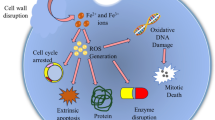
Abstract
Magnetic fluids, more specifically aqueous colloidal suspensions containing certain magnetic nanoparticles (MNPs), have recently been gaining special interest due to their potential use in clinical treatments of cancerous formations in mammalians. The technological application arises mainly from their hyperthermic behavior, which means that the nanoparticles dissipate heat upon being exposed to an alternating magnetic field (AMF). If the temperature is raised to slightly above 43 °C, cancer cells are functionally inactivated or killed; however, normal cells tend to survive under those same conditions, entirely maintaining their bioactivity. Recent in vitro studies have revealed that under simultaneous exposure to an AMF and magnetic nanoparticles, certain lines of cancer cells are bio-inactivated even without experiencing a significant temperature increase. This non-thermal effect is cell specific, indicating that MNPs, under alternating magnetic fields, may effectively kill cancer cells under conditions that were previously thought to be implausible, considering that the temperature does not increase more than 5 °C, which is also true in cases for which the concentration of MNPs is too low. To experimentally test for this effect, this study focused on the feasibility of inducing K562 cell death using an AMF and aqueous suspensions containing very low concentrations of MNPs. The assay was designed for a ferrofluid containing magnetite nanoparticles, which were obtained through the co-precipitation method and were functionalized with citric acid; the particles had an average diameter of 10 ± 2 nm and a mean hydrodynamic diameter of approximately 40 nm. Experiments were first performed to test for the ability of the ferrofluid to release heat under an AMF. The results show that for concentrations ranging from 2.5 to 1.0 × 103 mg L−1, the maximum temperature increase was actually less than 2 °C. However, the in vitro test results from K562 cells and suspensions containing these MNPs at concentrations varying within a narrower range from 2.5 to 10 mg L−1, typically under an AMF of 15 kA m−1 at 356 kHz, indicate efficient cytotoxic activity against malignant cells and inhibition of cell growth, even at very low hyperthermally induced temperature increases. The IC50 value varied with time, reaching 3.5 mg L−1 after 10 min under the AMF. Our results effectively demonstrate new prospective uses for such nanoparticles in advanced medical practices in oncology.
Graphical Abstract












Similar content being viewed by others
References
Andrade AL, Ferreira RV, Fabris JD, Domingues RZ (2011) Coating nanomagnetic particles for biomedical applications. In: Fazel R (ed) Biomedical engineering: frontiers and challenges. Nova Publishers, New York, pp 379–392. ISBN 978-953-307-309-5
Asín L, Ibarra MR, Tres S, Goya GF (2012) Controlled cell death by magnetic hyperthermia: effects of exposure time, field amplitude, and nanoparticle concentration. Pharm Res 29:1319–1327. doi:10.1007/s11095-012-0710-z
Asín L, Goya GF, Tres A, Ibarra MR (2013) Induced cell toxicity originates dendritic cell death following magnetic hyperthermia treatment. Cell Death Dis 4:e596. doi:10.1038/cddis.2013.121
Berry CC, Curtis ASG (2003) Functionalisation of magnetic nanoparticles for applications in biomedicine. J Phys D Appl Phys 36:R198–R206
Cavalli S, Carbajo D, Acosta M, Lope-Piedrafita S, Candiota AP, Arús C, Royo M, Albericio F (2012) Efficient γ-amino-proline-derived cell penetrating peptide–superparamagnetic iron oxide nanoparticle conjugates via aniline-catalyzed oxime chemistry as bimodal imaging nanoagents. Chem Commun 48:5322–5324. doi:10.1039/c2cc17937g
