Abstract
Introduction
Pediatric pilocytic astrocytomas (PAs) are low grade gliomas and the most common brain tumors in children. They often represent a therapeutic challenge when incompletely resected as they can recur and progress despite the use of several lines of chemotherapeutic agents or even radiation therapy. Genetic alterations leading to activation of the mitogen-activated-protein-kinase pathway are a hallmark of this disease and offer an interesting therapeutic alternative through the use of targeted inhibitors.
Methods
Here, we describe six children with sporadic PA who were treated with trametinib, a MEK inhibitor, following progression under conventional therapies. Retrospective chart review was performed.
Results
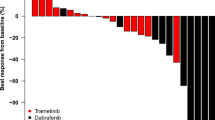
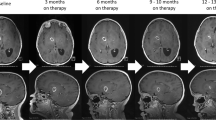
The median age at diagnosis was 2.3 years (y) old [range 11 months (m)–8.5 y old]. KIAA1549-BRAF fusion was identified in five cases, and hotspot FGFR1/NF1/PTPN11 mutations in one. All patients received at least one previous line of chemotherapy (range 1–4). The median time on treatment was 11 m (range 4–20). Overall, we observed two partial responses and three minor responses as best response; three of these patients are still on therapy. Treatment was discontinued in the patient with progressive disease. The most frequent toxicities were minor to moderately severe skin rash and gastro-intestinal symptoms. Two patients had dose reduction due to skin toxicity. Quality of life was excellent with decreased hospital visits and a close to normal life.
Conclusion
Trametinib appears to be a suitable option for refractory pediatric low-grade glioma and warrants further investigations in case of progression.



Similar content being viewed by others
Abbreviations
- Bx:
-
Biopsy
- Carbo–VCR:
-
Carboplatin–vincristine
- Carbo/VP16 IA:
-
Intra-arterial carboplatin and etoposide
- GBM:
-
Glioblastoma
- F:
-
Female
- GTR:
-
Gross total resection
- LND:
-
Lenalidomide
- M:
-
Male
- m:
-
Months
- MR:
-
Minor response
- Oxa:
-
Oxaliplatin
- PA:
-
Pilocytic astrocytoma
- PD:
-
Progressive disease
- PR:
-
Partial response
- STD:
-
Stable disease
- STR:
-
Subtotal resection
- TMZ:
-
Temozolomide
- TPCV:
-
Thioguanine, procarbazine, CCNU, vincristine
- VBL:
-
Vinblastine
- WHO:
-
World Health Organization
- XRT:
-
Radiotherapy
- y:
-
Years
References
Qaddoumi I, Sultan I, Gajjar A (2009) Outcome and prognostic features in pediatric gliomas: a review of 6212 cases from the Surveillance, Epidemiology, and End Results database. Cancer 115:5761–5770
Rickert CH, Paulus W (2001) Epidemiology of central nervous system tumors in childhood and adolescence based on the new WHO classification. Childs Nerv Syst 17:503–511
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
Ater JL, Zhou T, Holmes E, Mazewski CM, Booth TN, Freyer DR, Lazarus KH, Packer RJ, Prados M, Sposto R, Vezina G, Wisoff JH, Pollack IF (2012) Randomized study of two chemotherapy regimens for treatment of low-grade glioma in young children: a report from the Children’s Oncology Group. J Clin Oncol Off J Am Soc Clin Oncol 30:2641–2647. https://doi.org/10.1200/JCO.2011.36.6054
