Abstract
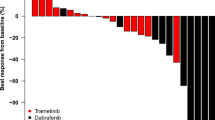
Reports suggest reasonable efficacy and minimal myelosuppression from combination imatinib and hydroxyurea for recurrent malignant glioma. We retrospectively reviewed 16 patients treated with this regimen who were evaluable for toxicity; 14 were also evaluable for response. The incidence of grade 3–4 hematologic toxicity was 25%. The best radiographic response, by Macdonald criteria, was partial response (PR) in three patients (21%), stable disease (SD) in four (29%), and progressive disease (PD) in seven (50%). One patient with a PR developed therapy-limiting hematologic toxicity on day 19 of treatment, progressing to grade 4 on day 64, and persisting until death on day 127 despite discontinuing both drugs. Another patient with PR and two of four patients with SD also developed grade 3 hematologic toxicity. All patients with grade 3–4 hematologic toxicity had disease control (PR or SD) as best radiographic response, whereas none with PD suffered grade 3–4 hematologic toxicity. Combining imatinib with hydroxyurea is effective in some patients with malignant glioma. However, myelosuppression can persist for months after discontinuing the regimen, precluding further chemotherapy. Disease control may also correlate with hematologic toxicity (p = 0.08), suggesting that glioma and marrow stem cells may share a common sensitivity to this chemotherapy regimen.



Similar content being viewed by others
References
Hermanson M, Funa K, Hartman M, Claesson-Welsh L, Heldin CH, Westermark B, Nister M (1992) Platelet-derived growth factor and its receptors in human glioma tissue: expression of messenger RNA and protein suggests the presence of autocrine and paracrine loops. Cancer Res 52:3213–3219
Raymond E, Brandes A, van Oosterom A, Dittrich C, Fumoleau P, Coudert B, Twelves C, de Balincourt C, Lacombe D, van den Bent M (2004) Multicentre phase II study of imatinib mesylate in patients with recurrent glioblastoma: An EORTC: NDDG/BTG Intergroup study [abstract]. J Clin Oncol 22:1501
Wen PY, Yung WK, Lamborn KR, Dahia PL, Wang Y, Peng B, Abrey LE, Raizer J, Cloughesy TF, Fink K, Gilbert M, Chang S, Junck L, Schiff D, Lieberman F, Fine HA, Mehta M, Robins HI, DeAngelis LM, Groves MD, Puduvalli VK, Levin V, Conrad C, Maher EA, Aldape K, Hayes M, Letvak L, Egorin MJ, Capdeville R, Kaplan R, Murgo AJ, Stiles C, Prados MD (2006) Phase I/II study of imatinib mesylate for recurrent malignant gliomas: North American Brain Tumor Consortium Study 99–08. Clin Cancer Res 12:4899–4907
Reardon DA, Egorin MJ, Quinn JA, Rich JN Sr., Gururangan I, Vredenburgh JJ, Desjardins A, Sathornsumetee S, Provenzale JM, Herndon JE 2nd, Dowell JM, Badruddoja MA, McLendon RE, Lagattuta TF, Kicielinski KP, Dresemann G, Sampson JH, Friedman AH, Salvado AJ, Friedman HS (2005) Phase II study of imatinib mesylate plus hydroxyurea in adults with recurrent glioblastoma multiforme. J Clin Oncol 23:9359–9368
Dresemann G (2005) Imatinib and hydroxyurea in pretreated progressive glioblastoma multiforme: a patient series. Ann Oncol 16:1702–1708
Levin VA (1992) The place of hydroxyurea in the treatment of primary brain tumors. Sem Oncol 19:34–39
Brada M, Hoang-Xuan K, Rampling R, Dietrich PY, Dirix LY, Macdonald D, Heimans JJ, Zonnenberg BA, Bravo-Marques JM, Henriksson R, Stupp R, Yue N, Bruner J, Dugan M, Rao S, Zaknoen S (2001) Multicenter phase II trial of temozolomide in patients with glioblastoma multiforme at first relapse. Ann Oncol 12:259–266
