Abstract
Background
Timely and sufficient M1 recruitment and M2 polarization are necessary for fibrosis during wound healing. The mechanism of how M2 mediates wound healing is worth exploring. Abnormally up-regulated connective tissue growth factor (CTGF) influences multiple organ fibrosis, including cardiac, pulmonary, hepatic, renal, and cutaneous fibrosis. Previous studies reported that M2 contributed to hepatic and renal fibrosis by secreting CTGF. It is worth discussing if M2 regulates fibrosis through secreting CTGF in wound healing.
Methods and results
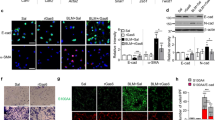
We established the murine wound model and inhibited macrophages during proliferation phase with clodronate liposomes in vivo. Macrophages depletion led to down-regulation of wound healing rates, collagen deposition, as well as expression of collagen 1/3 and Ki67. M2 was induced by interleukin-4 (IL-4) and measured by flow cytometry in vitro. Secreted pro-fibrotic and anti-fibrotic factors were tested by enzyme-linked immunosorbent assay (ELISA). M2 was polarized, which producing more CTGF, transforming growth factor-beta1 (TGF-β1), and IL-6, as well as less tumor necrosis factor-α (TNF-α) and IL-10. M2 CTGF gene was blocked using siCTGF. Effects of M2 on fibroblasts activities were detected by cell counting kit 8 (CCK8) and cellular wound healing assay. Expressions of related signaling pathway were assessed by western blotting. Blockade of CTGF in M2 deactivated fibroblasts proliferation and migration by regulating AKT, ERK1/2, and STAT3 pathway. Recombinant CTGF restored these effects.
Conclusions
Our research, for the first time, indicated that M2 promoted wound healing by secreting CTGF, which further mediating proliferation and migration of fibroblasts via AKT, ERK1/2, and STAT3 pathway.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included.
References
Rockey DC, Bell PD, Hill JA (2015) Fibrosis–a common pathway to organ injury and failure. New Engl J Med 372:1138–1149
Martin P, Leibovich SJ (2005) Inflammatory cells during wound repair: the good, the bad and the ugly. Trends Cell Biol 15:599–607
Hopkinson-Woolley J, Hughes D, Gordon S et al (1994) Macrophage recruitment during limb development and wound healing in the embryonic and foetal mouse. J Cell Sci 107:1159–1167
van Furth R, Nibbering PH, van Dissel JT et al (1985) The characterization, origin, and kinetics of skin macrophages during inflammation. J Invest Dermatol 85:398–402
Martin P, D’Souza D, Martin J et al (2003) Wound healing in the PU.1 null mouse–tissue repair is not dependent on inflammatory cells. Curr Biol 13:1122–1128
Tacke F, Randolph GJ (2006) Migratory fate and differentiation of blood monocyte subsets. Immunobiology 211:609–618
Ginhoux F, Tacke F, Angeli V (2006) Langerhans cells arise from monocytes in vivo. Nat Immunol 7:265–273
Klahr S, Morrissey J (2002) Obstructive nephropathy and renal fibrosis. Am J Physiol Renal Physiol 283:F861-875
Leask A (2013) CCN2: a novel, specific and valid target for anti-fibrotic drug intervention. Expert Opin Ther Targets 17:1067–1071
Kwan PO, Tredget EE (2017) Biological principles of scar and contracture. Hand Clin 33:277–292
Takigawa M (2018) An early history of CCN2/CTGF research: the road to CCN2 via hcs24, ctgf, ecogenin, and regenerin. J Cell Commun Signal 12:253–264
Lau LF (2016) Cell surface receptors for CCN proteins. J Cell Commun Signal 10:121–127
Montford JR, Furgeson SB (2017) A new CTGF target in renal fibrosis. Kidney Int 92:784–786
Shi C, Li G, Tong Y, Deng Y et al (2016) Role of CTGF gene promoter methylation in the development of hepatic fibrosis. Am J Transl Res 8:125–132
Kok HM, Falke LL, Goldschmeding R et al (2014) Targeting CTGF, EGF and PDGF pathways to prevent progression of kidney disease. Nat Rev Nephrol 10:700–711
Ferdoushi S, Paul D, Ghosh CK et al (2015) Correlation of connective tissue growth factor (CTGF/CCN2) with hepatic fibrosis in chronic hepatitis B. Mymensingh Med J 24:558–563
Makino K, Makino T, Stawski L et al (2017) Anti-connective tissue growth factor (CTGF/CCN2) monoclonal antibody attenuates skin fibrosis in mice models of systemic sclerosis. Arthritis Res Ther 19:134
Leask A (2017) CCN2 in skin fibrosis. Methods Mol Biol 1489:417–421
Hayakawa K, Ikeda K, Fujishiro M et al (2018) Connective tissue growth factor neutralization aggravates the psoriasis skin lesion: the analysis of psoriasis model mice and patients. Ann Dermatol 30:47–53
Alfaro MP, Deskins DL, Wallus M (2013) A physiological role for connective tissue growth factor in early wound healing. Lab Invest 93:81–95
Nakai K, Karita S, Igarashi J et al (2019) COA-Cl prevented TGF-β1-induced CTGF expression by Akt dephosphorylation in normal human dermal fibroblasts, and it attenuated skin fibrosis in mice models of systemic sclerosis. J Dermatol Sci 94:205–212
Henrot P, Truchetet ME, Fisher G et al (2019) CCN proteins as potential actionable targets in scleroderma. Exp Dermatol 28:11–18
Janis JE, Harrison B (2016) Wound healing: part I. Basic science. Plast Reconstr Surg 138:9S-17S
