Abstract
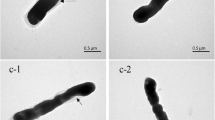
S-layer proteins in Lactic acid bacteria are not the only cell surface structures used for aggregation, but also plays significant role for intestinal tissue adhesion along with some other functional elements. In addition, it was determined that the properties of S-layer proteins differs not only between species but also the strains which belong to same species. In this work, presence and some functions of S-layer in lactic acid bacteria were determined, its effect on resistance to gastrointestinal enzymes, aggregation and adhesion ability were investigated as well. For this purpose S-layers of microorganisms were removed by 5 M LiCl treatment and size of the proteins were determined by SDS-PAGE analysis. The removal of S-layer proteins caused a change in the resistance of microorganisms to GIS enzymes. After the S-layer removal, two strains considerably lost their resistance to GIS enzymes. The strains mostly lost their aggregation ability in the absence of S-layer. The results showed that S-layer proteins are not the only structures involved in aggregation processes but, is a major mediator in Lactobacilli. Removal of S-layer had no effect on adhesion ability of W. cibaria DA28, the effect on L. casei DA4, L. coryniformis DA263 and L. plantarum DA140 was moderate, but the effect was high on L. plantarum DA100. The study showed that S-layer proteins play limited protection against GIS enzymes. In addition, absence of S-layer adversely affected aggregation and adhesion ability of strains.


Similar content being viewed by others
References
Vesterlund S, Paltta J, Karp M, Ouwehand A (2005) Adhesion of bacteria to resected human colonic tissue: quantitative analysis of bacterial adhesion and viability. Res Microbiol 156:238–244. https://doi.org/10.1016/j.resmic.2004.08.012
Yadav AK, Tyagi A, Kaushik JK, Saklani AC, Grover S, Batish VK (2013) Role of surface layer collagen binding protein from indigenous Lactobacillus plantarum 91 in adhesion and its anti-adhesion potential against gut pathogen. Microbiol Res 168:639–645. https://doi.org/10.1016/j.micres.2013.05.003
Alp D (2018) Investigation of some probiotic properties of lactic acid bacteria isolated from natural sources and determination of their ability to prevent pathogenic attachment in intestine model. Dissertation, Suleyman Demirel Universty
Harty DWS, Patrikakis M, Knox KW (1993) Identification of Lactobacillus strains isolated from patients with infective endocarditis and comparison of their surface-associated properties with those of other strains of the same species. Microbiol Ecolog Health Dis 6:191–201. https://doi.org/10.15739/IJAPR.030
McGrady JA, Butcher WG, Beighton D, Switalski LM (1995) Specific and charge interactions mediate collagen recognition by oral Lactobacilli. J Dent Res 74:649–657
Mukai T, Toba T, Ohori H (1996) Collagen binding of Bifidobacterium adolescentis. Curr Microbiol 34:326–331
Sara M, Sleytr UB (2000) S-layer proteins. J Bacteriol 182(4):859–868
Frece J, Kos B, Svetec IK, Zgaga Z, Mrsa V, Suskovic J (2005) Importance of s layer proteins in probiotic activity of lactobacillus acidophilus M92. J Appl Microbiol 98:285–292. https://doi.org/10.1111/j.1365-2672.2004.02473.x
Johnson B, Selle K, O’Flaherty S, Goh YJ, Klaenhammer T (2013) Identification of extracellular surface-layer associated proteins in Lactobacillus acidophilus NCFM. Microbiology 159:2269–2282. https://doi.org/10.1099/mic.0.070755-0
Polak-Berecka M, Wasko A, Paduch R (2014) The effect of cell surface components on adhesion ability of Lactobacillus rhamnosus. Antonie Leeuwenhoek 106:751–762. https://doi.org/10.1007/S10482-014-0245-X
Mobili P, Gerbino E, Tymczyszyn EE, Gómez-Zavaglia A (2010) S-layers in lactobacilli: structural characteristics and putative role in surface and probiotic properties of whole bacteria. Appl Microbiol Microb Biotechnol 22:1224–1234