Chang SM, Theodosopoulos P, Lamborn K, Malec M, Rabbitt J, Page M, Prados MD (2004) Temozolomide in the treatment of recurrent malignant glioma. Cancer 100:605–611
Yung WK, Albright RE, Olson J, Fredericks R, Fink K, Prados MD, Brada M, Spence A, Hohl RJ, Shapiro W, Glantz M, Greenberg H, Selker RG, Vick NA, Rampling R, Friedman H, Phillips P, Bruner J, Yue N, Osoba D, Zaknoen S, Levin VA (2000) A phase II study of temozolomide vs. procarbazine in patients with glioblastoma multiforme at first relapse. Br J Cancer 83:588–593
Brandes AA, Tosoni A, Amista P, Nicolardi L, Grosso D, Berti F, Ermani M (2004) How effective is BCNU in recurrent glioblastoma in the modern era? A phase II trial. Neurology 63:1281–1284
Dresemann G (2003) STI 571/hydroxyurea in progressive, pretreated glioblastoma (GB) patients (pts.) [abstract]. Proc Am Soc Clin Oncol 22:465
Reardon D, Friedman A, Herndon J, Quinn J, Rich J, Vrendenburgh J, Badruddoja M, Desjardins A, Sampson J, Gururanagan S, Dowell J, Affronti M, Allen D, Jackson S, Zeigler K, Silverman S, Tourt-Uhlig S, Silverman S, Salvado A, Nikolova Z, Bigner D, Friedman H (2004) Phase I/II trial of imatinib mesylate plus hydroxyurea in the treatment of patients with malignant glioma [abstract]. Neuro-oncol 6:381
Macdonald DR, Cascino TL, Schold SC Jr., Cairncross JG (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Fine HA, Kim L, Royce C, Draper D, Haggarty I, Ellinzano H, Albert P, Kinney P, Musib L, Thornton D (2005) Results from phase II trial of enzastaurin (LY317615) in patients with recurrent high grade gliomas [abstract]. Proc Am Soc Clin Oncol 1504
Druker BJ, Sawyers CL, Kantarjian H, Resta DJ, Reese SF, Ford JM, Capdeville R, Talpaz M (2001) Activity of a specific inhibitor of the BCR-ABL tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome. N Engl J Med 344:1038–1042
Lokeshwar N, Kumar L, Kumari M (2005) Severe bone marrow aplasia following imatinib mesylate in a patient with chronic myelogenous leukemia. Leuk Lymphoma 46:781–784
Lewalle P, Meuleman N, Verhest A, Bron D, Martiat P (2002) Infusion of peripheral blood stem cells collected at diagnosis, with maintenance of the treatment, resulted in Ph-negative recovery in a chronic myeloid leukaemia patient in persisting aplasia on STI-571 therapy. Br J Haematol 118:144–146
de Montalembert M, Begue P, Bernaudin F, Thuret I, Bachir D, Micheau M (1999) Preliminary report of a toxicity study of hydroxyurea in sickle cell disease. French Study Group on Sickle Cell Disease. Arch Dis Child 81:437–439
Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, Dewhirst MW, Bigner DD, Rich JN (2006) Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444:756–760
Hambardzumyan D, Squatrito M, Holland EC (2006) Radiation resistance and stem-like cells in brain tumors. Cancer Cell 10:454–456
Acknowledgments
This work was supported in part by the Lawrence J. Golden Memorial Fund for GBM Research. It was presented in abstract form and as a poster at the 2006 Society for Neuro-Oncology annual meeting in Orlando, Florida. Judy Lampron and Erika Simonelli provided key editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shah, G.D., Silver, J.S., Rosenfeld, S.S. et al. Myelosuppression in patients benefiting from imatinib with hydroxyurea for recurrent malignant gliomas. J Neurooncol 85, 217–222 (2007). https://doi.org/10.1007/s11060-007-9408-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-007-9408-1