Abstract
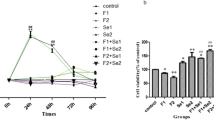
Prolonged and excessive fluoride exposure can lead to fluorosis. The kidney is one of the organs that are injured mostly due to fluoride-induced damage. Fluoride can induce DNA damage at cytotoxic concentrations. This study aims to determine the extent of NaF-induced DNA damage and to investigate the effect of vitamin E and selenium combination (ES) in preventing and repairing this damage. For this purpose, we administered different combinations of NaF and ES to NRK-52E cells and determined the effective concentrations of ES and the NaF IC50 values associated with different incubation times (3, 12, and 24 h) by using the MTT assay. The determined quantities of NaF IC50 in association with time and the NaF IC50 + ES combination were administered to the cells. The extent of DNA damage was determined with the comet assay and the expression levels of the Ku70/80 and PARP-1 genes were determined with the RT-qPCR method. DNA damage significantly increased in all experimental groups compared to the control group (p < 0.05). It was found out that the NaF and ES combination statistically reduced the DNA damage compared to the damage observed in the NaF-treated groups (p < 0.05). Treatment of the ES combination significantly increased the expressions of Ku70 and Ku80 genes involved in DNA repair (p < 0.05). We concluded that vitamin E and selenium can potentially be effective in the repair of fluoride-induced DNA damage based on the results of this in vitro study. Our results may shed light on the prevention of DNA damage associated with fluorosis.








Similar content being viewed by others
References
Anuradha CD, Kanno S, Hirano S (2001) Oxidative damage to mitochondria is a preliminary step to caspase-3 activation in fluoride-induced apoptosis in HL-60 cells. Free Radical Biol Med 1:367–373
Bai C, Chen T, Cui Y, Gong T, Peng X, Cui HM (2010) Effect of high fluorine on the cell cycle and apoptosis of renal cells in chickens. Biol Trace Elem Res 138:173–80
Barbier O, Arreola-Mendoza L, Del Razo LM (2010) Molecular mechanisms of fluoride toxicity. Chem Biol Interact 188:319–333
Ge Y, Chen L, Yin Z, Song X, Ruan T, Hua L, Liu J, Wang J, Ning H (2018) Fluoride-induced alterations of synapse-related proteins in the cerebral cortex of ICR offspring mouse brain. Chemosphere 201:874–883
Liu L, Zhang Y, Gu H, Zhang K, Ma L (2015) Fluorosis induces endoplasmic reticulum stress and apoptosis in osteoblasts in vivo. Biol Trace Elem Res 164(1):64–71
Xu H, Hu LS, Chang M, Jing L, Zhang XY, Li GS (2005) Proteomic analysis of kidney in fluoride-treated rat. Toxicol Lett 160:69–75
Song GH, Huang FB, Gao JP, Liu ML, Pang WB, Wb Li, Yan XY, Huo MJ, Yang X (2015) Effects of fluoride on DNA damage and caspase-mediated apoptosis in the liver of rats. Biol Trace Elem Res 166(2):173–82. https://doi.org/10.1007/s12011-015-0265-z
Yüksek V, Çetin S, Usta A, Kömüroğlu AU, Dede S (2017b) Effect of some vitamins on antioxidant/prooxidant parameters in sodium fluoride (NaF)-treated cell line (hFOB 1.19). Turkish J Veter Res 1(1):1-6.
Guney M, Oral B, Demirin H, Karahan N, Mungan T, Delibas N (2007) Protective effects of vitamins C and E against endometrial damage and oxidative stress in fluoride intoxication. Clin Exp Pharmacol Physiol 34(5–6):467–474
Yüksek V, Dede S, Taşpınar M, Çetin S (2017) The effects of certain vitamins on apoptosis and dna damage in sodium fluoride (NaF) administered renal and osteoblast cell lines. Fluoride 50(3):300–313
Hartlerode AJ, Scully R (2009) Mechanisms of double-strand break repair in somatic mammalian cells.Biochem J 423(2):157-68.