Creixell M, Bohorquez AC, Torres-Lugo M, Rinaldi C (2011) EGFR-targeted magnetic nanoparticle heaters kill cancer cells without a perceptible temperature rise. ACS Nano 5:7124–7129. doi:10.1021/nn201822b
Deng M, Huang Z, Zou Y, Yin G, Liu J, Gu J (2014) Fabrication and neuron cytocompatibility of iron oxide nanoparticles coated with silk-fibroin peptides. Coll Surf B 116:465–471. doi:10.1016/j.colsurfb.2014.01.021
Ding J, Tao K, Li J, Song S, Sun K (2010) Cell-specific cytotoxicity of dextran-stabilized magnetite nanoparticles. Coll Surf B 79:184–190. doi:10.1016/j.colsurfb.2010.03.053
Faraji AH, Wipf P (2009) Nanoparticles in cellular drug delivery. Bioorg Med Chem 17:2950–2962. doi:10.1016/j.bmc.2009.02.043
Ferreira RV, Pereira ILS, Cavalcante LCD, Gamarra LF, Carneiro SM, AmaroJr E, Fabris JD, Domingues RZ, Andrade AL (2010a) Synthesis and characterization of silica-coated nanoparticles of magnetite. Hyperfine Interact 195:265–274. doi:10.1007/s10751-009-0128-0
Ferreira RV, Fabris JD, Domingues RZ (2010b) Preparation of biocompatible ferrofluids for magnetic hyperthermia studies. Nanotechnol Res J 3:289–302
Frimpong RA, Dou JD, Pechan M, Hilt JZ (2010) Enhancing remote controlled heating characteristics in hydrophilic magnetite nanoparticles via facile co-precipitation. J Magn Magn Mater 322:326–331. doi:10.1016/j.jmmm.2009.09.050
Gamarra LF, Mamani JB, Carneiro SM, Fabris JD, Ferreira RV, Domingues RZ, Cornejo DR, Pontuschka WM, Amaro E (2010) Characterization of superparamagnetic iron oxide coated with silicone used as contrast agent for magnetic resonance image for the gastrointestinal tract. J Nanosci Nanotechnol 10:1153–1158. doi:10.1166/jnn.2010.1843
Goya GF, Lima E Jr, Arelaro AD, Torres T, Rechenberg HR, Rossi L, Marquina C, Ibarra MR (2008) Magnetic hyperthermia with Fe3O4 nanoparticles: The influence of particle size on energy absorption. IEEE Trans Magn 44:4444–4447. doi:10.1109/TMAG.2008.2003508
Gupta AK, Gupta M (2005) Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26:3995–4021. doi:10.1016/j.biomaterials.2004.10.012
Hajdú A, Szekeres M, Tóth IY, Bauer RA, Mihály J, Zupkó I, Tombácz E (2012) Enhanced stability of polyacrylate-coated magnetite nanoparticles in biorelevant media. Coll Surf B 94:242–249. doi:10.1016/j.colsurfb.2012.01.042
Harris L, Goff J, Carmichael A, Riffle J, Harburn J, Pierre TGS, Saunders M (2003) Magnetite nanoparticle dispersions stabilized with triblock copolymers. Chem Mater 15:1367–1377. doi:10.1021/cm020994n
Hergt R, Andra W (2007) Magnetic hyperthermia and thermoablation. In: Andra W, Nowak H (eds) Magnetism in medicine. A Handbook. WILEY-VCH, Weinheim, pp 550–567
Hergt R, Dutz S (2007) Magnetic particle hyperthermia-biophysical limitations of a visionary tumour therapy. J Magn Magn Mater 311:187–192. doi:10.1016/j.jmmm.2006.10.1156
Ho J, Al-Deen FM, Al-Abboodi A, Selomulya C, Xiang SD, Plebanski M, Forde GM (2011) N,N-Carbonyldiimidazole-mediated functionalization of superparamagnetic nanoparticles as vaccine carrier. Coll Surf B 83:83–90. doi:10.1016/j.colsurfb.2010.11.001
Hosseini F, Panahifar A, Adeli M, Amiri H, Lascialfari A, Orsini F, Doschak MR, Mahmoudi M (2013) Synthesis of pseudopolyrotaxanes-coated superparamagnetic iron oxide nanoparticles as new MRI contrast agent. Coll Surf B 103:652–657. doi:10.1016/j.colsurfb.2012.10.035
Huang SH, Juang RS (2011) Biochemical and biomedical applications of multifunctional magnetic nanoparticles: a review. J Nanopart Res 13:4411–4430. doi:10.1007/s11051-011-0551-4