Hynönen U, Palva A (2013) Lactobacillus surface layer proteins: structure, function and applications. Appl Microbiol Biotechnol. 97:5225–5243. https://doi.org/10.1007/s00253-013-4962-2
Wang H, Wei CX, Min L, Zhu LY (2018) Good or bad: gut bacteria in human health and diseases. Biotechnol Biotechnol Equip. https://doi.org/10.3389/fmicb.2018.02858
Goh JY, Klaenhammer TR (2010) Functional roles of aggregation-promoting- like factor in stress tolerance and adherence of Lactobacillus acidophilus NCFM. Appl Environ Microbiol 76(15):5005–5012. https://doi.org/10.1128/AEM.00030-10
Ossowski I, Satokari R, Reunanen J, Lebeer S, Keersmaecker S, Vanderleyden J, Vos WM, Palva A (2011) Functional characterization of a mucus-specific LPXTG surface adhesin from probiotic Lactobacillus rhamnosus GG. Appl Environ Microbiol 77(13):4465–4472. https://doi.org/10.1128/AEM.01958-09
Denkova R, Strinska H, Denkova Z (2014) Study on the adhesion of lactobacillus plantarum strains with probıotic properties to MDCK. J Fac Food Eng 13(3):214–217
Wasko A, Polak-Berecka M, Kuzdralinski A, Skrzypek T (2014) Variability of S-layer proteins in Lactobacillus helveticus strains. Anaerobe 25:53–60. https://doi.org/10.1016/j.anaerobe.2013.11.004
Lortal S, Heıjenoorkta J, Ruber R, Sleytr UB (1992) Slayer Of Lactobacillus helveticus ATCC 12046: isolation, chemical characterization and re-formation after extraction with lithium chloride. J General Microbiol 138:611–618
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Maragkoudakis PA, Zoumpopoulou G, Miaris C (2006) Probiotic potential of Lactobacillus strains isolated from dairy products. Int Dairy J 16:189–199
Tokatlı M, Gülgör G, Elmacı S, İşleyen N, Özçelik F (2015) In vitro properties of potential probiotic indigenous lactic acid bacteria originating from traditional pickles. BioMed Res Int 215:1–8: https://doi.org/10.1155/2015/315819
Kawther ELS, Tawfik NF, Dabiza MA, Sharaf OM, Effat BA (2010) In vitro assessment of gastrointestinal viability of potentially probiotic Lactobacilli. J American Sci 6:11–15
Eryılmaz F (2011) Identifıcation of potential probiotic properties of some lactic acid bacterial genera which is isolated from vaginal secretion. Dissertation University Of Ankara
Kos B, Suskovic J, Vukovic S, Simpraga M, Frece J, Matosic S (2003) Adhesion and aggregation ability of probiotic strain Lactobacillus acidophilus M92. J Appl Microbiol 94:981–987. https://doi.org/10.1046/j.1365-2672.2003.01915.x
Collado MC, Meriluoto J, Salminen S (2008) Adhesion and aggregation properties of probiotic and pathogen strains. Eur Food Res Technol 226:1065–1073. https://doi.org/10.1007/s00217-007-0632-x
Evans EM, Wrigglesworth JM, Burdett K, Pover WFR (1971) Studies on epithelial cells isolated from guinea pig small intestine. J Cell Biol 51:452–464
Sellwood R, Gibbons RA, Jones GW (1975) Adhesion of enteropathogenic Escherichia coli to pig intestinal brush borders: the existence of two pig phenotypes. J Med Microbiol 8:405–411
Ouwehand AC, Salminen S (2003) In vitro adhesion assays for probiotics and their in vivo relevance: a review. Microb Ecol Health Dis 15:175–184. https://doi.org/10.1080/08910600310019886
Kučan M, Gobin I, Merkov K, Jurčıć-Momčılovıć D, Frece J (2012) Testing the adhesion and colonization ability of Lactobacillus plantarum strain S1 to the mice intestinal epithelium. Int J Sanitary 6:25–30
Kang MS, Piao M, Shin BA, Lee HC, Oh JS (2006) Adhesion of Weissella cibaria to the epithelial cells and factors affecting its adhesion. J Bacteriol Virol 36:151–157
Schachtsiek M, Hammes WP, Hertel C (2004) Characterization Of Lactobacillus coryniformis DSM 20001T surface protein cpf mediating coaggregation with and aggregation among pathogens. Appl Environ Microbiol 70:7078–7085
Ristl R, Steiner K, Zarschler K, Zayni S, Messner P, Sch¨affer C (2011) The S-Layer Glycome-Adding to the Sugar Coat of Bacteria. Int J Microbiol 16 pages. doi: 10.1155/2011/127870.