Fell VL, Schild-Poulter C (2012) Ku regulates signaling to DNA damage response pathways through the Ku70 von Willebrand A domain. Mol Cell Biol 32(1):76–87. https://doi.org/10.1128/MCB.05661-11
Chaitanya GV, Steven AJ, Babu PP (2010) PARP-1 cleavage fragments: signatures of cell-death proteases in neurodegeneration. Cell Commun Signal 8:31. https://doi.org/10.1186/1478-811X-8-31
Chlubek D (2003) Fluoride and oxidative stress. Fluoride 36:217–228
Chaiprasongsuk A, Janjetovic Z, Kim TK, Jarrett SG, D'Orazio JA, Holick MF, Tang EKY, Tuckey RC, Panich U, Li W, Slominski AT (2019) Protective effects of novel derivatives of vitamin D3 and lumisterol against UVB-induced damage in human keratinocytes involve activation of Nrf2 and p53 defense mechanisms. Redox Biol 24:101206. https://doi.org/10.1016/j.redox.2019.101206
Wimalawansa SJ (2019) Vitamin D deficiency: effects on oxidative stress, epigenetics, gene regulation, and aging. Biology (Basel) 8(2):E30. https://doi.org/10.3390/biology8020030
McDowell LR, Williams SN, Hidiroglou N et al (1996) Vitamin E supplementation for the ruminant. Anim Feed Sci Technol 60:273–296
Altiner A, Atalay H, Bilal T (2017) Vitamin E as an antioxidant. Balıkesir Health Sci J 6(3):149–157
Çetin S, Yur F, Taşpinar M, Yüksek V (2019) The effects of some minerals on apoptosis and DNA damage in sodium fluoride-administered renal and osteoblast cell lines. Fluoride 52(3):362–378
Boutet E, Trouche D, Canitrot Y 2013 Neutral Comet Assay. Bio-protocol , Bio-protocol LCC, 2013, 3 (18). hal-01188318
Afanasieva K, Sivolob A (2018) Physical principles and new applications of comet assay. Biophys Chem 238:1–7. https://doi.org/10.1016/j.bpc.2018.04.003
García O, Romero I, González JE, Moreno DL, Cuétara E, Rivero Y, Gutiérrez A, Pérez CL, Alvarez A, Carnesolta D, Guevara I (2011) Visual estimation of the percentage of DNA in the tail in the comet assay: evaluation of different approaches in an intercomparison exercise. Mutat Res 720(1–2):14–21
Chomczynski P, Mackey K (1995) Modification of the TRI reagent procedure for isolation of RNA from polysaccharide- and proteoglycan-rich sources. Biotechniques 19(6):942–945
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT. Methods 25(4):402–408
Mullenix PJ, Denbesten PK, Schunior A, Kernan WJ (1995) Neurotoxicity of sodium fluoride in rats Neurotoxicol. Teratol 17(2):169–177
Liu YJ, Guan ZZ, Gao Q, Pei JJ (2011) Increased level of apoptosis in rat brains and SH- SY5Y cells exposed to excessive fluoride a mechanism connected with activating JNK phosphorylation. Toxicol Lett 204(2–3):183–189. https://doi.org/10.1016/j.toxlet.2011.04.030
Efe U, Dede S, Yüksek V, Çetin S (2020) Apoptotic and oxidative mechanisms in liver and kidney tissues of sheep with fluorosis. Biol Trace Element Res. https://doi.org/10.1007/s12011-020-02121-y
Chattopadhyay A, Podder S, Agarwal S, Bhattacharya S (2011) Fluoride-induced histopathology and synthesis of stress protein in liver and kidney of mice. Arch Toxicol 85(4):327–35. https://doi.org/10.1007/s00204-010-0588-7
Song GH, Gao JP, Wang CF, Chen CY, Yan XY, Guo M, Wang Y, Huang FB (2014) Sodium fluoride induces apoptosis in the kidney of rats through caspase mediated pathways and DNA damage. J Physiol Biochem 70(3):857–868
Dec K, Łukomska A, Maciejewska D, Jakubczyk K, Baranowska-Bosiacka I, Chlubek D, Wąsik A, Gutowska I (2017) The influence of fluorine on the disturbances of homeostasis in the central nervous system. Biol Trace Elem Res 177(2):224–234. https://doi.org/10.1007/s12011-016-0871-4
Xiong X, Liu J, He W, Xia T, He P, Chen X, Yang K, Wang A (2007) Dose-effect relationship between drinking water fluoride levels and damage to liver and kidney functions in children. Environ Res 103(1):112–116
