Abstract
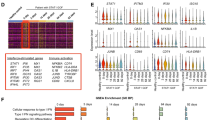
Sirt1, also known as the longevity gene, is an NAD+-dependent class III histone deacetylase that has been extensively studied in multiple areas of research including cellular metabolism, longevity, cancer, autoimmunity, and immunity. However, little is known about the function of Sirt1 in B cells. This study aimed to investigate the role of Sirt1 in the expression pattern of mRNAs in the resting B cells of mice. CD19+ B cell-specific inducible Sirt1 knockout (KO) mice were divided into tamoxifen-treated Sirt1 KO group (S19T) or control group (S19). mRNAs extracted from resting B cells of both groups were analyzed for differentially expressed genes (DEG) using microarray. DEG analysis showed significant differential expression of 20 genes, of which Hspa1a and Hspa1b showed the highest fold change (FC) in S19T compared with S19 (p value < 0.01 and FC > 3). Further, Kyoto Encyclopedia of Genes and Genomes analysis identified pathways associated with diseases, organismal systems, and antigen processing and presentation. Additionally, the pathways known to involve Hspa1a and Hspa1b were also activated in the S19T group. On the other hand, after in vitro stimulation with lipopolysaccharide, cell viability and IgM production were significantly decreased in Sirt1 KO B cells, while expressions of TNF-α, IL-6, and IL-10 were increased. In summary, our study reveals that Sirt1 may maintain the quiescent state in resting B cells by suppressing the increase of Hspa1a and Hspa1b. This work provides a foundation for further studies on the functional roles of Sirt1 in B cells.





Similar content being viewed by others
References
Kaeberlein M, McVey M, Guarente L (1999) The SIR2/3/4 complex and SIR2 alone promote longevity in Saccharomyces cerevisiae by two different mechanisms. Genes Dev 13(19):2570–2580
Haigis MC, Sinclair DA (2010) Mammalian sirtuins: biological insights and disease relevance. Ann Rev Pathol 5:253–295
Yang H, Bi Y, Xue L, Wang J, Lu Y, Zhang Z, Chen X, Chu Y, Yang R, Wang R (2015) Multifaceted modulation of SIRT1 in cancer and inflammation. Crit Rev Oncog 20(1–2):49–64
Li G, Xia Z, Liu Y, Meng F, Wu X, Fang Y, Zhang C, Liu D (2018) SIRT1 inhibits rheumatoid arthritis fibroblast-like synoviocyte aggressiveness and inflammatory response via suppressing NF-κB pathway. Biosci Rep 1:1–10. https://doi.org/10.1042/BSR20180541
Zhu X, Chu H, Jiang S, Liu Q, Liu L, Xue Y, Zheng S, Wan W, Qiu J, Wang J (2017) Sirt1 ameliorates systemic sclerosis by targeting the mTOR pathway. J Dermatol Sci 87(2):149–158
Chen X, Lu Y, Zhang Z, Wang J, Yang H, Liu G (2015) Intercellular interplay between Sirt1 signalling and cell metabolism in immune cell biology. Immunology 145(4):455–467
Zhang J, Lee S-M, Shannon S, Gao B, Chen W, Chen A, Divekar R, McBurney MW, Braley-Mullen H, Zaghouani H (2009) The type III histone deacetylase Sirt1 is essential for maintenance of T cell tolerance in mice. J Clin Investig 119(10):3048–3058
Owczarczyk AB, Schaller MA, Reed M, Rasky AJ, Lombard DB, Lukacs NW (2015) Sirtuin 1 regulates dendritic cell activation and autophagy during respiratory syncytial virus–induced immune responses. J Immunol 195:1637–1646
Alvarez Y, Rodríguez M, Municio C, Hugo E, Alonso S, Ibarrola N, Fernández N, Crespo MS (2012) Sirtuin 1 is a key regulator of the IL-12 p70/IL-23 balance in human dendritic cells. J Biol Chem M112:391839
Zhang Z, Xu J, Liu Y, Wang T, Pei J, Cheng L, Hao D, Zhao X, Chen H-Z, Liu D-P (2018) Mouse macrophage specific knockout of SIRT1 influences macrophage polarization and promotes angiotensin II-induced abdominal aortic aneurysm formation. J Genet Genomics 45(1):25–32
Goodnow CC, Vinuesa CG, Randall KL, Mackay F, Brink R (2010) Control systems and decision making for antibody production. Nat Immunol 11(8):681
Li R, Rezk A, Healy LM, Muirhead G, Prat A, Gommerman JL, Bar-Or A (2016) Cytokine-defined B cell responses as therapeutic targets in multiple sclerosis. Front Immunol 6:626