Bouffet E, Jakacki R, Goldman S, Hargrave D, Hawkins C, Shroff M, Hukin J, Bartels U, Foreman N, Kellie S, Hilden J, Etzl M, Wilson B, Stephens D, Tabori U, Baruchel S (2012) Phase II study of weekly vinblastine in recurrent or refractory pediatric low-grade glioma. J Clin Oncol Off J Am Soc Clin Oncol 30:1358–1363. https://doi.org/10.1200/JCO.2011.34.5843
Hwang EI, Jakacki RI, Fisher MJ, Kilburn LB, Horn M, Vezina G, Rood BR, Packer RJ (2013) Long-term efficacy and toxicity of bevacizumab-based therapy in children with recurrent low-grade gliomas. Pediatr Blood Cancer 60:776–782. https://doi.org/10.1002/pbc.24297
Campen CJ, Kranick SM, Kasner SE, Kessler SK, Zimmerman RA, Lustig R, Phillips PC, Storm PB, Smith SE, Ichord R, Fisher MJ (2012) Cranial irradiation increases risk of stroke in pediatric brain tumor survivors. Stroke 43:3035–3040. https://doi.org/10.1161/STROKEAHA.112.661561
Ullrich NJ, Robertson R, Kinnamon DD, Scott RM, Kieran MW, Turner CD, Chi SN, Goumnerova L, Proctor M, Tarbell NJ, Marcus KJ, Pomeroy SL (2007) Moyamoya following cranial irradiation for primary brain tumors in children. Neurology 68:932–938. https://doi.org/10.1212/01.wnl.0000257095.33125.48
Armstrong GT, Conklin HM, Huang S, Srivastava D, Sanford R, Ellison DW, Merchant TE, Hudson MM, Hoehn ME, Robison LL, Gajjar A, Morris EB (2011) Survival and long-term health and cognitive outcomes after low-grade glioma. Neurooncology 13:223–234. https://doi.org/10.1093/neuonc/noq178
Bar EE, Lin A, Tihan T, Burger PC, Eberhart CG (2008) Frequent gains at chromosome 7q34 involving BRAF in pilocytic astrocytoma. J Neuropathol Exp Neurol 67:878–887. https://doi.org/10.1097/NEN.0b013e3181845622
Forshew T, Tatevossian RG, Lawson AR, Ma J, Neale G, Ogunkolade BW, Jones TA, Aarum J, Dalton J, Bailey S, Chaplin T, Carter RL, Gajjar A, Broniscer A, Young BD, Ellison DW, Sheer D (2009) Activation of the ERK/MAPK pathway: a signature genetic defect in posterior fossa pilocytic astrocytomas. J Pathol 218:172–181. https://doi.org/10.1002/path.2558
Jacob K, Albrecht S, Sollier C, Faury D, Sader E, Montpetit A, Serre D, Hauser P, Garami M, Bognar L, Hanzely Z, Montes JL, Atkinson J, Farmer JP, Bouffet E, Hawkins C, Tabori U, Jabado N (2009) Duplication of 7q34 is specific to juvenile pilocytic astrocytomas and a hallmark of cerebellar and optic pathway tumours. Br J Cancer 101:722–733. https://doi.org/10.1038/sj.bjc.6605179
Jones DT, Kocialkowski S, Liu L, Pearson DM, Backlund LM, Ichimura K, Collins VP (2008) Tandem duplication producing a novel oncogenic BRAF fusion gene defines the majority of pilocytic astrocytomas. Cancer Res 68:8673–8677. https://doi.org/10.1158/0008-5472.CAN-08-2097
Jones DT, Kocialkowski S, Liu L, Pearson DM, Ichimura K, Collins VP (2009) Oncogenic RAF1 rearrangement and a novel BRAF mutation as alternatives to KIAA1549:BRAF fusion in activating the MAPK pathway in pilocytic astrocytoma. Oncogene 28:2119–2123
Pfister S, Janzarik WG, Remke M, Ernst A, Werft W, Becker N, Toedt G, Wittmann A, Kratz C, Olbrich H, Ahmadi R, Thieme B, Joos S, Radlwimmer B, Kulozik A, Pietsch T, Herold-Mende C, Gnekow A, Reifenberger G, Korshunov A, Scheurlen W, Omran H, Lichter P (2008) BRAF gene duplication constitutes a mechanism of MAPK pathway activation in low-grade astrocytomas. J Clin Investig 118:1739–1749