He LF, Chen JG (2006) DNA damage, apoptosis and cell cycle changes induced by fluoride in rat oral mucosal cells and hepatocytes. World J Gastroentero 12(7):1144–1148
Zhang M, Wang A, Xia T, He P (2008) Effects of fluoride on DNA damage, S-phase cell-cycle arrest and the expression of NF-kappaB in primary cultured rat hippocampal neurons. Toxicol Lett 179(1):1–5. https://doi.org/10.1016/j.toxlet.2008.03.002
He H, Wang H, Jiao Y, Ma C, Zhang H, Zhou Z (2015) Effect of sodium fluoride on the proliferation and gene differential expression in human RPMI8226 Cells. Biol Trace Elem Res 167(1):11–7
Otsuki S, Morshed SR, Chowdhury SA, Takayama F, Satoh T, Hashimoto K, Sugiyama K, Amano O, Yasui T, Yokote Y, Akahane K, Sakagami H (2005) Possible link between glycolysis and apoptosis induced by sodium fluoride. J Dent Res 84(10):919–23
Yan X, Yang X, Hao X, Ren Q, Gao J, Wang Y, Chang N, Qiu Y, Song G (2015) Sodium fluoride induces apoptosis in h9c2 cardiomyocytes by altering mitochondrial membrane potential and intracellular ROS level. Biol Trace Elem Res 166(2):210–215. https://doi.org/10.1007/s12011-015-0273-z
Pal S, Sarkar C (2014) Protective effect of resveratrol on fluoride induced alteration in protein and nucleic acid metabolism, DNA damage and biogenic amines in rat brain. Environ Toxicol Pharmacol 38:684–699. https://doi.org/10.1016/j.etap.2014.07.009
Yüksek V, Dede S, Usta A, Çetın S, Taşpınar M (2020) DNA damage-induced by sodium flouride(NaF) and the effect of cholecalciferol. Biocell 44(2):263–268
Lee JH, Jung JY, Jeong YJ, Park JH, Yang KH, Choi NK, Kim SH (2008) Kim WJ (2008) Involvement of both mitochondrial- and death receptor-dependent apoptotic pathways regulated by Bcl-2 family in sodium fluoride-induced apoptosis of the human gingival fibroblasts. Toxicology 243(3):340–7
Manivannan J, Sinha S, Ghosh M, Mukherjee A (2013) Evaluation of multi-endpoint assay to detect genotoxicity and oxidative stress in mice exposed to sodium fluoride. Mutat Res 751(1):59–65. https://doi.org/10.1016/j.mrgentox.2012.11.006
Do K, Chen AP (2013) Molecular pathways: targeting PARP in cancer treatment. Clin Cancer Res 19(5):977–84. https://doi.org/10.1158/1078-0432.CCR-12-0163
Huang Y, Shao Q, Luo X, Yang D, Zeng B, Xiang T, Ren G, Cheng Q (2018) Poly(ADP-ribose) polymerase-1 promotes recruitment of meiotic recombination-11 to chromatin and DNA double-strand break repair in Ku70-deficient breast cancer cells. FASEB J. 6:fj201800092R. https://doi.org/https://doi.org/10.1096/fj.201800092R
Nilov D, Maluchenko N, Kurgina T, Pushkarev S, Lys A, Kutuzov M, Gerasimova N, Feofanov A, Švedas V, Lavrik O, Studitsky VM (2020) Molecular mechanisms of PARP-1 inhibitor 7-methylguanine. Int J Mol Sci. https://doi.org/10.3390/ijms21062159
Urut F (2018) The Investigation of Cytotoxic Effect of Sodium Fluoride (NaF) in Renal Cell Line. Dissertation, Van Yüzüncü Yıl University
Ameeramja J, Panneerselvam L, Govindarajan V, Jeyachandran S, Baskaralingam V, Perumal E (2016) Tamarind seed coat ameliorates fluoride induced cytotoxicity, oxidative stress, mitochondrial dysfunction and apoptosis in A549 cells. J Hazard Mater 15(301):554–565. https://doi.org/10.1016/j.jhazmat.2015.09.037
Pramanik S, Saha D (2017) The genetic influence in fluorosis. Environ Toxicol Pharmacol 56:157–162
Daiwile AP, Tarale P, Sivanesan S, Naoghare PK, Bafana A, Parmar D, Kannan K (2019) Role of fluoride induced epigenetic alterations in the development of skeletal fluorosis. Ecotoxicol Environ Saf 169:410–417. https://doi.org/10.1016/j.ecoenv.2018.11.035