Molnarfi N, Schulze-Topphoff U, Weber MS, Patarroyo JC, Prod’homme T, Varrin-Doyer M, Shetty A, Linington C, Slavin AJ, Hidalgo J (2013) MHC class II–dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies. J Exp Med 210(13):2921–2937
Tangye SG, Brink R, Goodnow CC, Phan TG (2015) SnapShot: interactions between B cells and T cells. Cell 162(4):926–926.e921
Tangye SG, Ma CS, Brink R, Deenick EK (2013) The good, the bad and the ugly—T FH cells in human health and disease. Nat Rev Immunol 13(6):412
Ashwell JD, Defranco AL, Paul WE, Schwartz RH (1984) Antigen presentation by resting B cells: Radiosensitivity of the antigen-presentation function and two distinct pathways of T cell activation. J Exp Med 159(3):881–905
Airoldi I, Raffaghello L, Cocco C, Guglielmino R, Roncella S, Fedeli F, Gambini C, Pistoia V (2004) Heterogeneous expression of interleukin-18 and its receptor in B-cell lymphoproliferative disorders deriving from naive, germinal center, and memory B lymphocytes. Clin Cancer Res 10(1):144–154
Tellier J, Nutt SL (2018) Plasma cells: The programming of an antibody secreting machine. Eur J Immunol 49:30–37
Wang Q, Yan C, Xin M, Han L, Zhang Y, Sun M (2017) Sirtuin 1 (Sirt1) overexpression in BaF3 cells contributes to cell proliferation promotion, apoptosis resistance and pro-inflammatory cytokine production. Med Sci Monit 23:1477
Li H, Rajendran GK, Liu N, Ware C, Rubin BP, Gu Y (2007) SirT1 modulates the estrogen–insulin-like growth factor-1 signaling for postnatal development of mammary gland in mice. Breast Cancer Res 9(1):R1
Yasuda T, Wirtz T, Zhang B, Wunderlich T, Schmidt-Supprian M, Sommermann T, Rajewsky K (2013) Studying Epstein–Barr virus pathologies and immune surveillance by reconstructing EBV infection in mice. In: Cold Spring Harbor symposia on quantitative biology. Cold Spring Harbor Laboratory Press, p a020222
Melchers F, Braun V, Galanos C (1975) The lipoprotein of the outer membrane of Escherichia coli: a B-lymphocyte mitogen. J Exp Med 142(2):473–482
Fillatreau S (2018) B cells and their cytokine activities implications in human diseases. Clin Immunol 186:26–31
Aufricht C (2005) Heat-shock protein 70: molecular supertool? Pediatr Nephrol 20(6):707–713
Daugaard M, Rohde M, Jäättelä M (2007) The heat shock protein 70 family: highly homologous proteins with overlapping and distinct functions. FEBS Lett 581(19):3702–3710
Gothard LQ, Ruffner ME, Woodward JG, Park-Sarge O-K, Sarge KD (2003) Lowered temperature set point for activation of the cellular stress response in T-lymphocytes. J Biol Chem 278(11):9322–9326
Peng C-W, Zhao B, Chen H-C, Chou M-L, Lai C-Y, Lin S-Z, Hsu H-Y, Kieff E (2007) Hsp72 up-regulates Epstein-Barr virus EBNALP coactivation with EBNA2. Blood J 109(12):5447–5454
Chesnut RW, Grey H (1981) Studies on the capacity of B cells to serve as antigen-presenting cells. J Immunol 126(3):1075–1079
Mellman I, Steinman RM (2001) Dendritic cells: specialized and regulated antigen processing machines. Cell 106(3):255–258
Raposo G, Nijman HW, Stoorvogel W, Liejendekker R, Harding CV, Melief C, Geuze HJ (1996) B lymphocytes secrete antigen-presenting vesicles. J Exp Med 183(3):1161–1172
Nimmervoll B, Chtcheglova LA, Juhasz K, Cremades N, Aprile FA, Sonnleitner A, Hinterdorfer P, Vigh L, Preiner J, Balogi Z (2015) Cell surface localised Hsp70 is a cancer specific regulator of clathrin-independent endocytosis. FEBS Lett 589(19):2747–2753
Bendz H, Ruhland SC, Pandya MJ, Hainzl O, Riegelsberger S, Braüchle C, Mayer MP, Buchner J, Issels RD, Noessner E (2007) Human heat shock protein 70 enhances tumor antigen presentation through complex formation and intracellular antigen delivery without innate immune signaling. J Biol Chem 282(43):31688–31702
Williams JW, Elvington A, Kessler S, Wohltmann M, Wu GF, Randolph GJ (2019) B Cell-Mediated antigen presentation through mhc Class II Is dispensable for atherosclerosis progression. ImmunoHorizons 3(1):37–44
