Abstract
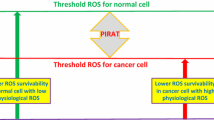
Iron oxide (Fe3O4) nanoparticles (NPs) are increasingly recognized for their utility in biomedical applications. However, little is known about the anticancer activity of Fe3O4 NPs. This study was designed to investigate whether Fe3O4 NPs induced toxicity in a cell-specific manner and determine the possible mechanisms of toxicity caused by Fe3O4 NPs in cancer cells. Fe3O4 NPs used in this study were synthesized by green method using α-d-glucose as a reducing agent. Prepared Fe3O4 NPs were spherical in shape with a smooth surface, were fairly distributed, and had an average diameter of 23 nm. Cytotoxicity of Fe3O4 NPs was examined against two types of cancer cells (human hepatocellular carcinoma HepG2 and human lung adenocarcinoma A549) and two normal cells (human lung fibroblast IMR-90 and rat hepatocytes). Fe3O4 NPs exerted distinct effects on cell viability via killing of cancer cells while posing no toxicity on normal cells. Fe3O4 NPs were found to induce depletion of glutathione and induction of reactive oxygen species (ROS) in both types of cancer cells (HepG2 and A549). Further, co-exposure of ascorbic acid significantly attenuated the Fe3O4 NPs-induced oxidative stress. The mRNA levels of tumor suppressor gene p53 and apoptotic genes (caspase-3 and caspase-9) were up-regulated in both types of cancer cells due to Fe3O4 NPs exposure. Protein level of p53, along with the higher activity of caspase-3 and caspase-9 enzymes, was also up-regulated by Fe3O4 NPs. Taken together, our data demonstrated that Fe3O4 NPs selectively induced apoptosis in cancer cells (HepG2 and A549) through up-regulation of p53 that might be mediated by ROS through which most of the anticancer drugs trigger apoptosis. The present study warrants further investigation on anticancer activity of Fe3O4 NPs in relevant animal models.






Similar content being viewed by others
References
Ahamed M, Akhtar MJ, Raja M, Ahmad I, Siddiqui MKJ, AlSalhi MS, Alrokayan SA (2011a) ZnO nanorod-induced apoptosis in human alveolar adenocarcinoma cells via p53, survivin and bax/bcl-2 pathways: role of oxidative stress. Nanomedicine 7:904–913. doi:10.1016/j.nano.2011.04.011
Ahamed M, Akhtar MJ, Siddiqui MA, Ahmad J, Musarrat J, Al-Khedhairy AA, Alrokayan SA (2011b) Oxidative stress mediated apoptosis induced by nickel ferrite nanoparticles in cultured A549 cells. Toxicology 283:101–108. doi:10.1016/j.tox.2011.02.010
Ahmad J, Ahamed M, Akhtar MJ, Alrokayan SA, Siddiqui MA, Musarrat J, Al-Khedhairy AA (2012) Apoptosis induction by amorphous silica nanoparticles mediated through reactive oxygen species generation in human liver cell line HepG2. Toxicol Appl Pharmacol 259:160–168. doi:10.1016/j.taap.2011.12.020
Akhtar MJ, Ahamed M, Kumar S, Ahmad J, Khan MAM, Alrokayan SA (2012) Zinc oxide nanoparticles selectively induce apoptosis in cancer cells through reactive oxygen species. Int J Nanomed 7:845–857. doi:10.2147/IJN.S29129
Bai W, Zhang Z, Tian W, He X, Ma Y, Zhao Y, Chai Z (2009) Toxicity of zinc oxide nanoparticles to zebrafish embryo: a physicochemical study of toxicity mechanism. J Nanopart Res 12:1645–1654. doi:10.1007/s11051-009-9740-9
Bosanquet AG, Bell PB (2004) Ex vivo therapeutic index by drug sensitivity assay using fresh human normal and tumor cells. J Exp Ther Oncol 4:145–154
Boyle P, Levin B (2009) World cancer report 2008. International Agency for Research on Cancer World Health Organization. Available at: http://www.iarc.fr/en/publications/pdfs-online/wcr
Chougule M, Patel AR, Sachdeva P, Jackson T, Singh M (2011) Anticancer activity of Noscapine, an opioid alkaloid in combination with cisplatin in human non-small cell lung cancer. Lung Cancer 71:271–282. doi:10.1016/j.lungcan.2010.06.002
Ellman GI (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. doi:10.1016/0003-9861(59)90090-6
Farnebo M, Bykov VN, Wiman KG (2010) The p53 tumor suppressor: a master regulator of diverse cellular processes and therapeutic target in cancer. Biochem Biophys Res Commun 396:85–89. doi:10.1016/j.bbrc.2010.02.152
Hanley C, Layne J, Punnoose A, Reddy KM, Coombs I, Coombs A, Feris K, Wingett D (2008) Preferential killing of cancer cells and activated human T cells using ZnO nanoparticles. Nanotechnology 19:295103. doi:10.1088/0957-4484/19/29/295103
Krishnakumar N, Sulfikkarali N, Rajendraprasad N, Karthikeyan S (2011) Enhanced anticancer activity of naringenin-loaded nanoparticles in human cervical (HeLa) cancer cells. Biomed Prev Nutr 1:223–231. doi:10.1016/j.bionut.2011.09.003
Lane DP, Cheok CF, Lain S (2010) p53-Based cancer therapy. Cold Spring Harb Perspect Biol 2:a001222. doi:10.1101/cshperspect.a001222
Lanone S, Boczkowski J (2006) Biomedical applications and potential health risks of nanomaterials: molecular mechanisms. Curr Mol Med 6:651–663