Illés E, Tombácz E (2006) The effect of humic acid adsorption on pH-dependent surface charging and aggregation of magnetite nanoparticles. J Coll Int Sci 295:115–123. doi:10.1016/j.jcis.2005.08.003
Ito A, Shinkai M, Honda H, Kobayashi T (2005) Medical application of functionalized magnetic nanoparticles. J Biosci Bioeng 100:1–11. doi:10.1263/jbb.100.1
Jadhav NV, Prasad AI, Kumar A, Mishra R, Dhara S, Babu KR, Prajapat NL, Ningthoujam RS, Pandey BN, Vatsa RK (2013) Synthesis of oleic acid functionalized Fe3O4 magnetic nanoparticles and studying their interaction with tumor cells for potential hyperthermia applications. Colloid Surf B Biointerfaces 108:158–168. doi:10.1016/j.colsurfb.2013.02.035
Jordan A, Scholz R, Wust P, Schirra H, Schiestel T, Schmidt H, Felix R (1999) Endocytosis of dextran and silan-coated magnetite nanoparticles and the effect of intracellular hyperthermia on human mammary carcinoma cells in vitro. J Magn Magn Mater 194:185–196. doi:10.1016/S0304-8853(98)00558-7
Kulshrestha P, Gogoia M, Bahadur D, Banerjee R (2012) In vitro application of paclitaxel loaded magnetoliposomes for combined chemotherapy and hyperthermia. Coll Surf B 96:1–7. doi:10.1016/j.colsurfb.2012.02.029
Laurent S, Forge D, Port M, Roch A, Robic C, Vander Elst L, Muller RN (2008) Magnetic iron oxide nanoparticles: synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem Rev 108:2064–2110. doi:10.1021/cr068445e
Laurent S, Dutz S, Häfeli UO (2011) Magnetic fluid hyperthermia: focus on superparamagnetic iron oxide nanoparticles. Adv Coll Interface Sci 166:8–23. doi:10.1016/j.cis.2011.04.003
Lee SJ, Jeong JR, Shin SC, Kim JC, Chang YH et al (2004) Nanoparticles of magnetic ferric oxides encapsulated with poly(D, L lactide-co-glycolide) and their applications to magnetic resonance imaging contrast agent. J Magn Magn Mater 272–276:2432–2433. doi:10.1016/j.jmmm.2003.12.416
MagForce AG (2010). http://www.magforce.de/en/unternehmen/ueber-uns.html. Accessed 6 Oct 2015
Mahmoudi M, Sant S, Wang F, Laurent S, Sen T (2011) Superparamagnetic iron oxide nanoparticles (SPIONs): development, surface modification and applications in chemotherapy. Adv Drug Deliv Rev 63:24–46. doi:10.1016/j.addr.2010.05.006
Marcos-Campos I, Asin L, Torres TE, Marquina C, Tres A, Ibarra MR, Goya GF (2011) Cell death induced by the application of alternating magnetic fields to nanoparticle-loaded dendritic cells. Nanotechnology 22:1–9. doi:10.1088/0957-4484/22/20/205101
Morup S, Topsoe E, Lipka L (1976) Modified theory for Mössbauer spectra of superparamagnetic particles: application to Fe3O4. J Phys Colloq 37:C6-287–C6-290. doi:10.1051/jphyscol:1976658
Mout R, Moyano DF, Rana S, Rotello VM (2012) Surface functionalization of nanoparticles for nanomedicine. Chem Soc Rev 41:2539–2544. doi:10.1039/C2CS15294K
Neuberger T, Schöpf B, Hofmann H, Hofmann M, Von Rechenberg B (2005) Superparamagnetic nanoparticles for biomedical applications: possibilities and limitations of a new drug delivery system. J Magn Magn Mater 293:483–496. doi:10.1016/j.jmmm.2005.01.064
Nguyen TD (2013) Portraits of colloidal hybrid nanostructures: controlled synthesis and potential applications. Coll Surf B 103:326–344. doi:10.1016/j.colsurfb.2012.10.049
Nyamjav D, Ivanisevic A (2005) Templates for DNA-templated Fe3O4 nanoparticles. Biomaterials 26:2749–2757. doi:10.1016/j.biomaterials.2004.07.025
Pankhurst QA, Connolly J, Jones SK, Dobson J (2003) Applications of magnetic nanoparticles in biomedicine. J Phys D 36:167
Reddy KR, Lee KP, Iyengar AG (2007) Synthesis and characterization of novel conducting composites of Fe3O4 nanoparticles and sulfonated polyanilines. J Appl Polym Sci 104:4127–4134. doi:10.1002/app.26020