Kang MS, Na HS, Oh JS (2005) Coaggregation ability of Weissella cibaria isolates with Fusobacterium nucleatum and their adhesiveness to epithelial cells. FEMS Microbiol Lett 253:323–329. https://doi.org/10.1016/j.femsle.2005.10.002
Yasui T, Yoda K, Kamiya T (1995) Analysis of S layer proteins of Lactobacillus brevis. FEMS Microbiol Lett 133:181–186
Sleytr UB, Schuster B, Egelseer EM, Pum D (2014) S-layers: principles and applications. FEMS Microbiol Rev 38:823–864. https://doi.org/10.1111/1574-6976.12063
De la Fuente-Núñez C, Mertens J, Smit J, Hancock REW (2012) The bacterial surface layer provides protection against antimicrobial peptides. Appl Environ Microbiol 78:5452–5456
Eslami N, Kermanshahi RK, Erfan M (2013) Studying the stability of s-layer protein of Lactobacillus Acidophilus ATCC 4356 in simulated gastrointestinal fluids using SDS-PAGE and circular dichroism. Iranian J Pharma Res 12:47–56
Meng J, Zhu X, Gao SM, Zhang QX, Sun Z, Lu RR (2014) Characterization of surface layer proteins and its role in probiotic properties of three Lactobacillus strains. Int J Biol Macromol 65:110–114. https://doi.org/10.1016/j.ijbiomac.2014.01.024
Beganovic J, Frece J, Kos B, Lebosˇ-Pavunc A, Habjanicˇ K, Suskovic J (2011) Functionality of the S-layer protein from the probiotic strain Lactobacillus helveticus M92. Antonie Leeuwenhoek 100:43–53. https://doi.org/10.1007/s10482-011-9563-4
Garrote GL, Delfederico L, Bibiloni R, Abraham AG, Perez PF, Semorile L, De Antoni GL (2004) Lactobacilli isolated from kefir grains: evidence of the presence of S-layer proteins. J Dairy Res 71:222–230. https://doi.org/10.1017/S0022029904000160
Li Q, Liu X, Dong M, Zhou J, Wang Y (2015) Aggregation and adhesion abilities of 18 lactic acid bacteria strains isolated from traditional fermented food. Int J Agricultural Policy Res 3:84–92
Chen X, Xu J, Shuai J, Chen J, Zhang Z, Fang W (2007) The S-layer proteins of Lactobacillus crispatus strain ZJ001 is responsible for competitive exclusion against Escherichia coli O157:H7 and Salmonella typhimurium. Int J Food Microbiol 115:307–312. https://doi.org/10.1016/j.ijfoodmicro.2006.11.007
Golowczyc MA, Mobili P, Garrote GL, Abraham AG, De Antoni GL (2007) Protective action of Lactobacillus kefir carrying S-layer protein against Salmonella enterica serovar Enteritidis. Int J Food Microbiol 118:264–273
Boris S, Suarez JE, Barbe's C, (1997) Characterization of the aggregation promoting factor from Lactobacillus gasseri, a vaginal isolate. J Appl Microbiol 83:413–420
Reniero R, Cocconcelli P, Bottazzi V, Morelli M (1992) High frequency of conjugation in Lactobacillus mediated by an aggregation-promoting factor. J General Microbiol 138:763–768
Ehrenfeld E, Kessler RE, Clewell DB (1986) Identification of pheromone-ınduced surface proteins in Streptococcus faecalis and evidence of a role for lipoteichoic acid in formation of mating aggregates. J Bacteriol 168(1):1–12
Aslım B, Onal D, Beyatlı Y (2007) Factors influencing autoaggregation and aggregation of Lactobacillus delbrueckii subsp. bulgaricus isolated from handmade yogurt. J Food Prot 70:223–227
Yüksekdağ ZN, Aslim B (2010) 9 Assessment of potential probiotic and starter properties of pediococcus spp. isolated from Turkish-type fermented sausages (sucuk). J Microbiol Biotechnol 20:161–168. https://doi.org/10.4014/jmb.0904.04019
Tareb R, Bernardeau M, Gueguen M, Vernoux JP (2013) In vitro characterization of aggregation and adhesion properties of viable and heat-killed forms of two probiotic Lactobacillus strains and interaction with foodborne zoonotic bacteria, especially Campylobacter jejuni. J Med Microbiol 62:637–649. https://doi.org/10.1099/jmm.0.049965-0
Alp D, Kuleaşan H (2019) Determination of excopolisaccharide production and cholesterol asiımilation abilities of lactic acid bacteria isolated from different sources. J Food 44(2):191–201. https://doi.org/10.15237/gida.GD18059
Carasi P, Ambrosis NM, De Antoni GL, Bressollier P, Urdaci MC, Serradell MA (2014) Adhesion properties of potentially probiotic Lactobacillus kefiri to gastrointestinal mucus. J Dairy Res 81:16–23. https://doi.org/10.1017/S0022029913000526
Zhang J, Gao J, Guo Y, Wu Z, Pan D (2017) Extraction of Lactobacillus acidophilus CICC 6074 S-layer proteins and their ability to inhibit enteropathogenic Escherichia coli. Curr Microbiol. https://doi.org/10.1007/s00284-017-1291-1
Author information
Authors and Affiliations
Contributions
DA: Designed study, conducted GIS enzyme resistance and aggregation tests, isolated the S-layer from strains, made adhesion ability test, isolated and prepared the S-layer proteins for SEM analysis and wrote the publication. HA: Wrote the manuscript, analyzed the data by statistical program, isolated the proteins by SDS-PAGE, prepared the S-layer proteins for SEM analysis. AKA: contributed to GIS enzyme resistance and aggregation tests and SDS-PAGE.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alp, D., Kuleaşan, H. & Korkut Altıntaş, A. The importance of the S-layer on the adhesion and aggregation ability of Lactic acid bacteria. Mol Biol Rep 47, 3449–3457 (2020). https://doi.org/10.1007/s11033-020-05430-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05430-6