Mycko MP, Cwiklinska H, Szymanski J, Szymanska B, Kudla G, Kilianek L, Odyniec A, Brosnan CF, Selmaj KW (2004) Inducible heat shock protein 70 promotes myelin autoantigen presentation by the HLA class II. J Immunol 172(1):202–213
Raman M, Chen W, Cobb M (2007) Differential regulation and properties of MAPKs. Oncogene 26(22):3100
Gong X, Luo T, Deng P, Liu Z, Xiu J, Shi H, Jiang Y (2012) Stress-induced interaction between p38 MAPK and HSP70. Biochem Biophys Res Commun 425(2):357–362
Ogasawara T, Yasuyama M, Kawauchi K (2003) Constitutive activation of extracellular signal-regulated kinase and p38 mitogen-activated protein kinase in B-cell lymphoproliferative disorders. Int J Hematol 77(4):364–370
Feng J, Liu S, Ma S, Zhao J, Zhang W, Qi W, Cao P, Wang Z, Lei W (2014) Protective effects of resveratrol on postmenopausal osteoporosis: regulation of SIRT1-NF-κB signaling pathway. Acta Biochim Biophys Sin 46(12):1024–1033
Schug TT, Xu Q, Gao H, Peres-da-Silva A, Draper DW, Fessler MB, Purushotham A, Li X (2010) Myeloid deletion of SIRT1 induces inflammatory signaling in response to environmental stress. Mol Cell Biol 30(19):4712–4721
Mosser DD, Morimoto RI (2004) Molecular chaperones and the stress of oncogenesis. Oncogene 23(16):2907
Beere HM, Wolf BB, Cain K, Mosser DD, Mahboubi A, Kuwana T, Tailor P, Morimoto RI, Cohen GM, Green DR (2000) Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome. Nat Cell Biol 2(8):469
Frezzato F, Accordi B, Trimarco V, Gattazzo C, Martini V, Milani G, Bresolin S, Severin F, Visentin A, Basso G (2016) Profiling B cell chronic lymphocytic leukemia by reverse phase protein array: focus on apoptotic proteins. J Leukoc Biol 100(5):1061–1070
Kim JY, Yenari MA (2013) The immune modulating properties of the heat shock proteins after brain injury. Anat Cell Biol 46(1):1–7
Kim JY, Han Y, Lee JE, Yenari MA (2018) The 70-kDa heat shock protein (Hsp70) as a therapeutic target for stroke. Expert Opin Ther Targets 22(3):191–199
Cangeri Di Naso F, Rosa Porto R, Sarubbi Fillmann H, Maggioni L, Vontobel Padoin A, Jacques Ramos R, Corá Mottin C, Bittencourt A, Anair Possa Marroni N, Homem Ivo, de Bittencourt Jr P (2015) Obesity depresses the anti-inflammatory HSP 70 pathway, contributing to NAFLD progression. Obesity 23(1):120–129
Park J-M, Han Y-M, Lee J-S, Ko KH, Hong S-P, Kim E-H, Hahm K-B (2015) Nrf2-mediated mucoprotective and anti-inflammatory actions of Artemisia extracts led to attenuate stress related mucosal damages. J Clin Biochem Nutr 56(2):132–142
Asea A, Kraeft S-K, Kurt-Jones EA, Stevenson MA, Chen LB, Finberg RW, Koo GC, Calderwood SK (2000) HSP70 stimulates cytokine production through a CD14-dependant pathway, demonstrating its dual role as a chaperone and cytokine. Nat Med 6(4):435
Wang Y, Kelly CG, Singh M, McGowan EG, Carrara A-S, Bergmeier LA, Lehner T (2002) Stimulation of Th1-polarizing cytokines, CC chemokines, maturation of dendritic cells, and adjuvant function by the peptide binding fragment of heat shock protein 70. J Immunol 169(5):2422–2429
Agrawal S, Gupta S (2011) TLR1/2, TLR7, and TLR9 signals directly activate human peripheral blood naive and memory B cell subsets to produce cytokines, chemokines, and hematopoietic growth factors. J Clin Immunol 31(1):89–98
Pistoia V (1997) Production of cytokines by human B cells in health and disease. Immunol Today 18(7):343–350
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation, the Ministry of Education, Korea (NRF-2017R1A6A1A03015713) and, in part, by the Konyang University Myunggok Research Fund of 2015-04.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Han, Y., Kang, Y., Yu, J. et al. Increase of Hspa1a and Hspa1b genes in the resting B cells of Sirt1 knockout mice. Mol Biol Rep 46, 4225–4234 (2019). https://doi.org/10.1007/s11033-019-04876-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-019-04876-7