Salehi S, Reed EF (2015) The divergent roles of macrophages in solid organ transplantation. Curr Opin Organ Transplant 20:446–453
Kamdem SD, Moyou-Somo R, Brombacher F (2018) Host regulators of liver fibrosis during human schistosomiasis. Front Immunol 9:2781
Ikezumi Y, Suzuki T, Karasawa T et al (2011) Identification of alternatively activated macrophages in new-onset paediatric and adult immunoglobulin A nephropathy: potential role in mesangial matrix expansion. Histopathology 58:198–210
Ikezumi Y, Suzuki T, Yamada T et al (2015) Alternatively activated macrophages in the pathogenesis of chronic kidney allograft injury. Pediatr Nephrol 30:1007–1017
Zhang S, Chen C, Ying J et al (2020) Alda-1, an aldehyde dehydrogenase 2 agonist, improves cutaneous wound healing by activating epidermal keratinocytes via Akt/GSK-3beta/beta-catenin pathway. Aesthetic Plast Surg 44:993–1005
Smith TD, Tse MJ, Read EL et al (2016) Regulation of macrophage polarization and plasticity by complex activation signals. Integr Biol (Camb) 8:946–955
Smigiel KS, Parks WC (2018) Macrophages, wound healing, and fibrosis: recent insights. Curr Rheumatol Rep 20:17
Edholm ES, Rhoo KH, Robert J (2017) Evolutionary aspects of macrophages polarization. Results Probl Cell Differ 62:3–22
Martinez FO, Gordon S (2014) The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep 6:13–13
Tang PM, Nikolic-Paterson DJ, Lan HY (2019) Macrophages: versatile players in renal inflammation and fibrosis. Nat Rev Nephrol 15:144–158
Carlson S, Helterline D, Asbe L et al (2017) Cardiac macrophages adopt profibrotic/M2 phenotype in infarcted hearts: Role of urokinase plasminogen activator. J Mol Cell Cardiol 108:42–49
Harrell CR, Markovic BS, Fellabaum C et al (2020) The role of Interleukin 1 receptor antagonist in mesenchymal stem cell-based tissue repair and regeneration. BioFactors 46:263–275
Zhu Z, Ding J, Ma Z et al (2017) Alternatively activated macrophages derived from THP-1 cells promote the fibrogenic activities of human dermal fibroblasts. Wound Repair Regen 25:377–388
Liu XC, Liu BC, Zhang XL et al (2007) Role of ERK1/2 and PI3-K in the regulation of CTGF-induced ILK expression in HK-2 cells. Clin Chim Acta 382:89–94
Yang M, Huang H, Li J et al (2007) Connective tissue growth factor increases matrix metalloproteinase-2 and suppresses tissue inhibitor of matrix metalloproteinase-2 production by cultured renal interstitial fibroblasts. Wound Repair Regen 15:817–824
Radhakrishnan SS, Blalock TD, Robinson PM et al (2012) Effect of connective tissue growth factor on protein kinase expression and activity in human corneal fibroblasts. Invest Ophthalmol Vis Sci 53:8076–8085
Sonnylal S, Xu SW, Leoni P et al (2010) Selective expression of connective tissue growth factor in fibroblasts in vivo promotes systemic tissue fibrosis. Arthritis Rheum 62:1523–1532
Chujo S, Shirasaki F, Kondo-Miyazaki M et al (2009) Role of connective tissue growth factor and its interaction with basic fibroblast growth factor and macrophage chemoattractant protein-1 in skin fibrosis. J Cell Physiol 220:189–195
Toda N, Mori K, Kasahara M et al (2017) Crucial role of mesangial cell-derived connective tissue growth factor in a mouse model of anti-glomerular basement membrane glomerulonephritis. Sci Rep 7:42114
Choi Y, Yoo JH, Lee JH et al (2020) Connective tissue growth factor (CTGF) regulates the fusion of osteoclast precursors by inhibiting Bcl6 in periodontitis. Int J Med Sci 17:647–656
Wang TT, Yuan JH, Ma JZ et al (2017) CTGF secreted by mesenchymal-like hepatocellular carcinoma cells plays a role in the polarization of macrophages in hepatocellular carcinoma progression. Biomed Pharmacother 95:111–119
Acknowledgements
We are indebted to all individuals who participated in or helped with this research project.
Author information
Authors and Affiliations
Contributions
All authors participated in the design, interpretation of the studies and analysis of the data and review of the manuscript; SMZ devoted to study design, data collection, and manuscript preparation. CYW and QW devoted to study design and data interpretation. Lu Wang participated in statistical analysis and paper revision. LL participated in statistical analysis and data interpretation. FZQ provided substantial advice in study design, division of labor, manuscript preparation, and funds collection.
Corresponding author
Ethics declarations
Conflict of interest
All Authors declare that they have no competing interest.
Ethical approval
All manipulations involving C57BL/6 mice were carried out in accordance with the Ethics Committee of the Zhongshan Hospital Fudan University and national ethical guidelines for experimental protocols on animals (Permission No. 2019-067).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, SM., Wei, CY., Wang, Q. et al. M2-polarized macrophages mediate wound healing by regulating connective tissue growth factor via AKT, ERK1/2, and STAT3 signaling pathways. Mol Biol Rep 48, 6443–6456 (2021). https://doi.org/10.1007/s11033-021-06646-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06646-w