Sievert AJ, Jackson EM, Gai X, Hakonarson H, Judkins AR, Resnick AC, Sutton LN, Storm PB, Shaikh TH, Biegel JA (2008) Duplication of 7q34 in pediatric low-grade astrocytomas detected by high-density single-nucleotide polymorphism-based genotype arrays results in a novel BRAF fusion gene. Brain Pathol. https://doi.org/10.1111/j.1750-3639.2008.00225.x
Sharma MK, Zehnbauer BA, Watson MA, Gutmann DH (2005) RAS pathway activation and an oncogenic RAS mutation in sporadic pilocytic astrocytoma. Neurology 65:1335–1336. https://doi.org/10.1212/01.wnl.0000180409.78098.d7
Maltzman TH, Mueller BA, Schroeder J, Rutledge JC, Patterson K, Preston-Martin S, Faustman EM (1997) Ras oncogene mutations in childhood brain tumors. Cancer Epidemiol Biomarkers Prev 6:239–243
Jones DT, Gronych J, Lichter P, Witt O, Pfister SM (2012) MAPK pathway activation in pilocytic astrocytoma. Cell Mol Life Sci 69:1799–1811. https://doi.org/10.1007/s00018-011-0898-9
Yu J, Deshmukh H, Gutmann RJ, Emnett RJ, Rodriguez FJ, Watson MA, Nagarajan R, Gutmann DH (2009) Alterations of BRAF and HIPK2 loci predominate in sporadic pilocytic astrocytoma. Neurology 73:1526–1531. https://doi.org/10.1212/WNL.0b013e3181c0664a
Jones DT, Hutter B, Jager N, Korshunov A, Kool M, Warnatz HJ, Zichner T, Lambert SR, Ryzhova M, Quang DA, Fontebasso AM, Stutz AM, Hutter S, Zuckermann M, Sturm D, Gronych J, Lasitschka B, Schmidt S, Seker-Cin H, Witt H, Sultan M, Ralser M, Northcott PA, Hovestadt V, Bender S, Pfaff E, Stark S, Faury D, Schwartzentruber J, Majewski J, Weber UD, Zapatka M, Raeder B, Schlesner M, Worth CL, Bartholomae CC, von Kalle C, Imbusch CD, Radomski S, Lawerenz C, van Sluis P, Koster J, Volckmann R, Versteeg R, Lehrach H, Monoranu C, Winkler B, Unterberg A, Herold-Mende C, Milde T, Kulozik AE, Ebinger M, Schuhmann MU, Cho YJ, Pomeroy SL, von Deimling A, Witt O, Taylor MD, Wolf S, Karajannis MA, Eberhart CG, Scheurlen W, Hasselblatt M, Ligon KL, Kieran MW, Korbel JO, Yaspo ML, Brors B, Felsberg J, Reifenberger G, Collins VP, Jabado N, Eils R, Lichter P, Pfister SM, International Cancer Genome Consortium PedBrain Tumor P (2013) Recurrent somatic alterations of FGFR1 and NTRK2 in pilocytic astrocytoma. Nat Genet 45:927–932. https://doi.org/10.1038/ng.2682
Eisenhardt AE, Olbrich H, Roring M, Janzarik W, Anh TN, Cin H, Remke M, Witt H, Korshunov A, Pfister SM, Omran H, Brummer T (2011) Functional characterization of a BRAF insertion mutant associated with pilocytic astrocytoma. Int J Cancer 129:2297–2303. https://doi.org/10.1002/ijc.25893
Varan A, Sen H, Aydin B, Yalcin B, Kutluk T, Akyuz C (2016) Neurofibromatosis type 1 and malignancy in childhood. Clin Genet 89:341–345. https://doi.org/10.1111/cge.12625
Miller C, Guillaume D, Dusenbery K, Clark HB, Moertel C (2017) Report of effective trametinib therapy in 2 children with progressive hypothalamic optic pathway pilocytic astrocytoma: documentation of volumetric response. J Neurosurg Pediatr 19:319–324. https://doi.org/10.3171/2016.9.PEDS16328
Banerjee A, Jakacki RI, Onar-Thomas A, Wu S, Nicolaides T, Young Poussaint T, Fangusaro J, Phillips J, Perry A, Turner D, Prados M, Packer RJ, Qaddoumi I, Gururangan S, Pollack IF, Goldman S, Doyle LA, Stewart CF, Boyett JM, Kun LE, Fouladi M (2017) A phase I trial of the MEK inhibitor selumetinib (AZD6244) in pediatric patients with recurrent or refractory low-grade glioma: a Pediatric Brain Tumor Consortium (PBTC) study. Neurooncology 19:1135–1144. https://doi.org/10.1093/neuonc/now282