Roca AG, Marco JF, del Puerto Morales M, Serna CJ (2007) Effect of nature and particle size on properties of uniform magnetite and maghemite nanoparticles. J Phys Chem C 111:18577–18584. doi:10.1021/jp075133m
Salunkhe AS, Khot VM, Pawar SH (2014) Magnetic hyperthermia with magnetic nanoparticles: a status review. Curr Top Med Chem 14:572–594. doi:10.2174/1568026614666140118203550
Saraswathy A, Nazeer SS, Jeevan M, Nimi N, Vijayakumar S, Harikrishnanc S, Harikrishna PR, Ramapurath V, Jayasreea S (2014) Citrate coated iron oxide nanoparticles with enhanced relaxivity for in vivo magnetic resonance imaging of liver fibrosis. Coll Surf B 117:216–224. doi:10.1016/j.colsurfb.2014.02.034
Schwertmann U, Cornell RM (2003) The iron oxides: structure, properties, occurrences and uses. WILEY-VCH, Weinheim
Sharifi I, Shokrollahi H, Amiri S (2012) Ferrite-based magnetic nanofluids used in hyperthermia applications. J Magn Magn Mater 324:903–915. doi:10.1016/j.jmmm.2011.10.017
Sousa ME, van Raap MBF, Rivas PC, Zélis PM, Girardin P, Pasquevich GA, Alessandrini JL, Muraca D, Sánchez FH (2013) Stability and relaxation mechanisms of citric acid coated magnetite nanoparticles for magnetic hyperthermia. J Phys Chem C 117:5436–5445. doi:10.1021/jp311556b
Stucki JW (1981) The quantitative assay of minerals for Fe2+ and Fe3+ using 1,10-phenanthroline. II. A photochemical method. Soil Sci Soc Am J 45:638–641
Tartaj P, Morales MP, Veintemillas-Verdaguer S, González-Carreno T, Serna CJ (2003) The preparation of magnetic nanoparticles for applications in biomedicine. J Phys D 36:R182–R197. doi:10.2136/sssaj1981
Thomas OC, Hedayati M, Zhou H, Zheng Y, Wabler M, Mihalic J, Geyh A, Ivkov R, Deweese TL (2011) Thermal and non-thermal effects of membrane bound ferromagnetic nanoparticles. Int J Radiat Oncol Biol Phys 81:S749–S750. doi:10.1016/j.ijrobp.2011.06.1242
Villanueva A, Presa P, Alonso JM, Rueda T, Martínez A, Crespo P, Morales MP, Gonzalez-Fernandez MA, Valdés J, Rivero G (2010) Hyperthermia HeLa cell treatment with silica-coated manganese oxide nanoparticles. J Phys Chem C 114:1976–1981. doi:10.1021/jp907046f
Wei Y, Yin G, Ma C, Huang Z, Chen X, Liao X, Yao Y, Yin H (2013) Synthesis and cellular compatibility of biomineralized Fe3O4 nanoparticles in tumor cells targeting peptides. Coll Surf B 107:180–188. doi:10.1016/j.colsurfb.2013.01.058
ZetaSizer Nano Series User Manual (2004) Malvern Instruments Ltd., Malvern, Worcestershire
Zhang L, He R, Gu HC (2006) Oleic acid coating on the monodisperse magnetite nanoparticles. Appl Surf Sci 253:2611–2617. doi:10.1016/j.apsusc.2006.05.023
Acknowledgments
This work was supported by the Minas Gerais State Agency for the Development of Scientific Research (FAPEMIG; Brazil), the National Institute of Science and Technology for Catalysis (INCT-Catálise; Brazil), and the National Council for Scientific and Technological Development (CNPq; Brazil). JDF would like to thank the Coordination of Superior Level Staff Improvement (CAPES; Brazil) for granting his Visiting Professorship at the Federal University of the Jequitinhonha and Mucuri Valleys (UFVJM; Brazil) under the PVNS program and the CNPq for Grant # 305755-2013-7. LCDC is currently a postdoctoral fellow at UFMG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferreira, R.V., Silva-Caldeira, P.P., Pereira-Maia, E.C. et al. Bio-inactivation of human malignant cells through highly responsive diluted colloidal suspension of functionalized magnetic iron oxide nanoparticles. J Nanopart Res 18, 92 (2016). https://doi.org/10.1007/s11051-016-3400-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-016-3400-7