:159–165. https://doi.org/10.1007/s11060-006-9165-6
Abrey LE, Batchelor TT, Ferreri AJ, Gospodarowicz M, Pulczynski EJ, Zucca E, Smith JR, Korfel A, Soussain C, DeAngelis LM, Neuwelt EA, O'Neill BP, Thiel E, Shenkier T, Graus F, van den Bent M, Seymour JF, Poortmans P, Armitage JO, Cavalli F, International Primary CNSLCG (2005) Report of an international workshop to standardize baseline evaluation and response criteria for primary CNS lymphoma. J Clin Oncol 23:5034–5043. https://doi.org/10.1200/JCO.2005.13.524
Ambady P, Fu R, Netto JP, Kersch C, Firkins J, Doolittle ND, Neuwelt EA (2017) Patterns of relapse in primary central nervous system lymphoma: inferences regarding the role of the neuro-vascular unit and monoclonal antibodies in treating occult CNS disease. Fluids Barriers CNS 14:16. https://doi.org/10.1186/s12987-017-0064-3
Jahnke K, Hummel M, Korfel A, Burmeister T, Kiewe P, Klasen HA, Muller HH, Stein H, Thiel E (2006) Detection of subclinical systemic disease in primary CNS lymphoma by polymerase chain reaction of the rearranged immunoglobulin heavy-chain genes. J Clin Oncol 24:4754–4757. https://doi.org/10.1200/JCO.2006.06.7165
Yang Y, Czernin J (2011) Contribution of imaging to cancer care costs. J Nucl Med 52(Suppl 2):86S–92S. https://doi.org/10.2967/jnumed.110.085621
Holdhoff M, Ambady P, Abdelaziz A, Sarai G, Bonekamp D, Blakeley J, Grossman SA, Ye X (2014) High-dose methotrexate with or without rituximab in newly diagnosed primary CNS lymphoma. Neurology 83:235–239. https://doi.org/10.1212/WNL.0000000000000593
Schulte-Altedorneburg G, Heuser L, Pels H (2012) MRI patterns in recurrence of primary CNS lymphoma in immunocompetent patients. Eur J Radiol 81:2380–2385. https://doi.org/10.1016/j.ejrad.2011.05.028
Ferreri AJ, Cwynarski K, Pulczynski E, Ponzoni M, Deckert M, Politi LS, Torri V, Fox CP, Rosee PL, Schorb E, Ambrosetti A, Roth A, Hemmaway C, Ferrari A, Linton KM, Ruda R, Binder M, Pukrop T, Balzarotti M, Fabbri A, Johnson P, Gorlov JS, Hess G, Panse J, Pisani F, Tucci A, Stilgenbauer S, Hertenstein B, Keller U, Krause SW, Levis A, Schmoll HJ, Cavalli F, Finke J, Reni M, Zucca E, Illerhaus G, International Extranodal Lymphoma Study G (2016) Chemoimmunotherapy with methotrexate, cytarabine, thiotepa, and rituximab (MATRix regimen) in patients with primary CNS lymphoma: results of the first randomisation of the International Extranodal Lymphoma Study Group-32 (IELSG32) phase 2 trial. Lancet Haematol 3:e217–227. https://doi.org/10.1016/S2352-3026(16)00036-3
Bromberg JEC, Issa S, Bakunina K, Minnema MC, Seute T, Durian M, Cull G, Schouten HC, Stevens WBC, Zijlstra JM, Baars JW, Nijland M, Mason KD, Beeker A, van den Bent MJ, Beijert M, Gonzales M, de Jong D, Doorduijn JK (2019) Rituximab in patients with primary CNS lymphoma (HOVON 105/ALLG NHL 24): a randomised, open-label, phase 3 intergroup study. Lancet Oncol 20:216–228. https://doi.org/10.1016/S1470-2045(18)30747-2
Harjama L, Kuitunen H, Turpeenniemi-Hujanen T, Haapasaari KM, Leppa S, Mannisto S, Karjalainen-Lindsberg ML, Lehtinen T, Eray M, Vornanen M, Haapasalo H, Soini Y, Jantunen E, Nousiainen T, Vasala K, Kuittinen O (2015) Constant pattern of relapse in primary central nervous lymphoma patients treated with high-dose methotrexate combinations. A Finnish retrospective study. Acta Oncol 54:939–943. https://doi.org/10.3109/0284186X.2014.990110
Ambady P, Fu R, Szidonya L, Peereboom DM, Doolittle ND, Neuwelt EA (2020) Impact of maintenance rituximab on duration of response in primary central nervous system lymphoma. J Neurooncol. https://doi.org/10.1007/s11060-020-03411-0
Neuwelt EA, Schiff D (2015) Primary CNS lymphoma: a landmark trial and the next steps. Neurology 84:1194–1195. https://doi.org/10.1212/WNL.0000000000001407
Funding
There was no funding for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
John P. Kirkpatrick reports grants from Varian Medical Systems. He is also an owner in limited partnership for ClearSight RT Products, not related to this work. Annick Desjardins owns stock in Istari Oncology. She has a consulting/advisory role for the following companies: Celgene, Istari Oncology and Orbus Therapeutics. She has received research funding from the following companies: Orbus Therapeutics, Genentech/Roche, Symphogen, and Triphase Accelerator Corp. Dina Randazzo has a consulting/advisory role for the following company: Optune. She has receiving research funding from the following company: Medicenna. Henry S. Friedman owns stock and other ownership interests in 2× Oncology and Istari Oncology. He has received honoraria from and has a consulting/advisory role for Genentech/Roche. He is on the speaker’s bureau for Genentech/Roche and has received travel, accommodations and expenses. David M. Ashley owns stock in Diverse Biotechnology. He has a consulting/advisory role for the following companies: Istari Oncology, Inc and The Jackson Laboratory. He has received research funding from the following companies: Midatech Pharma Plc. Katherine B. Peters has a consulting/advisory role for the following companies: Novocure, Agios, Eisai, Abbvie, Boehringer Ingelheim, and Monteris Medical. She has received research funding from the following companies: Agios, Abbvie, Bristol-Myers Squibb, Monteris Medical, and BioMimetix. Mallika P. Patel, Margaret O. Johnson, Patrick Healy, James E. Herndon II, Eric S. Lipp and Elizabeth S. Miller declared that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Patel, M.P., Kirkpatrick, J.P., Johnson, M.O. et al. Patterns of relapse after successful completion of initial therapy in primary central nervous system lymphoma: a case series. J Neurooncol 147, 477–483 (2020). https://doi.org/10.1007/s11060-020-03446-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-020-03446-3