Abstract
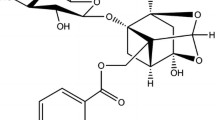
A combination of fluorescence, UV–Vis absorption, circular dichroism (CD), Fourier transform infrared (FT-IR) and molecular modeling approaches were employed to determine the interaction between lysionotin and bovine serum albumin (BSA) at physiological pH. The fluorescence titration suggested that the fluorescence quenching of BSA by lysionotin was a static procedure. The binding constant at 298 K was in the order of 105 L mol−1, indicating that a high affinity existed between lysionotin and BSA. The thermodynamic parameters obtained at different temperatures (292, 298, 304 and 310 K) showed that the binding process was primarily driven by hydrogen bond and van der Waals forces, as the values of the enthalpy change (ΔH°) and entropy change (ΔS°) were found to be −40.81 ± 0.08 kJ mol−1 and −35.93 ± 0.27 J mol−1 K−1, respectively. The surface hydrophobicity of BSA increased upon interaction with lysionotin. The site markers competitive experiments revealed that the binding site of lysionotin was in the sub-domain IIA (site I) of BSA. Furthermore, the molecular docking results corroborated the binding site and clarified the specific binding mode. The results of UV–Vis absorption, CD and FT-IR spectra demonstrated that the secondary structure of BSA was altered in the presence of lysionotin.








Similar content being viewed by others
References
Curry S, Mandelkow H, Brick P, Franks N (1996) Crystal structure of human serum albumin complexed with fatty acid reveals an asymmetric distribution of binding sites. Nat Struct Biol 5:827–835
Zhang YP, Peng M, Liu LL, Shi SY, Peng S (2012) Screening, identification, and potential interaction of active compounds from Eucommia ulmodies leaves binding with bovine serum albumin. J Agric Food Chem 60:3119–3125
Skrt M, Benedik E, Podlipnik C, Ulrih NP (2012) Interactions of different polyphenols with bovine serum albumin using fluorescence quenching and molecular docking. Food Chem 135:2418–2424
Zhang YP, Shi SY, Chen XQ, Zhang W, Huang KL, Peng MJ (2011) Investigation on the interaction between ilaprazole and bovine serum albumin without or with different C-ring flavonoids from the viewpoint of food–drug interference. J Agric Food Chem 59:8499–8506
Guo XJ, Han XW, Tong J, Guo C, Yang WF, Zhu JF, Fu B (2010) The investigation of the interaction between piracetam and bovine serum albumin by spectroscopic methods. J Mol Struct 966:129–135
Suksamrarn A, Poomsing P, Aroonrerk N, Punjanon T, Suksamrarn S, Kongkun S (2003) Antimycobacterial and antioxidant flavones from Limnophila geoffrayi. Arch Pharmcol Res 26:816–820
Chen JW, Zhu ZQ, Hu TX, Zhu DY (2002) Structure–activity relationship of natural flavonoids in hydroxyl radical-scavenging effects. Acta Pharmcol Sin 23:667–672
Bi SY, Yan LL, Pang B, Wang Y (2012) Investigation of three flavonoids binding to bovine serum albumin using molecular fluorescence technique. J Lumin 132:132–140
Feroz SR, Mohamad SB, Bujang N, Malek SNA, Tayyab S (2012) Multispectroscopic and molecular modeling approach to investigate the interaction of flavokawain B with human serum albumin. J Agric Food Chem 60:5899–5908
Zhang GW, Ma YD, Wang L, Zhang YP, Zhou J (2012) Multispectroscopic studies on the interaction of maltol, a food additive, with bovine serum albumin. Food Chem 133:264–270
Ghosh S, Jana S, Guchhait N (2012) Domain specific association of small fluorescent probe trans-3-(4-monomethylaminophenyl)-acrylonitrile (MMAPA) with bovine serum albumin (BSA) and its dissociation from protein binding sites by Ag nanoparticles: spectroscopic and molecular docking study. J Phys Chem 116:1155–1163
Zhang GW, Zhao N, Wang L (2010) Probing the binding of vitexin to human serum albumin by multispectroscopic techniques. J Lumin 131:880–887
Möller M, Denicola A (2002) Study of protein–ligand binding by fluorescence. Biochem Mol Biol Educ 30:309–312
Coi A, Bianucci AM, Bonomi F, Rasmussen P, Mura GM, Ganadu ML (2008) Structural perturbation of aB-crystallin by zinc and temperature related to its chaperone-like activity. Int J Biol Macromol 42:229–234
Yuan T, Weljie AM, Vogel HJ (1998) Tryptophan fluorescence quenching by methionine and selenomethionine residues of calmodulin: orientation of peptide and protein binding. Biochemistry 37:3187–3195
