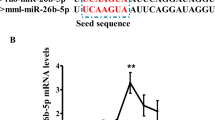
Abstract
Our previous studies have shown that miR-125b-5p was highly expressed and significantly upregulated during abdominal fat deposition in chickens. However, the role of miR-125b in the regulation of adipogenesis is not clear in chickens. Therefore, we evaluated the effects of miR-125b-5p on preadipocyte proliferation and differentiation and the interaction between miR-125b-5p and the acyl-CoA synthetase bubblegum family member 2 (ACSBG2) gene in adipogenesis in chicken abdominal adipose tissue. Here, transfection tests of miR-125b-5p mimic/inhibitor were performed in preadipocytes, and the effects of miR-125b-5p on preadipocytes proliferation and differentiation were analyzed. The target site of miR-125b-5p in the 3′UTR (untranslated region) of ACSBG2 were verified by a luciferase reporter assay. Our results showed that miR-125b-5p overexpression inhibited proliferation and reduced the number of cells in S phase and G2/M phase in preadipocytes; conversely, miR-125b-5p inhibition promoted the proliferation and increased the number of cells in S phase and G2/M phase. In adipocytes after induction, miR-125b-5p overexpression led to a notable increase in the accumulation of lipid droplets as well as in the concentration of triglycerides, while miR-125b-5p inhibition had the opposite effect. Furthermore, miR-125b-5p could directly bind to the 3'UTR of ACSBG2, and its overexpression could significantly repress the mRNA and protein expression of ACSBG2. These results indicate that miR-125b-5p can inhibit preadipocyte proliferation and can promote preadipocyte differentiation to affect adipogenesis in chicken abdominal adipose tissues, at least partially by downregulating ACSBG2.





Similar content being viewed by others
Data Availability
The data and material used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Bai S, Wang G, Zhang W, Zhang S, Rice BB, Cline MA, Gilbert ER (2015) Broiler chicken adipose tissue dynamics during the first two weeks post-hatch. Comp Biochem Physiol A Mol Integr Physiol 189:115–123
Abdalla BA, Chen J, Nie Q, Zhang X (2018) Genomic insights into the multiple factors controlling abdominal fat deposition in a chicken model. Front Genet 9:262
Ahn J, Lee H, Jung CH, Jeon TI, Ha TY (2013) MicroRNA-146b promotes adipogenesis by suppressing the SIRT1-FOXO1 cascade. EMBO Mol Med 5:1602–1612
Chen L, Hou J, Ye L, Chen Y, Cui J, Tian W, Li C, Liu L (2014) MicroRNA-143 regulates Adipogenesis by modulating the MAP2K5–ERK5 signaling. Sci Rep 4:3819
Ling HY, Wen GB, Feng SD, Tuo QH, Ou HS, Yao CH, Zhu BY, Gao ZP, Zhang L, Liao DF (2011) MicroRNA-375 promotes 3T3-L1 adipocyte differentiation through modulation of extracellular signal-regulated kinase signalling. Clin Exp Pharmacol Physiol 38:239–246
Jeong BC, Kang IH, Koh JT (2014) MicroRNA-302a inhibits adipogenesis by suppressing peroxisome proliferator-activated receptor γ expression. FEBS Lett 588:3427–3434
Karbiener M, Fischer C, Nowitsch S, Opriessnig P, Papak C, Ailhaud G, Dani C, Amri EZ, Scheideler M (2009) microRNA miR-27b impairs human adipocyte differentiation and targets PPARγ. Biochem Biophys Res Commun 390:247–251
Peng Y, Li H, Li X, Yu S, Xiang H, Peng J, Jiang S (2016) MicroRNA-215 impairs adipocyte differentiation and co-represses FNDC3B and CTNNBIP1. Int J Biochem Cell Biol 79:104–112
Cortinas L, Barroeta A, Villaverde C, Galobart J, Guardiola F, Baucells M (2005) Influence of the dietary polyunsaturation level on chicken meat quality: lipid oxidation. Poult Sci 84:48–55
Wang L, Cheng B, Li H, Wang Y (2019) Proteomics analysis of preadipocytes between fat and lean broilers. Br Poult Sci 60:522–529
Huang H, Liu R, Zhao G, Li Q, Zheng M, Zhang J, Li S, Liang Z, Wen J (2015) Integrated analysis of microRNA and mRNA expression profiles in abdominal adipose tissues in chickens. Sci Rep 5:16132
Yao J, Wang Y, Wang W, Wang N, Li H (2011) Solexa sequencing analysis of chicken pre-adipocyte microRNAs. Biosci Biotechnol Biochem 75:54–61
