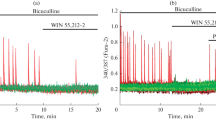
The effects of intracerebral intraventricular administration of the nonselective cannabinoid CB1 receptor WIN 55.212-2 (1 μM, 2 μl) on seizure activity were studied in conscious guinea pigs. Changes in local field potential and convulsive discharges evoked by electrical stimulation of the perforant path were recorded simultaneously in the hippocampus, entorhinal cortex, medial septal area, and amygdala after prior administration of WIN 55.212-2 or the organic solvent DMSO. In 80% of tests, WIN 55.212-2 was found to block the development of seizure activity. Repeated injections of agonist for 30 days increased the amplitude of local field potentials and the power of the theta rhythm in all structures recorded. Administration of kainic acid provoked status epilepticus in control animals, while status epilepticus did not arise in guinea pigs given daily agonist injections for 25–30 days. The possible mechanisms of the protective effects of WIN 55.212-2 are discussed.
Similar content being viewed by others
References
E. V. Astasheva and V. G. Kichigina, “Activation of the glutamatergic system of the medial septal area accelerates epileptogenesis,” Zh. Vyssh. Nerv. Deyat., 59, No. 6, 743–749 (2009).
A. Ameri and T. Simmet, “Effects of 2-arachidonylglycerol, an endogenous cannabinoid, on neuronal activity in rat hippocampal slices,” Arch. Pharmacol., 361, 265–272 (2000).
L. Calderazzo, E. A. Cavalheiro, G. Macchi, M. Molinari, and M. Bentivoglio, “Branched connections to the septum and to the entorhinal cortex from the hippocampus, amygdala, and diencephalon in the rat,” Brain Res. Bull., 40, 245–251 (1996).
L. A. Cenquizca and L. W. Swanson, “Spatial organization of direct hippocampal field CA1 axonal projections to the rest of the cerebral cortex,” Brain Res. Rev, 56, 1–26 (2007).
L. V. Colom, A. Garcia, E. R. Sambria, M. T. Castaneda, and M. G. Perez-Cordova, “Septo-hippocampal networks in chronically epileptic rats: potential antiepileptic effects of theta rhythm generation,” J. Neurophysiol., 95, 3645–3653 (2006).
L. S. Deshpande, R. E. Blair, J. M. Ziobro, S. Sombati, B. R. Martin, and R. J. DeLorenzo, “Endocannabinoids block status epilepticus in cultured hippocampal neurons,” Eur. J. Pharmacol., 58, 52–59 (2007).
L. S. Deshpande, S. Sombati, R. E. Blair, D. S. Carter, B. R. Martin, and R. J. DeLorenzo, “Cannabinoid CB1 receptor antagonists cause status epilepticus-like activity in the hippocampal neuronal culture model of acquired epilepsy,” Neurosci. Lett., 411, 11–16 (2007).
T. Esch, A Michalsen, and G. B. Stefano, “Endocannabinoids as molecular instruments of health promotion,” Med. Monatsschr. Pharmacol., 29, 397–403 (2006).
I. Ferencz, G. Leanza, A. Nanobashvili, Z. Kokaia, M. Kokaia, and O. Lindvall, “Septal cholinergic neurons suppress seizure development in hippocampal kindling in rats: comparison with noradrenergic neuron,” Neurosci., 102, 819–832 (2001).
T. F. Freund, I. Katona, and D. Piomelli, “Role of endogenous cannabinoids in synaptic signaling,” Physiol. Rev., 83, 1017–1066 (2003).
V. F. Kitchigina and M. V. Butuzova, “Theta activity of septal neurons during different epileptic phases: the same frequency but different significance?” Exp. Neurol., 216, 449–458 (2009).
S. Koda, S. Takeda, H. Onimaru, S. Akada, and A. Sakamoto, “Cannabinoid suppressed bicuculline-induced convulsion without respiratory depression in the brainstem-spinal cord preparation from newborn rats,” Biomed. Res., 26, 241–247 (2005).
C. Leranth, D. Carpi, G. Buzsaki, and J. Kiss, “The entorhino-septosupramammillary nucleus connection in the rat: morphological basis of a feedback mechanism regulating hippocampal theta rhythm,” Neurosci., 88, 701–718 (1999).