Lu W, Shen Y, Xie A, Zhang W (2010) Green synthesis and characterization of superparamagnetic Fe3O4 nanoparticles. J Magn Magn Mater 322:1828–1833. doi:10.1016/j.jmmm.2009.12.035
Lu X, Qian J, Zhou H, Gan Q, Tang W, Lu J, Yuan J, Liu C (2011) In vitro cytotoxicity and induction of apoptosis by silica nanoparticles in human HepG2 hepatoma cells. Int J Nanomed 6:1889–1901. doi:10.2147/IJN.S24005
Moldeus P (1978) Isolation and use of liver cells. In: Fleischer S, Pscker L (eds) Methods in enzymology. Academic Press, New York, p 60
Mossman T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63. doi:10.1016/0022-1759(83)90303-4
Murdock RC, Braydich-Stolle L, Schrand AM, Schlager JJ, Hussain SM (2008) Characterization of nanomaterial dispersion in solution prior to in vitro exposure using dynamic light scattering technique. Toxicol Sci 101:239–253. doi:10.1093/toxsci/kfm240
Nel A, Xia T, Madler L, Li N (2006) Toxic potential of materials at the nanolevel. Science 311:622–627. doi:10.1126/science.1114397
Ng KW, Khoo SK, Heng BC, Setyawati MI, Tan EC, Zhao X, Xiong S, Fang W, Leong DT, Loo JS (2011) The role of the tumor suppressor p53 pathway in the cellular DNA damage response to zinc oxide nanoparticles. Biomaterials 32:8218–8225. doi:10.1016/j.biomaterials.2011.07.036
Nie S, Xing Y, Kim GJ, Simons JW (2007) Nanotechnology applications in cancer. Annu Rev Biomed Eng 9:257–288. doi:10.1146/annurev.bioeng.9.060906.152025
Park JH, Lee S, Kim JH, Park K, Kim K, Kwon IC (2008) Polymeric nanomedicine for cancer therapy. Prog Polymer Sci 33:113–137. doi:10.1016/j.progpolymsci.2007.09.003
Patterson AL (1939) The Scherrer formula for X-ray particle size determination. Phys Rev 56:978–982. doi:10.1103/PhysRev.56.978
Premanathan M, Karthikeyan K, Jeyasubramanian K, Manivannan G (2011) Selective toxicity of ZnO nanoparticles toward Gram positive bacteria and cancer cells by apoptosis through lipid peroxidation. Nanomedicine 7:184–192. doi:10.1016/j.nano.2010.10.001
Rasmussen JW, Martinez E, Louka P, Wingett DG (2010) Zinc oxide nanoparticles for selective destruction of tumor cells and potential for drug delivery applications. Expert Opin Drug Deliv 7:1063–1077. doi:10.1517/17425247.2010.502560
Sánchez-Pérez Y, Chirino YI, Osornio-Vargas AR, Morales-Bárcenas R, Gutiérrez-Ruíz C, Vázquez-López I, Garcia-Cuellar CM (2009) DNA damage response of A549 cells treated with particulate matter (PM10) of urban air pollutants. Cancer Lett 278:192–200. doi:10.1016/j.canlet.2009.01.010
Sharma V, Shukla RK, Saxena N, Parmar D, Das M, Dhawan A (2009) DNA damaging potential of zinc oxide nanoparticles in human epidermal cells. Toxicol Lett 185:211–218. doi:10.1016/j.toxlet.2009.01.008
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85. doi:10.1016/0003-2697(85)90442-7
Timmer JC, Salvesen GS (2007) Caspase substrates. Cell Death Differ 14:66–72. doi:10.1038/sj.cdd.4402059
Tosi G, Costantino L, Ruozi B, Forni F, Vandelli MA (2008) Polymeric nanoparticle for the drug delivery to the central nervous system. Expert Opin Drug Deliv 5:155–174. doi:10.1517/17425247.5.2.155
Trachootham D, Alexandre J, Huang P (2009) Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nat Rev Drug Discov 8:579–591. doi:10.1038/nrd2803
Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L, Newman J, Reczek EE, Weissleder R, Jacks T (2007) Restoration of p53 function leads to tumor regression in vivo. Nature 445:661–665. doi:10.1038/nature05541
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27:612–661. doi:10.1016/S0891-5849(99)00107-0
Wang M, Thanou M (2010) Targeting nanoparticles to cancer. Pharmacol Res 62:90–99. doi:10.1016/j.phrs.2010.03.005
WHO (2010) Cancer. World Health Organization. http://www.who.int/mediacentre/factsheets/fs297/en/. Retrieved 1 May 2011
Xia T, Kovochich M, Liong M, Mädler L, Gilbert B, Shi H, Yeh JI, Zink JI, Nel AE (2008) Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano 2:2121–2134. doi:10.1021/nn800511k
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V, Cordon-Cardo C, Lowe SW (2007) Senescence and tumor clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445:656–660. doi:10.1038/nature05529
Youle RJ, Strasser A (2008) The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 9:47–59. doi:10.1038/nrm2308
Acknowledgments
This work was supported by King Abdulaziz City for Science and Technology (KACST) under the National Plan for Science and Technology (NPST) (Grant No. 10-NAN1201-02).
Conflict of interest
The authors declare that there are no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahamed, M., Alhadlaq, H.A., Khan, M.A.M. et al. Selective killing of cancer cells by iron oxide nanoparticles mediated through reactive oxygen species via p53 pathway. J Nanopart Res 15, 1225 (2013). https://doi.org/10.1007/s11051-012-1225-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-012-1225-6