Drobysheva A, Klesse LJ, Bowers DC, Rajaram V, Rakheja D, Timmons CF, Wang J, Koral K, Gargan L, Ramos E, Park JY (2017) Targeted MAPK pathway inhibitors in patients with disseminated pilocytic astrocytomas. J Natl Compr Cancer Netw 15:978–982. https://doi.org/10.6004/jnccn.2017.0139
Wagner LM, Myseros JS, Lukins DE, Willen CM, Packer RJ (2018) Targeted therapy for infants with diencephalic syndrome: a case report and review of management strategies. Pediatr Blood Cancer. https://doi.org/10.1002/pbc.26917
Geoerger B, Moertel CL, Whitlock J, McCowage GB, Kieran MW, Broniscer A, Hargrave DR, Hingorani P, Kilburn LB, Mueller S, Tseng L, Nebot N, Dasgupta K, Russo MW, Fox E (2018) Phase 1 trial of trametinib alone and in combination with dabrafenib in children and adolescents with relapsed solid tumors or neurofibromatosis type 1 (NF1) progressive plexiform neurofibromas (PN). J Clin Oncol Off J Am Soc Clin Oncol 36
Blumenschein GR Jr, Smit EF, Planchard D, Kim DW, Cadranel J, De Pas T, Dunphy F, Udud K, Ahn MJ, Hanna NH, Kim JH, Mazieres J, Kim SW, Baas P, Rappold E, Redhu S, Puski A, Wu FS, Janne PA (2015) A randomized phase II study of the MEK1/MEK2 inhibitor trametinib (GSK1120212) compared with docetaxel in KRAS-mutant advanced non-small-cell lung cancer (NSCLC)dagger. Ann Oncol 26:894–901. https://doi.org/10.1093/annonc/mdv072
Kim KB, Kefford R, Pavlick AC, Infante JR, Ribas A, Sosman JA, Fecher LA, Millward M, McArthur GA, Hwu P, Gonzalez R, Ott PA, Long GV, Gardner OS, Ouellet D, Xu Y, DeMarini DJ, Le NT, Patel K, Lewis KD (2013) Phase II study of the MEK1/MEK2 inhibitor trametinib in patients with metastatic BRAF-mutant cutaneous melanoma previously treated with or without a BRAF inhibitor. J Clin Oncol Off J Am Soc Clin Oncol 31:482–489. https://doi.org/10.1200/JCO.2012.43.5966
Rizzo D, Ruggiero A, Amato M, Maurizi P, Riccardi R (2016) BRAF and MEK inhibitors in pediatric glioma: new therapeutic strategies, new toxicities. Expert Opin Drug Metab Toxicol 12:1397–1405. https://doi.org/10.1080/17425255.2016.1214710
Boull C, Hook K, Moertel C, Maguiness S (2017) Cutaneous reactions in children treated with the mitogen-activated protein kinase extracellular signal-regulated kinase inhibitor trametinib for neural tumors. Pediatr Dermatol 34:90–94. https://doi.org/10.1111/pde.13038
Dombi E, Baldwin A, Marcus LJ, Fisher MJ, Weiss B, Kim A, Whitcomb P, Martin S, Aschbacher-Smith LE, Rizvi TA, Wu J, Ershler R, Wolters P, Therrien J, Glod J, Belasco JB, Schorry E, Brofferio A, Starosta AJ, Gillespie A, Doyle AL, Ratner N, Widemann BC (2016) Activity of selumetinib in neurofibromatosis type 1-related plexiform neurofibromas. N Engl J Med 375:2550–2560. https://doi.org/10.1056/NEJMoa1605943
Acknowledgements
We would like to acknowledge the generous financial support of the Westmount Old Timers which made this study possible.
Funding
This study was funded by a grant from the Westmount Old Timers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kondyli, M., Larouche, V., Saint-Martin, C. et al. Trametinib for progressive pediatric low-grade gliomas. J Neurooncol 140, 435–444 (2018). https://doi.org/10.1007/s11060-018-2971-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2971-9