Hemmateenejad B, Shamsipur M, Samari F, Khayamian T (2012) Combined fluorescence spectroscopy and molecular modeling studies on the interaction between harmalol and human serum albumin. J Pharm Biomed Anal 67–68:201–208
Lakowicz JR (2006) Principles of fluorescence spectroscopy, 3rd edn. Springer, New York
Zhang GW, Zhao N, Hu X, Tian J (2010) Interaction of alpinetin with bovine serum albumin: probing of the mechanism and binding site by spectroscopic methods. Spectrochim Acta A 76:410–417
Sudlow G, Birkett DJ, Wade DN (1976) Further characterization of specific drug binding sites on human serum albumin. Mol Pharmcol 12:1052–1061
Sjoholm I, Ekman B, Kober A, Ljungstedt-Pahlman I, Seiving B, Sjodin T (1979) Binding of drugs to human serum albumin: XI. The specificity of three binding sites as studied with albumin immobilized in microparticles. Mol Pharmacol 16:767–777
Zhang GW, Wang L, Fu P, Hu MM (2011) Mechanism and conformational studies of farrerol binding to bovine serum albumin by spectroscopic methods. Spectrochim Acta A 82:424–431
Ross PD, Subramanian S (1981) Thermodynamics of protein association reactions: forces contributing to stability. Biochemistry 20:3096–3102
Qi ZD, Zhou B, Xiao Q, Chuan S, Liu Y, Dai J (2008) Interaction of rofecoxib with human serum albumin: determination of binding constants and the binding site by spectroscopic methods. J Photochem Photobiol A 193:81–88
Föster T, Sinanoglu O (1966) Modern quantum chemistry. Academic Press, New York
Shahabadi N, Maghsudi M, Kiani Z, Pourfoulad M (2011) Multispectroscopic studies on the interaction of 2-tert-butylhydroquinone (TBHQ), a food additive, with bovine serum albumin. Food Chem 124:1063–1068
Mehrabi M, Ghobadi S, Khodarahmi RJ (2009) Spectroscopic study on the interaction of celecoxib with human carbonic anhydrase II: thermodynamic characterization of the binding process. J Photochem Photobiol B 97:161–168
Polet H, Steinhardt J (1968) Binding-induced alterations in ultraviolet absorption of native serum albumin. Biochemistry 7:1348–1356
Li DJ, Zhu M, Xu C, Ji BM (2011) Characterization of the baicalein–bovine serum albumin complex without or with Cu2+ or Fe3+ by spectroscopic approaches. Eur J Med Chem 46:588–599
He WY, Li Y, Tang JH, Luan F, Jin J, Hu ZD (2006) Comparison of the characterization on binding of alpinetin and cardamonin to lysozyme by spectroscopic methods. Int J Biol Macromol 39:165–173
Madaeni SS, Rostami E (2008) Spectroscopic investigation of the interaction of BSA with cationic surfactants. Chem Eng Technol 31:1265–1271
Shahabadi N, Maghsudi M, Rouhani S (2012) Study on the interaction of food colourant quinoline yellow with bovine serum albumin by spectroscopic techniques. Food Chem 135:1836–1841
Sandhya B, Hegde AH, Kalanur SS, Katrahalli U, Seetharamappa J (2011) Interaction of triprolidine hydrochloride with serum albumins: thermodynamic and binding characteristics, and influence of site probes. J Pharm Biomed Anal 54:1180–1186
Khan SN, Islam B, Yennamalli R, Sulta A, Subbarao N, Khan AU (2008) Interaction of mitoxantrone with human serum albumin: spectroscopic and molecular modeling studies. Eur J Pharm Sci 35:371–382
Liu XH, Xi PX, Chen FJ, Xu ZH, Zeng ZZ (2008) Spectroscopic studies on binding of 1-phenyl-3-(coumarin-6-yl)-sulfonylurea to bovine serum albumin. J Photochem Photobiol B 92:98–102
Zhang GW, Wang L, Pan JH (2012) Probing the binding of the flavonoid diosmetin to human serum albumin by multispectroscopic techniques. J Agric Food Chem 60:2721–2729
Acknowledgments
We gratefully acknowledge the financial support of the National Natural Science Foundation of China (Nos. 21167013 and 31060210), the Natural Science Foundation of Jiangxi Province (20114BAB204019), and the Research Program of State Key Laboratory of Food Science and Technology of Nanchang University (Nos. SKLF-ZZB-201305, SKLF-ZZA-201302 and SKLF-KF-201203).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, Y., Zhang, G. & Yan, J. Detection of interaction between lysionotin and bovine serum albumin using spectroscopic techniques combined with molecular modeling. Mol Biol Rep 41, 1693–1702 (2014). https://doi.org/10.1007/s11033-013-3018-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-3018-0