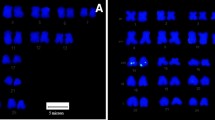
Abstract
Bacterial artificial chromosome (BAC) library is an important genomic resource useful in targeted marker development, positional cloning, physical mapping and a substrate for genome sequencing for better understanding the genome organization of a species. The present manuscript elucidates the improvement in protocols for economical and efficient BAC insert DNA isolation, BAC end sequencing and FISH for physical localization on the metaphase chromosome complements. BAC clones of Clarias magur, maintained in 384—well plate format in our laboratory, were used in this study. The protocols gave consistent and efficient results. We use routinely these protocols for BAC insert DNA extraction, generating end sequence data of the clone and constructing DNA probes to hybridize on the metaphase spreads of C. magur using FISH for physical their localization.




Similar content being viewed by others
References
Shizuya H, Birren B, Kim U, Mancino V, Slepak T, Tachiiri Y, Simon ML (1992) Cloning and stable maintenance of 300-kilobase-pair fragments of human DNA in Escherichia coli using an F-factor-based vector. Proc Natl Acad Sci USA 10:831–810
Stone NE, Fan JB, Willour V, Pennacchio LA, Warrington JA, de La AH, Chapelle A, Lehesjoki A, Cox DR, Myers RM (1996) Construction of a 750-kb bacterial clone contig and restriction map in the region of human chromosome 21 containing the progressive myoclonus epilepsy gene. Genome Res 6:218–225
Boysen C, Simon M, Hood L (1997) Analysis of the 1.1-Mb human α/δ T-cell receptor locus with bacterial artificial chromosome clones. Genome Res 7:330–338
Venter JC, Smith HO, Hood L (1996) A new strategy for genome sequencing. Nature 381:364–366
Poulsen TS, Johnsen HE (2004) BAC end sequencing. Methods Mol Biol 255:157–161
IUCN Red List of Threatened Species (2010) e.T168255A6470089. https://www.iucnredlist.org/species/168255/6470089. Accessed 23 April 2019.
Sambrook J (2001) Molecular cloning: a laboratory manual. In: Sambrook J, Russell DW (eds). Cold Spring Harbor Laboratory, Cold Spring Harbor, New York.
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain terminating inhibitors. Proc Natl Acad Sci USA 74:5463
Mathew CGP (1984) Radiolabeling of DNA by Nick Translation. In: Walker JM (ed) Nucleic acids. Methods in molecular biology, vol 2. Humana Press, Totowa.
Winterfeld G, Roser M (2007) Deposition of ribosomal DNAs in the chromosome of perennial oats (Poaceae: Aveneae). Bot J Linn Soc 155:193–210
Sinnett D, Richer C, Baccichet A (1998) Isolation of stable bacterial artificial chromosome DNA using a modified alkaline lysis method. Biotechniques 24:752–754
Li Y, Xu P, Zhao Z, Wang J, Zhang Y, Sun XW (2011) Construction and characterization of the BAC library for common carp Cyprinus carpio L. and establishment of microsynteny with zebrafish Danio rerio. Mar Biotechnol 13:706–712
Zhao S (2000) Human BAC Ends. Nucleic Acids Res 28:129–132
Krzywinski M, Bosdet I, Smailus D, Chiu R, Mathewson C, Wye N et al (2004) A set of BAC clones spanning the human genome. Nucleic Acids Res 32:3651–3660
Qiaoping Y, Feng L, Joseph H, Victoria Z, Maria-Ines B, Qb J, Rod W, Robin B (2000) Anchoring of rice BAC clones to the rice genetic map in silico. Nucleic Acids Res 28:3636–3641
Zhao S, Shatsman S, Ayodeji B, Geer K, Tsegaye G, Krol M et al (2001) Mouse BAC ends quality assessment and sequence analyses. Genome Res 11:1736–1745
Osoegawa B, Zhu CL, Shu T, Ren Q, Cao GM, Vessere MM et al (2004) BAC resources for the rat genome project. Genome Res 14:780–785
Cameron RA, Samanta M, Yuan A, He D, Davidson E (2009) SpBase: the sea urchin genome database and web site. Nucleic Acids Res 37:750–754
Fernandez-Pozo N, Menda N, Edwards JD, Saha S, Tecle IY, Strickler SR et al (2015) The Sol Genomics Network (SGN)—from genotype to phenotype to breeding. Nucleic Acids Res 43:1036–1041
Beier S, Himmelbach A, Colmsee C, Zhang XQ, Barrero RA, Zhang Q et al (2017) Construction of a map-based reference genome sequence for barley, Hordeum vulgare L. Sci Data 4:1–24
Chapus C, Edwards SV (2009) Genome evolution in Reptilia: in silico chicken mapping of 12,000 BAC-end sequences from two reptiles and a basal bird. BMC Genomics 14:10
Genet C, Dehais P, Palti Y, Gao G, Gavory F, Wincker P, Quillet E, Boussaha M (2011) Analysis of BAC-end sequences in rainbow trout: content characterization and assessment of synteny between trout and other fish genomes. BMC Genomics 12:314
Cao Y, Kimura S, Itoi T, Honda K, Ohtake H, Omasa T (2012) Fluorescence in situ hybridization using bacterial artificial chromosome (BAC) clones for the analysis of chromosome rearrangement in Chinese hamster ovary cells. Methods 56:418–423
Acknowledgements
Authors are highly thankful to the Director, ICAR-National Bureau of Fish Genetic Resources, Lucknow, for infrastructure support and encouragement for taking up the work. The authors are also thankful to the CABin Scheme of ICAR, implemented through CABin Division of ICAR-IASRI, New Delhi, for financial support of the project in which the work was undertaken.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is a no any financial or non-financial conflict of interest among the authors for publication of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kumar, R., Baisvar, V.S., Kushwaha, B. et al. Improved protocols for BAC insert DNA isolation, BAC end sequencing and FISH for construction of BAC based physical map of genes on the chromosomes. Mol Biol Rep 47, 2405–2413 (2020). https://doi.org/10.1007/s11033-020-05283-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05283-z