Wang W, Du ZQ, Cheng B, Wang Y, Yao J, Li Y, Cao Z, Luan P, Wang N, Li H (2015) Expression profiling of preadipocyte microRNAs by deep sequencing on chicken lines divergently selected for abdominal fatness. PLoS One 10:e0117843
Ma X, Sun J, Zhu S, Zhenwei D, Li D, Li W, Li Z, Tian Y, Kang X, Sun G (2020) MiRNAs and mRNAs analysis during abdominal preadipocyte differentiation in chickens. Animals 10:468
Sun YM, Lin KY, Chen YQ (2013) Diverse functions of miR-125 family in different cell contexts. J Hematol Oncol 6:6
Guan Y, Yao H, Zheng Z, Qiu G, Sun K (2011) MiR-125b targets BCL3 and suppresses ovarian cancer proliferation. Int J Cancer 128:2274–2283
Wang YD, Cai N, Wu X, Cao H, Xie L, Zheng P (2013) OCT4 promotes tumorigenesis and inhibits apoptosis of cervical cancer cells by miR-125b/BAK1 pathway. Cell Death Dis 4:e760
Cheng X, Xi QY, Wei S, Wu D, Ye RS, Chen T, Qi QE, Jiang QY, Wang SB, Wang LN (2016) Critical role of miR-125b in lipogenesis by targeting stearoyl-CoA desaturase-1 (SCD-1). J Anim Sci 94:65–76
Rockstroh D, Löffler D, Kiess W, Landgraf K, Körner A (2016) Regulation of human adipogenesis by miR125b-5p. Adipocyte 5:283–297
Giroud M, Pisani DF, Karbiener M, Barquissau V, Ghandour RA, Tews D, Fischer-Posovszky P, Chambard JC, Knippschild U, Niemi T (2016) miR-125b affects mitochondrial biogenesis and impairs brite adipocyte formation and function. Mol Metab 5:615–625
Ouyang D, Ye Y, Guo D, Yu X, Chen J, Qi J, Tan X, Zhang Y, Ma Y, Li Y (2015) MicroRNA-125b-5p inhibits proliferation and promotes adipogenic differentiation in 3T3-L1 preadipocytes. Acta Biochim Biophys Sin 47:355–361
He J, Xu Q, Jing Y, Agani F, Qian X, Carpenter R, Li Q, Wang XR, Peiper SS, Lu Z (2012) Reactive oxygen species regulate ERBB2 and ERBB3 expression via miR-199a/125b and DNA methylation. EMBO Rep 13:1116–1122
Ren C, Xie R, Yao Y, Yu M, Chang F, Xing L, Zhang Y, Liu Y, Wang S, Farooque M (2019) MiR-125b suppression inhibits apoptosis and negatively regulates sema4d in avian leukosis virus-transformed cells. Viruses 11:728
Chen Y, Zhao Y, Jin W, Li Y, Zhang Y, Ma X, Sun G, Han R, Tian Y, Li H (2019) MicroRNAs and their regulatory networks in Chinese Gushi chicken abdominal adipose tissue during postnatal late development. BMC Genomics 20:778
Moriya-Sato A, Hida A, Inagawa-Ogashiwa M, Wada MR, Sugiyama K, Shimizu J, Yabuki T, Seyama Y, Hashimoto N (2000) Novel acyl-CoA synthetase in adrenoleukodystrophy target tissues. Biochem Biophys Res Commun 279:62–68
Steinberg SJ, Morgenthaler J, Heinzer AK, Smith KD, Watkins PA (2000) Very long-chain acyl-CoA synthetases. human “bubblegum” represents a new family of proteins capable of activating very long-chain fatty acids. J Biol Chem 275:35162–35169
Chen K, He H, Xie Y, Zhao L, Zhao S, Wan X, Yang W, Mo Z (2015) miR-125a-3p and miR-483-5p promote adipogenesis via suppressing the RhoA/ROCK1/ERK1/2 pathway in multiple symmetric lipomatosis. Sci Rep 5:11909
Fajas L (2003) Adipogenesis: a cross-talk between cell proliferation and cell differentiation. Ann Med 35:79–85
Lefterova MI, Lazar MA (2009) New developments in adipogenesis. Trends Endocrinol Metab 20:107–114
Huang J, Jiang D, Zhao S, Wang A (2019) Propranolol suppresses infantile hemangioma cell proliferation and promotes apoptosis by upregulating miR-125b expression. Anti-Cancer Drugs 30:501–507
Liu LH, Li H, Li JP, Zhong H, Zhang HC, Chen J, Xiao T (2011) miR-125b suppresses the proliferation and migration of osteosarcoma cells through down-regulation of STAT3. Biochem Biophys Res Commun 416:31–38
Xu N, Brodin P, Wei T, Meisgen F, Eidsmo L, Nagy N, Kemeny L, Ståhle M, Sonkoly E, Pivarcsi A (2011) MiR-125b, a microRNA downregulated in psoriasis, modulates keratinocyte proliferation by targeting FGFR2. J Invest Dermatol 131:1521–1529
Sun X, Li M, Sun Y, Cai H, Lan X, Huang Y, Bai Y, Qi X, Chen H (2016) The developmental transcriptome sequencing of bovine skeletal muscle reveals a long noncoding RNA, lncMD, promotes muscle differentiation by sponging miR-125b. Biochim Biophys Acta 1863:2835–2845
