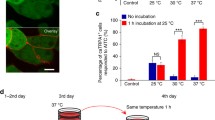
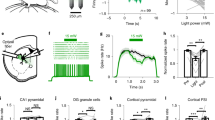
Thermogenetics appeared recently as an evolutionary advance in methods of optical stimulation of nerve cells and uses focused light to activate light-sensitive cation channels in the neuron membrane. Light activation and opening of light-sensitive channels, which can be expressed genetically in any type of neuron, induces neuron membrane depolarization and a resultant action potential. In contrast to classical methods of optogenetics, which use the visible spectrum, thermogenetics uses channels sensitive to warming. This provides the opportunity for additional activation of these channels not only using IR light, but also other methods of warming nervous tissue, such as ultrasound or microwave radiation. The penetrability of living tissue to stimulation with IR radiation is an order of magnitude greater than that for visible spectrum radiation, which allows thermogenetics methods to be used in vivo without invasive surgical methods to “clear the way” for optogenetic stimulation. On the other hand, the thermal nature of the stimulation imposes additional limits on use of the method, as heating must be sufficiently gentle so as not to induce heat shock at the cellular level, and the threshold of activation must be sufficiently high for channel opening not to occur spontaneously at normal physiological temperatures.
Similar content being viewed by others
References
Adamantidis, A. R., Zhang, F., de Lecea, L., and Deisseroth, K., “Optogenetics: opsins and optical interfaces in neuroscience,” Cold Spring Harb. Protoc., 2014, 815–822 (2014).
Bath, D. E., Stowers, J. R., Hörmann, D., et al., “FlyMAD: rapid thermogenetic control of neuronal activity in freely walking Drosophila,” Nat. Methods, 11, No. 7, 756–762 (2014).
Caterina, M. J., Rosen, T. A., Tominaga, M., et al., “A capsaicin-receptor homologue with a high threshold for noxious heat,” Nature, 398, No. 6726, 436–441 (1999).
Chen, R., Romero, G., Christiansen, M. G., et al., “Wireless magnetothermal deep brain stimulation,” Science, 347, No. 6229, 1477–1480 (2015).
Chen, S. et al., “TRP channel mediated neuronal activation and ablation in freely behaving zebrafish,” Nat. Methods, 13, No. 2, 147–150 (2016).
Chugunov, A. O., Volynsky, P. E., Krylov, N. A., et al., “Temperaturesensitive gating of TRPV1 channel as probed by atomistic simulations of its trans- and juxtamembrane domains,” Sci. Rep., 6, 33112 (2016).
Cordero-Morales, J. F., Gracheva, E. O., and Julius, D., “Cytoplasmic ankyrin repeats of transient receptor potential A1 (TRPA1) dictate sensitivity to thermal and chemical stimuli,” Proc. Natl. Acad. Sci. USA, 108, No. 46, E1184–E1191 (2011).
DeBow, S. and Colbourne, F., “Brain temperature measurement and regulation in awake and freely moving rodents,” Methods, 30, No. 2003, 167–171.
Ermakova, Y. G., Lanin, A. A., Fedotov, I. V., et al., “Thermogenetic neurostimulation with single-cell resolution,” Nat. Commun., 8, 15362 (2017).
Fedotov, I. V., Safronov, N. A., Ermakova, Y. G., et al., “Fiber-optic control and thermometry of single-cell thermosensation logic,” Sci. Rep., 5, 15737 (2015).
Gau, P., Poon, J., Ufret-Vincenty, C., et al., “The zebrafish ortholog of TRPV1 is required for heat-induced locomotion,” J. Neurosci., 33, No. 12, 5249–5260 (2013).
Gracheva, E. O., Cordero-Morales, J. F., González-Carcacía, J. A., et al., “Ganglion-specific splicing of TRPV1 underlies infrared sensation in vampire bats,” Nature, 476, No. 7358, 88–91 (2011).
Gracheva, E. O., Ingolia, N. T., Kelly, Y. M., et al., “Molecular basis of infrared detection by snakes,” Nature, 464, No. 7291, 1006–1011 (2010).
Hamada, F. N., Rosenzweig, M., Kang, K., et al., “An internal thermal sensor controlling temperature preference in Drosophila,” Nature, 454, 217–220 (2008).
Ibsen, S., Tong, A., Schutt, C., et al., “Sonogenetics is a non-invasive approach to activating neurons in Caenorhabditis elegans,” Nat. Commun., 6, 8264 (2015).
Kohatsu, S., Koganezawa, M., and Yamamoto, D., “Female contact activates male-specific interneurons that trigger stereotypic courtship behavior in Drosophila,” Neuron, 69, No. 3, 498–508 (2011).
Lanin, A. A., Fedotov, I. V., Ermakova Yu, G., et al., “Fiber-optic electron-spin-resonance thermometry of single laser-activated neurons,” Opt. Lett., 41, No. 23, 5563–5566 (2016a).
Lanin, A. A., Stepanov, E. A., Tikhonov, R. A., et al., “A compact laser platform for nonlinear Raman microspectroscopy: multimodality through broad chirp tunability,” J. Raman Spectrosc., 47, 1042–1048 (2016b).