Wu CX, Wang YH, Li Y, Guan ZZ, Qi XL (2018) Changes of DNA repair gene methylation in blood of chronic fluorosis patients and rats. J Trace Elem Med Biol 50:223–228. https://doi.org/10.1016/j.jtemb.2018.07.010
Mikkelsen K, Apostolopoulos VB (2018) Vitamins and Ageing. Subcell Biochem 90:451–470. https://doi.org/10.1007/978-981-13-2835-0_15
Dugué PA, Bassett JK, Brinkman MT, Southey MC, Joo JE, Wong EM, Milne RL, English DR, Giles GG, Boussioutas A, Mitchell H, Hodge AM (2019) Dietary intake of nutrients involved in one-carbon metabolism and risk of gastric cancer: a prospective study. Nutr Cancer 71(4):605–614. https://doi.org/10.1080/01635581.2019.1577982
Lee EJ, Oh SY, Kim MK, Ahn SH, Son BH, Sung MK (2009) Modulatory effects of alpha- and gamma-tocopherols on 4-hydroxyestradiol induced oxidative stresses in MCF-10A breast epithelial cells. Nutr Res Pract 3(3):185–91. https://doi.org/10.4162/nrp.2009.3.3.185
Knaś M, Szajda SD, Snarska J, Zalewska-Szajda B, Walejko P, Borzym-Kluczyk M, Knaś-Karaszewska K, Kepka A, Chojnowska S, Waszkiewicz N, Zimnoch M, Maj J, Hryniewicka A, Dudzik D, Witkowshi S, Puchalski Z, Zwierz K (2009) Colon cancer releases alpha-tocopherol from its O-glycosides better than normal colon tissue. Hepatogastroenterology 56(90):339–342
Rocha KC, Vieira ML, Beltrame RL, Cartum J, Alves SI, Azzalis LA, Junqueira VB, Pereira EC, Fonseca FL (2016) Impact of selenium supplementation in neutropenia and immunoglobulin production in childhood cancer patients. J Med Food. 19(6):560–568. https://doi.org/10.1089/jmf.2015.0145
Favrot C, Beal D, Blouin E, Leccia MT, Roussel AM, Rachidi W (2018) Age-Dependent protective effect of selenium against UVA irradiation in primary human keratinocytes and the associated DNA repair signature. Oxid Med Cell Longev 2018:5895439. https://doi.org/10.1155/2018/5895439
Yildiz A, Kaya Y, Tanriverdi O (2019) effect of the interaction between selenium and zinc on dna repair in association with cancer prevention. J Cancer Prev 24(3):146–154. https://doi.org/10.15430/JCP.2019.24.3.146
Rusolo F, Capone F, Pasquale R, Angiolillo A, Colonna G, Castello G, Costantini M, Costantini S (2017) Comparison of the seleno-transcriptome expression between human non-cancerous mammary epithelial cells and two human breast cancer cell lines. Oncol Lett 13(4):2411–2417. https://doi.org/10.3892/ol.2017.5715
Zhu W, Cromie MM, Cai Q, Lv T, Singh K, Gao W (2014) Curcumin and vitamin E protect against adverse effects of benzo[a]pyrene in lung epithelial cells. PLoS One 9(3):e92992. https://doi.org/10.1371/journal.pone.0092992.eCollection
Ertilav K, Nazıroğlu M, Ataizi ZS, Braidy N (2019) Selenium enhances the apoptotic efficacy of docetaxel through activation of TRPM2 channel in DBTRG glioblastoma cells. Neurotox Res 35(4):797–808. https://doi.org/10.1007/s12640-019-0009-5
Xu T, Liu Y, Deng Y, Meng J, Li P, Xu X, Zeng J (2016) Insulin combined with selenium inhibit p38MAPK/CBP pathway and suppresses cardiomyocyte apoptosis in rats with diabetic cardiomyopathy. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 32(7):926–30
Pu X, Wang Z, Zhou S, Klaunig JE (2016) Protective effects of antioxidants on acrylonitrile-induced oxidative stress in female F344 rats. Environ Toxicol 31(12):1808–1818. https://doi.org/10.1002/tox.22182
Funding
This study was supported by a Grant from the Scientific Research Projects Presidency of Van Yüzüncü Yıl University (THD-2018-7148).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yüksek, V., Çetin, S. & Usta, A. The effect of vitamin E and selenium combination in repairing fluoride-induced DNA damage to NRK-52E cells. Mol Biol Rep 47, 7761–7770 (2020). https://doi.org/10.1007/s11033-020-05852-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05852-2