A. Ludányi, L. Eross, S. Czirják, J. Vajda, P. Halász, M. Watanabe, M. Palkovits, Z. Maglóczky, T. F. Freund, and I. Katona, “Downregulation of the CB1 cannabinoid receptor and related molecular elements of the endocannabinoid system in epileptic human hippocampus,” J. Neurosci., 28, No. 12, 2976–2990 (2008).
B. Lutz, “On-demand activation of the endocannabinoid system in the control of neuronal excitability and epileptiform seizures,” Biochem. Pharmacol., 68, 1691–1698 (2004).
G. Marsicano, S. Goodenough, K. Monory, H. Hermann, M. Eder, A. Cannich, S. C. Azad, M. G. Cascio, S. O. Gutiérrez, M. van der Stelt, M. L. López-Rodriguez, M. L. Casanova, G. Schütz,W. Zieglgänsberger, V. Di Marzo, C. Behl, and B. Lutz, “CB1 cannabinoid receptors and on-demand defense against excitotoxicity,” Science, 302, 84–88 (2003).
G. Marsicano and R. Kuner, “Anatomical distribution of receptors, ligands and enzymes in the brain and in the spinal cord: circuitries and neurochemistry,” in: Cannabinoids and the Brain, A. Köfalve (ed.), Springer, New York (2008), pp. 161–201.
J. W. Miller, G. m. Turner, and B. C. Gray, “Anticonvulsant effects of the experimental induction of hippocampal theta activity,” Epilepsy Res., 18, 195–204 (1994).
K. Monory, F. Massa, M. Egertová, M. Eder, H. Blaudzun, R. Westenbroek, W. Kelsch, W. Jacob, R. Marsch, M. Ekker, J. Long, J. L. Rubenstein, S. Goebbels, K. A. Nave, M. During, M. Klugmann, B. Wölfel, H. U. Dodt, W. Zieglgänsberger, C. T. Wotjak, K. Mackie, M. R. Elphick, G. Marsicano, and B. Lutz, “The endocannabinoid system controls key epileptogenic circuits in the hippocampus,” Neuron, 51, 455–466 (2006).
T. Nagayama, A. D. Sinor, R. P. Simon, J. Chen, S. H. Graham, K. Jin, and D. A. Greenberg, “Cannabinoids and neuroprotection in global and focal cerebral ischemia and in neuronal cultures,” J. Neurosci., 19, 2987–2995 (1999).
D. Panikashvili, C. Simeonidou, S. Ben-Shabat, L. Hanus, A. Breuer, R. Mechoulam, and E. Shohami, “An endogenous cannabinoid (2-AG) is neuroprotective after brain injury,” Nature, 413, 527–531 (2001).
R. J. Racine, “Modification of seizure activity by electrical stimulation. II. Moor seizure,” EEG Clin. Neurophysiol., 32, 281–294 (1972).
E. T. Tzavara, M. Wade, and G. G. Nomikos, “Biphasic effects of cannabinoids on acetylcholine release in the hippocampus: site and mechanism of action,” J. Neurosci., 23, 9374–9384 (2003).
O. S. Vinogradova, “Expression, control and probable functional significance of the neuronal theta-rhythm,” Progr. Neurobiol., 45, 523–583 (1995).
M. J. Wallace, R. E. Blair, K. W. Falenski, B. R. Martin, and R. J. De-Lorenzo, “The endogenous cannabinoid system regulates seizure frequency and duration in a model of temporal lobe epilepsy,” J. Pharmacol. Exp. Ther., 307, 129–137 (2003).
M. J. Wallace, J. L. Wiley, B. R. Martin, and J. R. DeLorenzo, “Assessment of the role of CB1 receptors in cannabinoid anticonvulsant effects,” Eur. J. Pharmacol., 428, 51–57 (2001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Vysshei Nervnoi Deyatel’nosti imeni I. P. Pavlova, Vol. 61, No. 1, pp. 94–101, January–February, 2011.
Rights and permissions
About this article
Cite this article
Shubina, L.V., Kichigina, V.F. Protective Effects of the CB1 Receptor Agonist WIN 55.212-2 during Development of Seizure Activity in the Brain in Models of Temporal Epilepsy in Vivo. Neurosci Behav Physi 42, 582–587 (2012). https://doi.org/10.1007/s11055-012-9604-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-012-9604-0