Liao, M., Cao, E., Julius, D., and Cheng, Y., “Structure of the TRPV1 ion channel determined by electron cryo-microscopy,” Nature, 504, No. 7478, 107–112 (2013).
Liu, X., Ramirez, S., Pang, P. T., et al., “Optogenetic stimulation of a hippocampal engram activates fear memory recall,” Nature, 484, 381–385 (2012).
Mishra, A., Salari, A., Berigan, B. R., et al., “The Drosophila Gr28bD product is a non-specific cation channel that can be used as a novel thermogenetic tool,” Sci. Rep., 8, No. 1, 901 (2018).
Nikitin, E. S., Roshchin, M. V., Ierusalimskii, V. N., et al., “Optogenetic stimulation of pyramidal neuron axons in the visual cortex and hippocampus in living brain slices,” Zh. Vyssh. Nerv. Deyat., 67, No. 5, 101–108 (2017).
Oda, M., Kurogi, M., Kubo, Y., and Saitoh, O., “Sensitivities of two zebrafish TRPA1 paralogs to chemical and thermal stimuli analyzed in heterologous expression systems,” Chem. Senses, 41, No. 3, 261–272 (2016).
Paulsen, C. E., Armache, J. P., Gao, Y., et al., “Structure of the TRPA1 ion channel suggests regulatory mechanisms,” Nature, 525, No. 7570, 552 (2015a).
Paulsen, C. E., Armache, J. P., Gao, Y., et al., “Structure of the TRPA1 ion channel suggests regulatory mechanisms,” Nature, 520, No. 7548, 511–517 (2015b).
Prakash, R., Yizhar, O., Grewe, B., et al., “Two-photon optogenetic toolbox for fast inhibition, excitation and bistable modulation,” Nat. Methods, 9, 1171–1179 (2012).
Pulver, S. R., Pashkovski, S. L., Hornstein, N. J., et al., “Temporal dynamics of neuronal activation by channelrhodopsin-2 and TRPA1 determine behavioral output in Drosophila larvae,” J. Neurophysiol., 101, No. 6, 3075–3788 (2009).
Richter, C. P. and Tan, X., “Photons and neurons,” Hear. Res., 311, 72–88 (2014).
Roshchin, M., Ermakova, Y. G., Lanin, A. A., et al., “Thermogenetic stimulation of single neocortical pyramidal neurons transfected with TRPV1-L channels,” Neurosci. Lett., 687, 153–157 (2018).
Safronov, N. A., Fedotov, I. V., Ermakova Yu, G., et al., “Microwaveinduced thermogenetic activation of single cells,” Appl. Phys. Letts., 106, No. 16, 163702 (2015).
Stanley, S. A., Gagner, J. E., Damanpour, S., et al., “Radio-wave heating of iron oxide nanoparticles can regulate plasma glucose in mice,” Science, 336, No. 6081, 604–608 (2012).
Takahashi, N. and Mori, Y., “TRP channels as sensors and signal integrators of redox status changes,” Front. Pharmacol., 2, 58 (2011).
Tseeb, V., Suzuki, M., Oyama, K., et al., “Highly thermosensitive Ca dynamics in a HeLa cell through IP(3) receptors,” HFSP J, 3, No. 2, 117–123 (2009).
Voronin, A. A. and Zheltikov, A. M., “Ionization penalty in nonlinear optical bioimaging,” Phys. Rev. E Stat. Nonlin. Soft. Matter. Phys, 81, No. 5, Pt 1, 051918 (2010).
Wang, Y. Y., Chang, R. B., Waters, H. N., et al., “The nociceptor ion channel TRPA1 is potentiated and inactivated by permeating calcium ions,” J. Biol. Chem., 283, No. 47, 32691–32703 (2008).
Wetsel, W. C., “Sensing hot and cold with TRP channels,” Int. J. Hyperthermia, 27, No. 4, 388–398 (2011).
Yang, F. and Zheng, J., “High temperature sensitivity is intrinsic to voltage-gated potassium channels,” eLife, 3, e03255 (2014).
Yao, J., Liu, B., and Qin, F., “Rapid temperature jump by infrared diode laser irradiation for patch-clamp studies,” Biophys. J., 96, 3611–3619 (2009).
Yarmolenko, P. S., Moon, E. J., Landon, C., et al., “Thresholds for thermal damage to normal tissues: An update,” Int. J. Hyperthermia, 27, 320–343 (2011).
Yizhar, O. et al., “Optogenetics in neural systems,” Neuron, 71, No. 1, 9–34 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Vysshei Nervnoi Deyatel’nosti imeni I. P. Pavlova, Vol. 70, No. 1, pp. 133–140, January–February, 2020.
Rights and permissions
About this article
Cite this article
Ermakova, Y.G., Roshchin, M.V., Lanin, A.A. et al. Thermogenetics as a New Direction in Controlling the Activity of Neural Networks. Neurosci Behav Physi 50, 1018–1023 (2020). https://doi.org/10.1007/s11055-020-01001-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-020-01001-1