Cui Y, Xiao Z, Han J, Sun J, Ding W, Zhao Y, Chen B, Li X, Dai J (2012) MiR-125b orchestrates cell proliferation, differentiation and migration in neural stem/progenitor cells by targeting Nestin. BMC Neurosci 13:116
Wahid F, Shehzad A, Khan T, Kim YY (2010) MicroRNAs: synthesis, mechanism, function, and recent clinical trials. Biochim Biophys Acta 1803:1231–1243
Ren J, Jin P, Wang E, Marincola FM, Stroncek DF (2009) MicroRNA and gene expression patterns in the differentiation of human embryonic stem cells. J Transl Med 7:20
Xin F, Li M, Balch C, Thomson M, Fan M, Liu Y, Hammond SM, Kim S, Nephew KP (2008) Computational analysis of microRNA profiles and their target genes suggests significant involvement in breast cancer antiestrogen resistance. Bioinformatics 25:430–434
Lal A, Navarro F, Maher CA, Maliszewski LE, Yan N, O'Day E, Chowdhury D, Dykxhoorn DM, Tsai P, Hofmann O (2009) miR-24 inhibits cell proliferation by targeting E2F2, MYC, and other cell-cycle genes via binding to “seedless” 3′UTR microRNA recognition elements. Mol Cell 35:610–625
Kim YJ, Hwang SH, Cho HH, Shin KK, Bae YC, Jung JS (2012) MicroRNA 21 regulates the proliferation of human adipose tissue-derived mesenchymal stem cells and high-fat diet-induced obesity alters microRNA 21 expression in white adipose tissues. J Cell Physiol 227:183–193
Lopes-Marques M, Machado AM, Ruivo R, Fonseca E, Carvalho E, Castro LFC (2018) Expansion, retention and loss in the Acyl-CoA synthetase “Bubblegum”(Acsbg) gene family in vertebrate history. Gene 664:111–118
Watkins PA, Maiguel D, Jia Z, Pevsner J (2007) Evidence for 26 distinct acyl-coenzyme A synthetase genes in the human genome. J Lipid Res 48:2736–2750
Pei Z, Oey NA, Zuidervaart MM, Jia Z, Li Y, Steinberg SJ, Smith KD, Watkins PA (2003) The acyl-CoA synthetase “bubblegum” (lipidosin): further characterization and role in neuronal fatty acid beta-oxidation. J Biol Chem 278:47070–47078
Guo L, Cui H, Zhao G, Liu R, Li Q, Zheng M, Guo Y, Wen J (2018) Intramuscular preadipocytes impede differentiation and promote lipid deposition of muscle satellite cells in chickens. BMC Genomics 19:838
D’Andre HC, Paul W, Shen X, Jia X, Zhang R, Sun L, Zhang X (2013) Identification and characterization of genes that control fat deposition in chickens. J Anim Sci Biotechnol 4:43
Qin Y, Zhao J, Min X, Wang M, Luo W, Wu D, Yan Q, Li J, Wu X, Zhang J (2014) MicroRNA-125b inhibits lens epithelial cell apoptosis by targeting p53 in age-related cataract. Biochim Biophys Acta 1842:2439–2447
Wu N, Xiao L, Zhao X, Zhao J, Wang J, Wang F, Cao S, Lin X (2012) miR-125b regulates the proliferation of glioblastoma stem cells by targeting E2F2. FEBS Lett 586:3831–3839
Krek A, Grün D, Poy MN, Wolf R, Rosenberg L, Epstein EJ, MacMenamin P, Da Piedade I, Gunsalus KC, Stoffel M (2005) Combinatorial microRNA target predictions. Nat Genet 37:495
Funding
This study was funded by a grant from the National Natural Science Foundation of China (Nos. 32072692 and 31572356), the Program for Innovation Research Team of Ministry of Education (IRT16R23) and the Scientific Studio of Zhongyuan Scholars (No. 30601985).
Author information
Authors and Affiliations
Contributions
Conceptualization, G.L.; data curation, Y.L. and G.S.; formal analysis, W.J.; funding acquisition, G.L., Y.T., and X.K.; investigation, G.L., Y.C., W.J., and B.Z.; resources, H.L. and X.K.; software, Z.L. and W.L.; validation, Y.Z. and B.Z.; visualization, Y.C.; writing—original draft, S.F.; writing—review and editing, G.L. and Y.C.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Informed Consent
We confirm that this manuscript has not been published elsewhere and all authors agree to publish this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, G., Chen, Y., Jin, W. et al. Effects of miR-125b-5p on Preadipocyte Proliferation and Differentiation in Chicken. Mol Biol Rep 48, 491–502 (2021). https://doi.org/10.1007/s11033-020-06080-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-06080-4